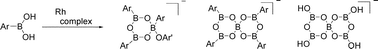
Ar–B(OH)2
(1a: Ar = C6H4OMe-4, 1b: Ar = C6H3Me2-2,6) react immediately with Rh(OC6H4Me-4)(PMe3)3
(2) in 5 ∶ 1 molar ratio at room temperature to generate [Rh(PMe3)4]+[B5O6Ar4]−
(3a: Ar = C6H4OMe-4, 3b: Ar = C6H3Me2-2,6). p-Cresol (92%/Rh), anisole (80%/Rh) and H2O (364%/Rh) are formed from 1a and 2. The reaction of 1a with 2 for 24 h produces [Rh(PMe3)4]+[B5O6(OH)4]
−
(4) as a yellow solid. This is attributed to hydrolytic dearylation of once formed 3a because the direct reaction of 3a with excess H2O forms 4. An equimolar reaction of 2 with phenylboroxine (PhBO)3 causes transfer of the 4-methylphenoxo ligand from rhodium to boron to produce [Rh(PMe3)4]+[B3O3Ph3(OC6H4Me-4)]−
(5). Arylboronic acids 1a and 1b react with Rh(OC6H4Me-4)(PR3)3
(6: R = Et, 8: R = Ph) and with Rh(OC6H4Me-4)(cod)(PR3)
(11: R =
iPr, 12: R = Ph) to form [Rh(PR3)4]+[B5O6Ar4]−
(7a: R = Et, Ar = C6H4OMe-4, 7b: R = Et, Ar = C6H3Me2-2,6, 9a: R = Ph, Ar = C6H3Me2-2,6) and [Rh(cod)(PR3)(L)]+[B5O6Ar4]−
(13b: R =
iPr, L = acetone, Ar = C6H3Me2-2,6, 14a: R = Ph, L = PPh3, Ar = C6H4OMe-4, 14b: R = Ph, L = PPh3, Ar = C6H3Me2-2,6), respectively. Hydrolysis of 14a yields [Rh(cod)(PPh3)2]+[B5O6(OH)4]−
(15) quantitatively.

 Please wait while we load your content...
Please wait while we load your content...