Hydroxy group interactions in stannylated carbohydrates. Structures and thermal stabilities of 5-deoxy-5-C-(Ph3Sn)-1,2-O-isopropylidene-α-D-xylofuranose, 5-deoxy-5-C-(IPh2Sn)-1,2-O-isopropylidene-α-D- and -L-xylofuranose, and 5-deoxy-5-C-(I2PhSn)-1,2-O-isopropylidene-α-D- and -L-xylofuranose
Abstract
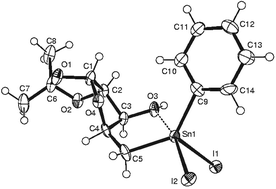
Structures and thermal stabilities of 5-deoxy-5-C-(Ph3Sn)-1,2-O-isopropylidene-α-D-xylofuranose, [(D)-3], 5-deoxy-5-C-(IPh2Sn)-1,2-O-isopropylidene-α-D- and -L-xylofuranose, [(D)-4 and (L)-4], and 5-deoxy-5-C-(I2PhSn)-1,2-O-isopropylidene-α-D- and -L-xylofuranose, [(D)-5 and (L)-5], are reported. The hydroxy groups in the stannylated xylofuranose derivatives, 3–5, exhibit roles as Lewis bases, Brønsted acids and hydrogen bonding centres. The 5-deoxy-1,2-O-isopropylidene-α-D- and –L-xylofuran-5-yl

 Please wait while we load your content...
Please wait while we load your content...