Four novel polymeric coordination networks have been obtained through self-assembly processes involving alkoxo-bridged copper(II) species as nodes, and anionic cyano-complexes as linkers: ∞2[{Cu2(pa)2}{M(CN)2}2]
(M = Ag, 1; Au, 2), ∞3[{Cu4(mea)4}{Au(CN)2}4·H2O]
3, and ∞3[{Cu2(pa)2}{Ni(CN)4}]
4
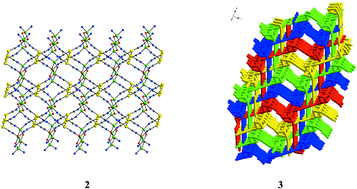
(pa = deprotonated propanolamine; mea = deprotonated monoethanolamine). The supramolecular architectures of compounds 1, 2 and 3 are sustained by argentophilic or strong aurophilic interactions. The solid-state architectures of 1 and 2, which are isomorphous, consist of infinite layers, constructed from binuclear alkoxo-bridged nodes and [M(CN)2]− spacers. The layers are stacked in an offset parallel mode, and are further interconnected through Ag⋯Ag or Au⋯Au contacts (1: Ag⋯Ag 3.015 Å; 2: Au⋯Au 3.069 Å). Compound 3 consists of unique fourfold interpenetrating diamondoid nets. The diamondoid topology is built of heterocubane {Cu4O4} nodes, which are connected by [Au(CN)2]− rods. The Cu–O distances within the {Cu4O4} node vary between 1.927(2) and 2.679(1)
Å, showing unsymmetric bridging of the copper atoms. Aurophilic interactions are established between the bridging and terminal [Au(CN)2]− metalloligands, and connect the interpenetrating nets, resulting in infinite chains of gold atoms (the Au⋯Au distances vary between 3.253 and 3.305 Å). Compound 4 exhibits a 3-D network constructed from {Cu2(pa)2]2+ nodes connected by square-planar [Ni(CN)4]2− ions. Compounds 1, 2 and 4 are weakly paramagnetic. The cryomagnetic investigation of 3 reveals a gradual increase, followed by a decrease of the χMT product, as the temperature is lowered. A superposition of ferro- (J1
=
+20.8 cm−1) and antiferromagnetic (J2
=
−6.4) interactions within the tetranuclear node was found. Antiferromagnetic interactions are established between the tetranuclear nodes (θ
=
−2.99 K).

 Please wait while we load your content...
Please wait while we load your content...