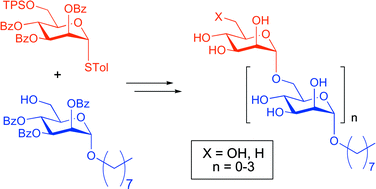
Mycobacterium tuberculosis is the cause of the deadly human disease tuberculosis. In studies over the last 40 years it has been revealed that this organism possesses a complex cell wall including glycophospholipids such as the phosphatidylinositiol mannosides (PIMs), lipomannan (LM) and lipoarabinomannan (LAM). These glycolipids all contain a common α-1,6-linked mannoside core, and the higher PIMs and LAM possess α-1,2-linked mannosyl residues. It has been shown that simple α-1,6-linked oligomannosides can act as substrates for α-1,6-mannosyltransferases in mycobacteria. Here we report a simple iterative synthesis of a series of hydrophobic octyl α-1,6-linked oligomannosides from mono- through to tetrasaccharides. We have utilized a single thioglycoside donor and alcohol acceptor. Further, we have developed conditions for the conversion of each of these compounds to the 6-deoxy congeners. Deoxygenation of the 6-position of the terminal mannosyl residue should prevent these compounds acting as substrates for the abundant α-1,6-mannosyltransferases in mycobacteria and should permit detection of the elusive α-1,2-mannosyltransferase activity responsible for elaboration of LM to mature LAM and the biosynthesis of the higher PIMs.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...