Photochemical transformations of 2,2′-(1,2-phenylenedivinylene)dipyrroles
Abstract
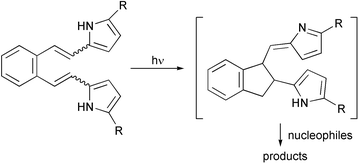
The photochemistry of 2,2′-(1,2-phenylenedivinylene)dipyrroles (4a,b) has been investigated under various conditions. After excitation of the starting material, an electron transfer followed by hydrogen transfer and radical combination occurs giving 2-{[1-(2H-pyrrol-2-ylidene)methinyl]-2-indanyl}pyrrole (14) as the intermediate. The intermediate could not be isolated, but trapped by nucleophiles, like

 Please wait while we load your content...
Please wait while we load your content...