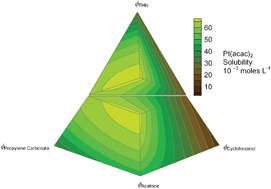
The solubility of platinum(II) acetylacetonate (Pt(acac)2) in various solvent mixtures of acetone, cyclohexanol, propylene carbonate and 1,2,3,4-tetrahydronaphthalene (THN) have been determined at 25 °C and 1 atm, using thermogravimetric analysis (TGA) and inductively coupled plasma mass spectrometry (ICP-MS). The densities of the saturated solutions were determined by pycnometry. The entropy of fusion and melting point of Pt(acac)2 were determined by differential scanning calorimetry (DSC) to be 41.4 J mol−1 K−1 and 512.5 K, respectively. From this data, the ideal mole fraction at saturation (xideal) of Pt(acac)2 was calculated to be 0.0278, which is more than twice the highest reported solubility of that compound. In this effort, mixtures of acetone, propylene carbonate and THN exhibit cosolvency for Pt(acac)2, with the greatest measured molar solubility of Csat
= 0.0675 mol L−1 occurring at the 1 ∶ 1 ∶ 1 v ∶ v ∶ v ratio of these three solvents. A new equation was developed for activity coefficients in solvent mixtures and used to predict ln γ for Pt(acac)2 to within 3.6% of the measured values. The concept of Intercomponent Mixture Parameters (IMPs) is introduced in relation to Scheffé mixture polynomials as applied to the prediction of activity coefficients in solvent mixtures.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...