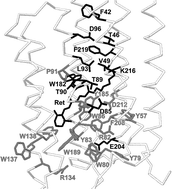
In the last decade, several genome sequencing projects revealed the existence of previously unknown photoreceptors. Among those are eukaryotic rhodopsins of haloarchaeal type, mostly represented by fungal sequences. We have classified and analyzed seventy-seven of these fungal proteins, which show a high similarity of their putative transmembrane regions to those of bacteriorhodopsin. Those sequences can be divided into the two subgroups, fungal rhodopsins (RDs) and opsin-related proteins (ORPs), the latter lacking the lysine residue necessary for retinal binding. We have analyzed the conservation pattern of the residues known to have functional or structural importance in bacteriorhodopsin and discussed dramatic differences in the conservation between RDs and ORPs. We found many cases of multiple forms of RDs and/or ORPs and examined possible reasons for such multiplicity. For some species the reason may lie in functional photobiological diversification, while for the others it follows the pattern of evolutionary recent genome duplication and possible functional redundancy.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...