Synthesis, characterization and catalytic reactivity of ruthenium nanoparticles stabilized by chiral N-donor ligands
Abstract
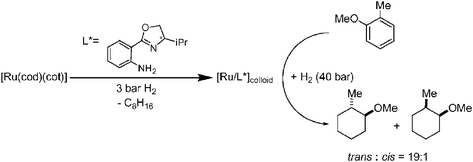
The decomposition of the organometallic precursor [Ru(cod)(cot)] (cod = 1,5-cyclooctadiene; cot = 1,3,5-cyclooctatriene) under mild conditions (room temperature, 3 bars H2) and in the presence of optically pure ligands, L*, namely (R)-2-aminobutanol 1, amino(oxazolines) (2, 3), hydroxy(oxazoline) (4) and bis(oxazolines) (5–8), leads to stable ruthenium nanoparticles exhibiting a mean diameter between 1.6–2.5 nm. These nanoparticles can be isolated and re-dispersed. They display different mean sizes, shapes and dispersions depending on the stabilizer nature. These new colloids (Ru1–Ru18) have been characterized by both solid state and molecular chemistry techniques, including TEM/HRTEM, WAXS, elemental analysis, and IR and NMR spectroscopy. To further characterize the surface state of these particles, their catalytic behaviour has been examined in the reduction of organic prochiral unsaturated substrates. Although the asymmetric induction obtained is modest, it reveals the influence of the asymmetric ligand coordinated at the surface of the particles.

 Please wait while we load your content...
Please wait while we load your content...