Absolute rate constants for the formation of nitrogen-centred radicals from chloramines/amides and their reactions with antioxidants
Abstract
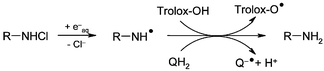
Pulse radiolysis techniques have been employed to investigate the one-electron reduction of a variety of chloramines and chloramides. These include models for the side-chain of Lys (6-aminohexanoic acid chloramine and α-N-acetyl-Lys chloramine), Gly chloramine, β-alanine chloramine and two models of protein backbone amides, the chloramides of cyclo-(Gly)2 and cyclo-(Ala)2. The molar absorption coefficients and stabilities of these chloramines/amides have been determined. The one-electron reduction of these chloramine/amide species by hydrated electrons occurs with second-order rate constants of the order of 109–1010 M−1 s−1, and results in cleavage of the N–Cl bonds to yield nitrogen-centred radicals and chloride ions (as measured by high performance ion chromatography). The reactivities of the nitrogen-centred radicals have been investigated with the readily oxidisable quenchers, hydroquinone and Trolox. These quenchers were used as models of the in vivo antioxidants, ubiquinol-10 and α-tocopherol, and react with second-order rate constants between 2 × 107 and 1 × 108 M−1 s−1. No evidence was obtained in these pulse radiolysis experiments for a rapid rearrangement of the oxidising nitrogen-centred radicals to reducing carbon-centred radicals, though such reactions have been indicated in previous EPR studies.

 Please wait while we load your content...
Please wait while we load your content...