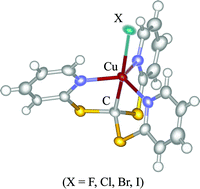
A complete series of copper(II) halide complexes [CuX(tptm)]
(X = F (1), Cl (2), Br (3), I (4); tptm = tris(2-pyridylthio)methyl) with a novel CuII–C(sp3) bond has been prepared by the reactions of [Cu(tptm)(CH3CN)]PF6
(5·PF6) with corresponding halide sources of KF or n-Bu4NX (X = Cl, Br, I), and the trigonal bipyramidal structures have been confirmed by X-ray crystallography and/or EPR spectroscopy. The iodide complex 4 easily liberates the iodide anion in acetonitrile forming the acetonitrile complex 5 as a result. The EPR spectra of the complexes showed several superhyperfine structures that strongly indicated the presence of spin density on the halide ligands through the Cu–X bond. The results of DFT calculations essentially matched with the X-ray crystallographic and the EPR spectroscopic results. Cyclic voltammetry revealed a quasi-reversible reduction wave for CuII/CuI indicating a trigonal pyramidal coordination for CuI states. A coincidence of the redox potential for all [CuX(tptm)]0/+ processes indicates that the main oxidation site in each complex is the tptm ligand.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...