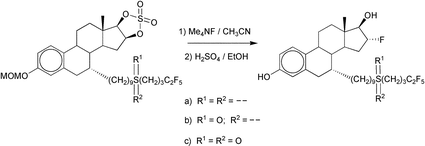
We prepared a new series of 7α-substituted derivatives of 16α-fluoroestradiol, based on the very potent antiestrogen 7α-{9-[(4,4,5,5,5-pentafluoropentyl)sulfinyl]nonyl}-estra-1,3,5(10)-triene-3,17β-diol (ICI 182,780; Faslodex™). The latter consist of estradiol functionalized with a side chain at the 7α-position, conferring interesting pharmaceutical properties for endocrine therapy of estrogen receptor (ER) positive breast cancer. The considerable advantages of ICI 182,780 over other selective ER-modulators (SERMs) already used in hormonal therapy, lead us to develop three new 16α-fluoro derivatives with potential use in positron emission tomography (PET), for the imaging of ER densities in breast tumors. Introduction of the long side chain at the 7α-position was accomplished by Cu(I)-promoted conjugate addition of a Grignard reagent to 6-dehydro-19-nortestosterone. Subsequent oxidation of the 17-hydoxy group and A-ring aromatization gave a 7α-substituted estrone derivative. Further addition to complete the side chain gave the ICI 182,780 mimics that were converted to the reactive 16β,17β-cyclic sulfates, i.e. the key intermediates for the 18F-labeling reaction. Opening of the cyclic sulfates via nucleophilic fluorination with Me4NF, followed by rapid hydrolysis in acidic ethanol of the protecting ether and sulfate groups, yielded the desired 16α-fluoro PET derivatives of ICI 182,780. The latter procedure is readily adapted for radiolabeling with 18F by substituting Me4NF for 18F− in acetonitrile.

 Please wait while we load your content...
Please wait while we load your content...