Photoinduced energy and electron transfer in fullerene–oligophenyleneethynylene systems: dependence on the substituents of the oligomer unit
Abstract
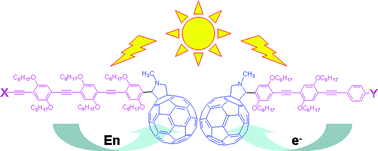
The photophysical properties of
* Corresponding authors
a
Molecular Photoscience Group, Istituto per la Sintesi Organica e la Fotoreattività, Consiglio Nazionale delle Ricerche, Via Gobetti 101, Bologna, Italy
E-mail:
armaroli@isof.cnr.it
Fax: +39 051 6399844
Tel: +39 051 6399820
b
Groupe de Chimie des Fullerènes et des Systèmes Conjugués, Laboratoire de Chimie de Coordination du CNRS, 205 route de Narbonne, Toulouse Cedex 4, France
E-mail:
jfnierengarten@lcc-toulouse.fr
Fax: +33 (0) 5 61 55 30 03
Tel: +33 (0) 5 61 33 31 00
The photophysical properties of

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
J. N. Clifford, T. Gu, J. Nierengarten and N. Armaroli, Photochem. Photobiol. Sci., 2006, 5, 1165 DOI: 10.1039/B611321D
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
