Structure and bonding in the isoelectronic series CnHnP5−n+: is phosphorus a carbon copy?†
Abstract
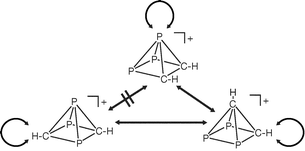
The relative stabilities of different isomers of the isoelectronic series CnHnP5−n+ have been investigated using G3X theory. The results indicate that all species containing one or more phosphorus atom adopt a three-dimensional nido geometry, in marked contrast to the planar structure favoured by the all-carbon analogue. Within isomeric nido clusters, a strong correlation between total energy and the nucleus-independent chemical shift (NICS) indicates that three-dimensional aromaticity plays a significant role in determining the stability of the cluster. In the context of these nido clusters, the extent to which phosphorus is a carbon copy proves to be highly dependent on the global electronic environment. The first isolobal substitution of CH by P causes a complete switch from localised to delocalised bonding, accompanied by a transition from a two- to a three-dimensional structure, with the phosphorus atom showing a strong preference for the unique apical site. In contrast, further increasing the phosphorus content causes no further change in structure or bonding, suggesting that, at the basal sites, phosphorus is a rather better carbon copy. The low-energy pathways for interconversion of apical and basal atoms previously identified in C2H2P3+ prove to be a general feature of all members of the series.

 Please wait while we load your content...
Please wait while we load your content...