The purple photosynthetic bacteria contain a large variety of sensory and regulatory proteins, and those responding to light are among the most interesting. These currently include bacteriophytochrome (Bph), sensory rhodopsin (SR), and photoactive yellow protein (PYP), which all appear to function as light sensors. We herein interpret new findings within the context of current knowledge. For greater detail, the reader is referred to comprehensive reviews on these topics. Of the three proteins, only PYP has been well-characterized in terms of structure and physical-chemical properties in the purple bacteria, although none have well-defined functions. New findings include a cluster of six genes in the Thermochromatium tepidum genome that encodes presumed sensory rhodopsin and phototaxis proteins. T. tepidum also has a gene for PYP fused to bacteriophytochrome and diguanylate cyclase domains. The genes for PYP and its biosynthetic enzymes are associated with those for gas vesicle formation in Rhodobacter species, suggesting that one function of PYP is to regulate cell buoyancy. The association of bacteriophytochrome genes with those for reaction centers and light-harvesting proteins in Rhodopseudomonas palustris suggests that the photosynthetic antenna as well as the reaction center are regulated by Bphs. Furthermore, Rc. centenum PPR is reversibly photobleached at 702 nm rather than red-shifted as in other phytochromes, suggesting that PPR senses the intensity of white light rather than light quality. PYP from Halorhodospira
(aka Ectothiorhodospira)
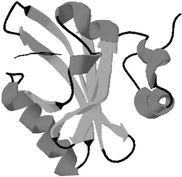
halophila is of special interest because it has become the structural prototype for the PAS domain, a motif that is found throughout the phylogenetic tree and which plays important roles in many signaling pathways. Thus, the structural and photochemical characterization of PYP, utilizing site-directed mutagenesis, provides insights into the mechanism of signal transduction.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...