Novel properties of ionic liquids in selective sulfur removal from fuels at room temperature
Abstract
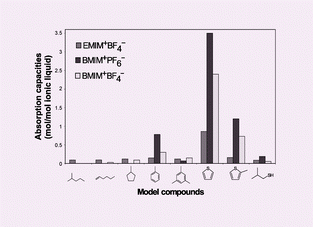
The ionic liquids, 1-ethyl-3-methylimidazolium tetrafluoroborate, 1-butyl-3-methylimidazolium hexafluorophosphate and 1-butyl-3-methylimidazolium tetrafluoroborate were found to be effective for the selective removal of sulfur-containing compounds from transportation fuels such as gasoline at room temperature. S-containing compounds with a C5 aromatic ring are favorably absorbed over C6 aromatics, while S-containing non-aromatic compounds are poorly absorbed. The ionic liquids are regenerated from absorbed S-containing compounds by distillation or by dissolution in water. These ionic liquids are air- and moisture-stable at low temperature and non-corrosive. Therefore, the ionic liquids can be used in multiple cycles for the removal of S-containing compounds from fuels. The absorption capacity of the ionic liquids for S-containing compounds is sensitive to the structure of both the anion and cation of the ionic liquids, which is manifested by the significant inhibiting effect of methyl group substitution on the aromatic ring.

 Please wait while we load your content...
Please wait while we load your content...