Synthesis and thermal behaviour of α,α′-didecyloligothiophenes
Abstract
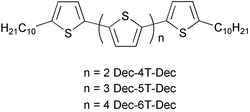
α,α′-Didecylquater-, -quinque- and -sexi-thiophenes were synthesized by Kumada cross-coupling and oxidative coupling reactions. For the former reaction Pd(dppf)Cl2 was found to be a more efficient catalyst than the usually applied Ni(dppp)Cl2. Thermal behaviour of all new oligothiophenes was investigated by differential scanning calorimetry and polarizing optical microscopy. It was shown that all these compounds possess not only crystal phases but also high temperature ordered smectic mesophases and that the clearing point increases linearly with the number of conjugated thiophene rings. A degree of order in the crystal phase was estimated on the basis of thermodynamic data. The highest degree of order was proposed for α,α′-didecylquaterthiophene, which explains why the mobility of end-α,α′-capped quaterthiophenes in FET (field effect transistor) devices is comparable or sometimes better than those of corresponding quinque- and sexi-thiophene derivatives.

 Please wait while we load your content...
Please wait while we load your content...