Solid-state molecular syntheses: complete reactions without auxiliaries based on the new solid-state mechanism†
Abstract
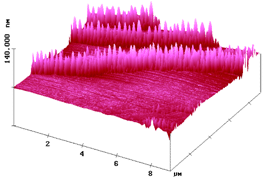
Molecular crystals including salts undergo gas–solid, solid–solid, thermal, photochemical and catalyzed reactions if the crystal lattice allows for long-range anisotropic molecular migrations, if the product phase can form fast enough and if crystal disintegration provides fresh surface. This ingenious three-step solid-state mechanism is derived from atomic force microscopy (AFM) studies, the results of which correlate strictly with the crystal packing. It is secured by the results of various supporting experiments. The experimental mechanism is at variance with Schmidt's “topochemistry” hypothesis with its well-known failures which are now easily settled. Topotactic reactions without molecular migrations are very rare and not at all typical for solid-state reactions. AFM scrutiny excluded several alleged cases of topotaxy and secured three of them with molecular precision. Very detailed solid-state effects that go down to the molecular level emerge from AFM investigations with single crystals on their different faces and are discussed. The efficiency of gas–solid and solid–solid reactions is unexpectedly high. Thus, the yield in one product is 100% due to favorable kinetics in most cases. These waste-free syntheses are fast, require rather simple equipment, save resources and are unbeatable in their wealth for sustainable environmental protection, inasmuch as they do not require workup procedures. The scaling up for industrial application is prepared at the kg level. Numerous selected examples of preparative interest out of more than 1000 are presented.
- This article is part of the themed collection: Molecular Crystal Engineering

 Please wait while we load your content...
Please wait while we load your content...