A chiral molecular recognition approach to the formation of optically active quaternary centres in aza-Henry reactions
Abstract
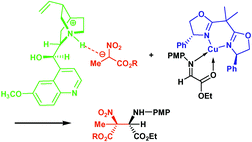
An approach to asymmetric catalysis based on chiral molecular recognition by the combination of chiral Lewis acids and chiral organocatalysis for the formation of optically active quarternary centers in the aza-Henry reaction is presented; this procedure leads to products with up to 98% ee and a diastereomeric ratio of 14 : 1 in excellent yields with catalyst loadings of 5 mol%.

 Please wait while we load your content...
Please wait while we load your content...