CF4defluorination by Cp2Ln–H: a DFT study†
Abstract
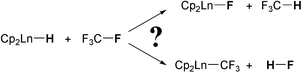
The reaction of CF4 with Cp2Ln–H has been studied with DFT(B3PW91) calculations for the entire family of lanthanide elements. The reaction paths for H/F exchange (formation of CF3H and Cp2Ln–F) and alkylation (formation of Cp2Ln–CF3 and HF) have been determined. Even though a transition state for formation of Cp2Ln–CF3 has been located, Cp2Ln–CF3 reacts with no energy barrier with HF to give Cp2Ln–F and CF3H. The products of the reactions of H/F exchange and

 Please wait while we load your content...
Please wait while we load your content...