Microwave assisted Leimgruber–Batcho reaction for the preparation of indoles, azaindoles and pyrroylquinolines
Abstract
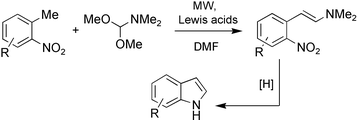
The development of enhanced conditions for Lewis acid catalysed Leimgruber–Batcho indole synthesis using microwave acceleration is described. This approach has permitted the preparation of a variety of heteroaromatic

 Please wait while we load your content...
Please wait while we load your content...