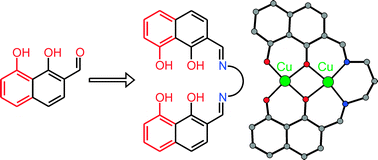
A facile synthesis of 2-formyl-1,8-naphthalenediol is reported. Its potential as a general precursor for the preparation of unsymmetrical multidentate chelating ligand systems based on 1,8-naphthalenediol is demonstrated by the synthesis of the dinucleating ligand L4−
(H4L
=
N,N′-bis(2-(1,8-naphthalenediol)methylidene)propylenediamine). Reaction of H4L with copper acetate results in the formation of the unsymmetrical dinuclear CuII complex [LCu2]
(3), which has been structurally characterized by single-crystal X-ray diffraction. One CuII ion is coordinated by a N2O2 compartment of L4− and the other CuII ion is coordinated by an O4 compartment of L4− while they are bridged by two aryloxide functions of L4−. A dimerization of two molecules of 3 to a tetranuclear entity 32 occurs through formation of weak apical Cu–O interactions. Analysis of the temperature dependent magnetic susceptibility measurements (2–290 K) established a strong intradimer exchange coupling J12
=
−371 cm−1. This strong superexchange interaction fits nicely in a magneto-structural correlation which has been established for dinuclear bis(phenoxide)-bridged CuII complexes demonstrating the electronic equivalence of the aryloxides of a phenol and 1,8-naphthalenediol.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...