Kinetics and products of the OH radical-initiated reaction of 3-methyl-2-butenal
Abstract
Kinetics and products of the gas-phase reaction of OH radicals with 3-methyl-2-butenal [(CH3)2C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHCHO] have been investigated at room temperature and atmospheric pressure of air. Using a relative rate method with methacrolein as the reference compound, a rate constant for the reaction of OH radicals with 3-methyl-2-butenal of (6.21 ± 0.18)
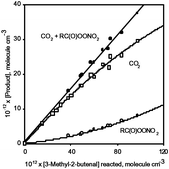
× 10−11 cm3 molecule−1 s−1 at 296 ± 2 K was measured, where the indicated error does not include the uncertainty in the rate constant for the methacrolein reference compound. Products of this reaction were investigated using in situ Fourier transform infrared (FT-IR) spectroscopy and solid phase microextraction (SPME) fibers coated with O-(2,3,4,5,6-pentafluorobenzyl)hydroxylamine for on-fiber derivatization of carbonyl compounds, with subsequent thermal desorption and analysis by gas chromatography. The products observed and the molar formation yields were: glyoxal, 40 ± 3%; acetone, 74 ± 6%; 2-hydroxy-2-methylpropanal, 4.6 ± 0.7%; CO2, 39% initially, decreasing to 30% at greater extents of reaction; peroxyacyl nitrate(s)
[RC(O)OONO2], 5–8%, increasing with the extent of reaction and with the sum of the CO2 and RC(O)OONO2 yields being 38 ± 6%; and organic nitrates [RONO2], 8.5 ± 2.3%. The formation of these products is readily explained by a reaction mechanism based on those previously formulated for the corresponding reactions of the α,β-unsaturated aldehydes acrolein, crotonaldehyde and methacrolein. Based on the mechanism proposed, at room temperature H-atom abstraction from the CHO group accounts for 40 ± 6% of the overall reaction, and OH radical addition to the carbon atoms of the C
CHCHO] have been investigated at room temperature and atmospheric pressure of air. Using a relative rate method with methacrolein as the reference compound, a rate constant for the reaction of OH radicals with 3-methyl-2-butenal of (6.21 ± 0.18)
× 10−11 cm3 molecule−1 s−1 at 296 ± 2 K was measured, where the indicated error does not include the uncertainty in the rate constant for the methacrolein reference compound. Products of this reaction were investigated using in situ Fourier transform infrared (FT-IR) spectroscopy and solid phase microextraction (SPME) fibers coated with O-(2,3,4,5,6-pentafluorobenzyl)hydroxylamine for on-fiber derivatization of carbonyl compounds, with subsequent thermal desorption and analysis by gas chromatography. The products observed and the molar formation yields were: glyoxal, 40 ± 3%; acetone, 74 ± 6%; 2-hydroxy-2-methylpropanal, 4.6 ± 0.7%; CO2, 39% initially, decreasing to 30% at greater extents of reaction; peroxyacyl nitrate(s)
[RC(O)OONO2], 5–8%, increasing with the extent of reaction and with the sum of the CO2 and RC(O)OONO2 yields being 38 ± 6%; and organic nitrates [RONO2], 8.5 ± 2.3%. The formation of these products is readily explained by a reaction mechanism based on those previously formulated for the corresponding reactions of the α,β-unsaturated aldehydes acrolein, crotonaldehyde and methacrolein. Based on the mechanism proposed, at room temperature H-atom abstraction from the CHO group accounts for 40 ± 6% of the overall reaction, and OH radical addition to the carbon atoms of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond accounts for 53 ± 4% of the overall reaction. Hence 93 ± 8% of the reaction products and pathways are accounted for.
C bond accounts for 53 ± 4% of the overall reaction. Hence 93 ± 8% of the reaction products and pathways are accounted for.

 Please wait while we load your content...
Please wait while we load your content...