Covalent modification of vapour-grown carbon nanofibers via direct Friedel–Crafts acylation in polyphosphoric acid
Abstract
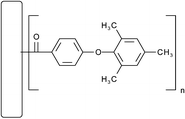
Electrophilic functionalization of vapour-grown carbon nanofibers (VGCNF) was accomplished via Friedel–Crafts acylation with 2,4,6-trimethylphenoxybenzoic acid in polyphosphoric acid using the improved conditions that we previously described. The progress of the reaction was conveniently monitored with FT-IR spectroscopy following the growth of the keto-carbonyl band at 1664 cm−1 associated with the product. In addition to scanning electron microscopic and UV-vis spectroscopic data, the combined results from the elemental analysis and thermogravimetric analysis further suggested that there were 3 arylcarbonyl groups covalently attached to the nanotube structure for every 100 carbon sites. Because of the presence of significant hydrogen content in the starting VGCNF, the covalent attachment of the arylcarbonyl groups most probably occurred at the sp2C–H sites.

 Please wait while we load your content...
Please wait while we load your content...