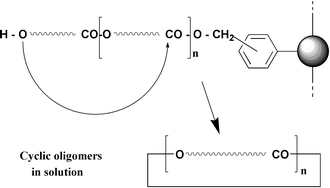
Attachment of ω-hydroxyalkanecarboxylic acids to Merrifield beads followed by treatment with a catalytic amount of di-n-butyltin oxide in chlorobenzene at 133 °C for 4–18 h brought about the formation of the corresponding cyclic oligomers (COs) as the main (>92%) soluble products in yields of 56–83%. Similar results were obtained with polymer-supported (PS) ricinoleic acid, which has a secondary hydroxy group, but attempts to carry out analogous reactions with PS lithocholic acid failed. PS 20-hydroxyicos-10-enoic acid and N-(11-hydroxyundecanoyl)-11-aminoundecanoic acid reacted well to give COs, the cyclic monomers being the major products. Samples of these cyclic monomers were isolated. The latter two hydroxy acids were also successfully assembled on the beads (the former using olefin metathesis) and then cyclo-oligomerised. There are several possible applications for this PS synthetic method. Thus, the COs might be
used, via transesterifications, for the preparation of soluble combinatorial libraries of macrocycles, or used as starting materials for the small-scale combinatorial synthesis of copolyesters. For both of these applications the fact that the PS syntheses afford families of COs is not a problem because the applications involve the establishment of equilibria into which all of the COs can feed. Finally, these PS reactions may provide a useful approach to the small-scale synthesis of macrocyclic lactones with 15 or more ring atoms, since with these ring sizes the cyclised monomers are formed in >67% yield.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...