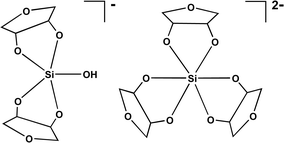
Addition of furanoidic sugars having vicinal cis-diol functionality to aqueous alkaline silicate solutions results in the spontaneous formation of a family of organosilicate complexes in which silicon is either penta- or hexacoordinated. Silicon complexation occurs with 1,4-anhydroerythritol, apiose and ribose, as well as with ribonucleosides (adenosine, cytidine, guanosine) and ribonucleotides (including ATP and NAD+). Silicon-29 and carbon-13 NMR analysis indicates the occurrence of three main complexes containing pentacoordinated silicon. Two hexacoordinated silicon species are also observed in most instances, these being favoured over the pentacoordinated complexes as solution pH is increased, temperature decreased, or the sugar-to-silicon concentration ratio increased. Ribose, which has two vicinal cis-dihydroxy sites for binding silicon, yields a much wider array of complexes.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...