Asymmetric direct aldol reaction catalyzed by an l-prolinamide derivative: considerable improvement of the catalytic efficiency in the ionic liquid†
Abstract
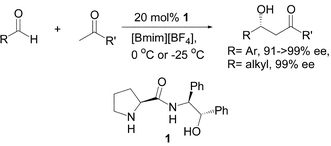
The asymmetric direct aldol reactions of a wide scope of aldehydes with unmodified ketones in the presence of 20 mol% (S,S,S)-pyrrolidine-2-carboxylic acid (2′-hydroxyl-1′,2′-diphenyl-ethyl)-amine (1) were performed in ionic liquids; aldol products with 91 to >99% ees for aromatic aldehydes and 99% ees for alphatic aldehydes were offered by the present procedure.

 Please wait while we load your content...
Please wait while we load your content...