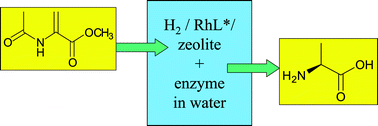
The growing awareness of the pressing need for greener, more sustainable technologies has focused attention on the use of atom efficient catalytic methodologies for the manufacture of fine chemicals and pharmaceuticals. Another aspect which is receiving increasing attention is the use of alternative reaction media that circumvent the problems associated with many of the traditional volatile organic solvents. The use of nonconventional reaction media also provides opportunities for facilitating the recovery and recycling of the catalyst. The state of the art in the use of alternative reaction media for green, sustainable organic synthesis is reviewed. Liquid–liquid biphasic catalysis provides an industrially attractive method for the recovery and recycling of catalysts as an alternative to the more traditional solid heterogeneous catalysts. Various approaches to liquid–liquid biphasic catalysis—aqueous biphasic, fluorous biphasic, supercritical carbon dioxide, ionic liquids and various combinations thereof—are reviewed and compared. “The best solvent is no solvent” but if a solvent is needed then water has a lot to recommend it and catalysis in aqueous biphasic systems is an industrially attractive methodology which has found broad application. Similarly, supercritical carbon dioxide is an interesting reaction medium in the context of green chemistry and catalysis in various mono- and biphasic systems involving this solvent are reviewed. Fluorous biphasic systems and ionic liquids also have advantages in certain situations and the advantages and limitations of these media are compared. The ultimate in clean catalytic technologies is to telescope multistep syntheses into one-pot in the form of catalytic cascade processes. Examples of such catalytic cascades involving both chemo- and biocatalytic conversions are presented. Biocatalysis has a distinct advantage in this context in that the reactions all take place at or close to ambient temperature and pressure. In emulation of natural processes, where several different enzymes are compartmentalised in the cell, it can be advantageous to immobilise the various catalysts in such a cascade process. In this context, a novel and effective method for the immobilisation of enzymes as cross-linked enzyme aggregates (CLEAs) is discussed and the use of a combi CLEA, containing two enzymes, for the one-pot conversion of benzaldehyde to S-mandelic acid is reported.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...