Production of H2O2 in CO2 and its use in the direct synthesis of propylene oxide†
Abstract
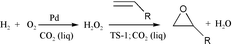
Hydrogen peroxide is generally considered to be a green oxidant, as it is relatively non-toxic and breaks down in the environment to non-toxic by-products. However, the current method for production, the sequential hydrogenation and oxidation of an alkyl anthraquinone, is capital-intensive, produces significant volumes of waste, and consumes sizeable quantities of energy during the purification and concentration of the product. We have designed CO2-soluble catalysts, and have successfully generated H2O2 from H2 and O2 in carbon dioxide–water mixtures. The use of CO2 as solvent allows for use of reasonable concentrations of H2 and O2 without danger of explosion, and homogeneous catalysis eliminates diffusional limitations to reaction. Although aqueous hydrogen peroxide is an important product in its own right, H2O2 is also a benign intermediate for a variety of oxidation reactions. We have shown that H2O2 generated in situ from H2 and O2 in CO2 cleanly epoxidizes propylene to propylene oxide (PO). PO is currently produced either via the chlorohydrin route, which produces two pounds of waste per pound of PO, or via one of several “peroxidation” processes, where a co-product must be produced with the desired PO and hence waste volumes and energy requirements are much higher than desired. Because those factors that render the current AQ process for H2O2 less than green also contribute to its relatively high cost, generation of H2O2 from H2 and O2 in CO2 not only permits green synthesis of an important commodity, but also opens other applications for use of this green oxidant.

 Please wait while we load your content...
Please wait while we load your content...