Reactivity and the mechanisms of reactions of β-sultams with nucleophiles
Abstract
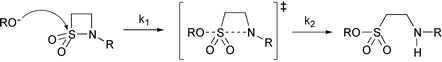
Ethane-1,2-sultam has a pKa of 12.12 ± 0.06 at 30 °C and its rate of alkaline hydrolysis shows a pH-dependence reflecting this so that the observed pseudo first-order rate constant at pHs above the pKa are pH independent. There is no evidence of neighbouring group participation in the hydrolysis of either N-α-carboxybenzylethane-1,2-sultam or N-(hydroxyaminocarbonylmethyl)-2-benzylethane-1,2-sultam. Oxyanions, but not amines or thiols, react with N-benzoylethane-1,2-sultam in water by a nucleophilic ring opening reaction confirmed by product analysis and kinetic solvent isotope effects. A Brønsted plot for this reaction has two distinct correlations with βnuc = 0.52 and 0.65 for weak and strong bases, respectively, although a statistically corrected plot may indicate a single correlation.

 Please wait while we load your content...
Please wait while we load your content...