Polymorphs of a pyrazole nitronyl nitroxide and its complexes with metal(ii) hexafluoroacetylacetonates†
Abstract
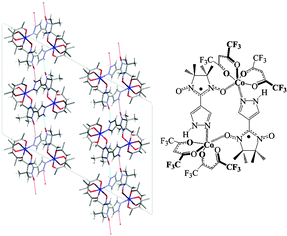
The synthesis of pyrazol-4-yl nitronyl nitroxide is reported, along with the crystal structures of its polymorphs and coordination compounds with nickel(II) and cobalt(II) hexafluoroacetylacetonate. The polymorphic forms of the pure radical show very different magnetic properties: the alpha form shows only antiferromagnetic interactions while those of the beta form reveal competing ferro- and anti-ferromagnetic interactions. This phenomenon can be traced to the relative orientations of the spin carriers in the crystal. These, in turn, are determined in large measure by the hydrogen bonding networks, where a competition between [N–H⋯O] and [N–H⋯N] hydrogen bonds is observed. This delicate balance is also seen in the coordination chemistry of the radical acting as a

 Please wait while we load your content...
Please wait while we load your content...