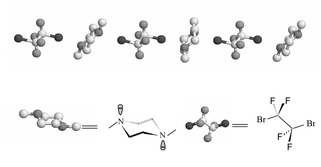
On the basis of our results, which concern sp3N⋯Br–Rf [Rf = per(poly)fluoroalkyl], sp3O⋯I–Rf, sp2O⋯I–Rf, sp3N⋯I–Rf, sp2N⋯I–Rf and interactions of sp2N⋯I–Rf with different substituents, a brief profile of the interactions between electronegative atoms (N, O, S) and halogen atoms (I, Br) of fluorine-containing alkylates is determined. The first example of an aliphatic fluorine-containing donor-acceptor supramolecule that is based on the N⋯Br–Rf interaction is reported in this paper. Our X-ray structure shows that 1,2-dibromotetrafluoroethane and 1,4-dimethylpiperazine alternately form endless chains depending on the N⋯Br–Rf interactions. The distance between Br and N is 2.864(3)
Å, which is considerably longer than the average covalent bond between N and Br, but it is also definitively shorter than the sum of the corresponding van der Waals radii of N and Br. From two other crystals, we successfully obtained precise data on the sp3O⋯X–Rf and sp2O⋯X–Rf interactions. Furthermore, an investigation of the substituent group effect is presented. We also report a valuable method to recrystallize and collect X-ray data of co-crystals that are easily disordered.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...