Thioester analogues of peptidoglycan fragment MurNAc-L-Ala-γ-D-Glu as substrates for peptidoglycan hydrolase MurNAc-L-Ala amidase
Abstract
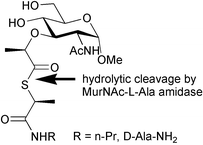
MurNAc-L-amidase is one of a family of peptidoglycan hydrolases which catalyses the breakdown of bacterial peptidoglycan. Analogues of the peptidoglycan fragment MurNAc-L-Ala-γ-D-Glu containing S-thiolactic acid in place of L-alanine were synthesised as thioester substrates for this enzyme. Triphenylmethanethiol was used to develop a stereoselective synthesis of S-thiolactic acid, which was elaborated synthetically into MurNAc-dipeptide analogues. MurNAc-S-thioacetyl-N-propylamide 13 and MurNAc-S-thiolactyl-2R-alaninamide 16 were found not to be substrates for recombinant MurNAc-L-Ala amidases CwlA from Bacillus subtilis and Ply21 from bacteriophage TP21, however, turnover of tripeptide thioester S-propionylthiolactyl-γ-D-Glu-L-Lys-OMe 21 was observed using amidase Ply21. Therefore, recognition of the amino acid at position 3 of the pentapeptide sidechain appears to be important for enzymatic turnover.

 Please wait while we load your content...
Please wait while we load your content...