Rotational spectra and structure of the Ar2–H2S complex: pulsed nozzle Fourier transform microwave spectroscopic and ab initio studies
Abstract
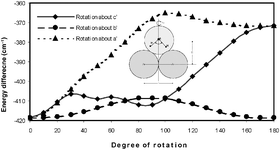
This paper reports the rotational spectrum and structure of the Ar2–H2S complex and its HDS and D2S isotopomers. The ground state structure has heavy-atom C2v symmetry with the two Ar atoms indistinguishable and H2S freely rotating as evinced by the fact that asymmetric top energy levels with Kp = odd levels are missing. The rotational constants for the parent isotopomer are: A = 1733.115(1) MHz, B = 1617.6160(5) MHz and C = 830.2951(2) MHz. Unlike the Ar–H2S complex, the Ar2–H2S does not show an anomalous isotopic shift in rotational constants on deuterium substitution. However, the intermolecular potential is still quite floppy, leading to very different centrifugal distortion constants for the three isotopomers. The Ar–Ar and Ar–c.m.(H2S) distances are determined to be 3.820 Å and 4.105 Å, respectively. The A rotational constants for Ar2–H2S/HDS/D2S isotopomers are very close to each other and to the B constant of free Ar2, indicating that H2S does not contribute to the moment of inertia about the a-axis. Ab initio calculations at MP2 level with aug-cc-pVQZ basis set lead to an equilibrium C2v minimum structure with the Ar–Ar line perpendicular to the H–H line and the S away from Ar2. The centrifugal distortion constants, calculated using the ab initio force field, are in reasonable agreement with the experimental values. However, they do not show the variation observed for different isotopmers. The binding energy of Ar2–H2S has been determined to be 507 cm−1 (6.0 kJ mol−1) by CBS extrapolation after correcting for basis set superposition error. Potential energy scans point out that the barrier for internal rotation of H2S about its b axis is only 10 cm−1 and it is below the zero point energy (13.5 cm−1) in this torsional degree of freedom. Internal rotation of H2S about its a- and c-axes also have small barriers of about 50 cm−1 only, suggesting that H2S is extremely floppy within the complex.

 Please wait while we load your content...
Please wait while we load your content...