Strong electronic coupling in intermolecular (charge-transfer) complexes. Mechanistic relevance to thermal and optical electron transfer from aromatic donors
Abstract
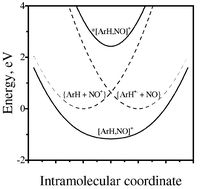
Intermolecular electron transfer from various arene donors (ArH) to the nitrosonium acceptor (NO+) proceeds via a series of unusual [1∶1] precursor complexes. Quantitative analysis of the accompanying pair of charge-transfer absorption bands (hνH and hνL) reveals the presence of strong electronic interactions between the donor/acceptor moieties in [ArH,NO+] complexes with the coupling elements lying in the range: Hab = 1.4 ± 0.5 eV. In the context of Sutin's development of Marcus–Hush theory, these dyads represent a Robin–Day Class III system for the 2-step transformation of the reactant diabatic state {ArH + NO+} to the final diabatic state {ArH+˙ + NO˙}, in which the unusually high values of Hab characterize a series of intermolecular (precursor) complexes that lie in a single (potential-energy) minimum. As such, the thermal electron transfer between the arene donor and the nitrosonium acceptor occurs first via the redistribution of a pair of electrons in the formation of the [ArH, NO]+ complex with Xmin = 1.0, and then followed by its homolytic dissociation to the {ArH+˙ and NO˙} products. This conclusion is experimentally confirmed by X-ray crystallographic and IR analyses of the precursor complexes that identify: (i) the strongly perturbed donor/acceptor moieties structurally akin to arene cation radicals (ArH+˙) and the reduced nitric oxide (NO˙), and (ii) the degree of charge transfer (Z) that is complete (100%). Optical electron transfer via the direct photoactivation of the precursor complex to the identical Franck–Condon state {ArH+˙, NO˙}* occurs independent of whether the high-energy (hνH) or the low-energy (hνL) absorption band is irradiated.

 Please wait while we load your content...
Please wait while we load your content...