Synthesis and optimization of tri(propylene glycol) glycerolate diacrylate cross-linked polystyrene resin in polypeptide synthesis: role of the macromolecular support in solid phase peptide synthesis
Abstract
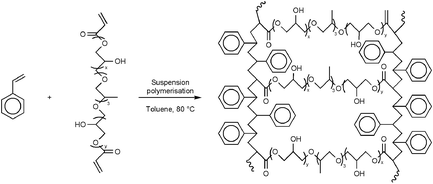
A novel tri(propylene glycol) glycerolate diacrylate cross-linked polystyrene support for solid phase peptide synthesis was prepared by aqueous radical suspension polymerization. The peptides were grown from the hydroxy functionality of the cross-linker in the polymer and this makes it unique among other styrene-based polymer supports that are currently used in polypeptide synthesis. The role of the polymer support in peptide synthesis was established by studies delineating the optimization of synthetic steps involved in solid phase synthesis. The optimization studies include C-terminal amino acid incorporation, Nα-Fmoc and Boc-deprotection, acylation reactions and the removal of the target peptide from the support. The dependence between the nature and extent of cross-linking of the polymer backbone and the reactivity of the attached amino groups was investigated by carrying out a reactivity study on amide bond formation compared with Merrifield resin. The resin-like behavior of the new support was studied by synthesizing a ‘difficult’ sequence of the (34–42) fragment of β-amyloid peptide (1–42) and compared with commercially available Merrifield and Sheppard resins. The synthetic utility of the support was established by synthesizing a 23-residue NR 2B peptide substrate of Ca2+/calmodulin binding peptide in high yield and purity. Better solvation of the resin beads, enhanced coupling efficiency in the peptide synthetic steps and the high yield and purity of the peptides synthesized highlights the positive role of the cross-linker in the new polystyrene support.

 Please wait while we load your content...
Please wait while we load your content...