Synthesis of glycero amino acid-based surfactants. Part 2.1 Lipase-catalysed synthesis of 1-O-lauroyl-rac-glycero-3-O-(Nα-acetyl-L-amino acid) and 1,2-di-O-lauroyl-rac-glycero-3-O-(Nα-acetyl-L-amino acid) derivatives
Abstract
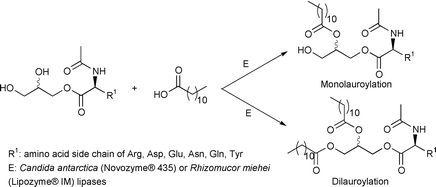
An enzymatic procedure for the synthesis of mono- and dilauroylated amino acid glyceride conjugates is presented. The reaction consisted of the lipase-catalysed esterification of one or two hydroxy groups of the amino acid glyceryl ester derivatives by lauric acid. Solvent-free media with continuous removal of the water produced at atmospheric pressure were the reaction systems of choice. It is found that the mono- and dilauroylation yields and enzymatic activity depended crucially on the reaction temperature, the molar ratio of lauric acid to amino acid glyceryl ester, and concentration of phosphate salts using Candida antarctica (Novozym® 435) and Rhizomucor miehei (Lipozyme® IM) lipases as catalysts. These reaction variables are identified and studied systematically using Boc–Arg–OGl as a model substrate and modulated accordingly for the target amino acid-based compounds. As in conventional glycerides, spontaneous intramolecular acyl-migration reactions are observed for the lauroyl and aminoacyl moieties of the glycerol backbone. This reaction led to a number of regioisomers, which are identified and quantified by mono- and bidimensional NMR techniques. The method developed allowed us to prepare glycero conjugates of arginine, aspartic acid, glutamic acid, asparagine, glutamine and tyrosine in isolated yields of the regioisomeric mixtures ranging from 22 to 69%.

 Please wait while we load your content...
Please wait while we load your content...