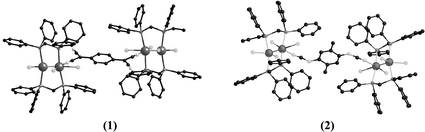
The syntheses, spectroscopic properties, redox chemistry, and solid-state structures of products obtained from the reaction of Re2Cl4(dppm)2
(dppm = bis(diphenylphosphino) methane) with the polycyano acceptors TCNQ (7,7,8,8-tetracyanoquinodimethanido) and DM-DCNQI (2,5-dimethyl-N,N′-dicyanoquinonediimine) are described. The compounds [Re2Cl4(dppm)2]2(μ-TCNQ), 1, and [Re2Cl4(dppm)2]2(μ-DM-DCNQI), 2, have been prepared by reaction of two equivalents of Re2Cl4(dppm)2 with TCNQ and DMDCNQI, respectively, in THF or CH2Cl2. A single-crystal X-ray crystallographic study of [Re2Cl4(dppm)2]2(μ-TCNQ)·10THF revealed the presence of a trans-μ2 bidentate mode for the bridging TCNQ ligand that joins two Re2Cl4(dppm)2 molecules through equatorial positions. In a similar fashion, the compound [Re2Cl4(dppm)2]2(μ-DM-DCNQI)·10THF consists of two Re2 units coordinated to the two nitrile positions of the DM-DCNQI ligand. The electronic properties of both compounds are unusual in that they exhibit intense, broad absorptions that span the near-IR region and extend into the mid-IR. The electrochemistry of the compounds consists of numerous oxidation and reduction processes in the range of +2.0 to −2.0 V as determined by cyclic voltammetry. Both 1 and 2 exhibit temperature independent paramagnetism (TIP) with large χTIP values of 7.29 × 10−3 and 6.23 × 10−3 emu mol−1, respectively.

 Please wait while we load your content...
Please wait while we load your content...