Characterisation of gallium(iii)-acetate complexes in aqueous solution: A potentiometric, EXAFS, IR and molecular orbital modelling study
Abstract
The aqueous gallium(III)-acetate system was studied in 0.6 M Na(Cl) at 25 °C using a multi-technique approach, including potentiometry, IR spectroscopy, EXAFS spectroscopy and molecular orbital calculations. The potentiometric data were satisfactorily explained by a model which includes one mononuclear and one binuclear complex. The corresponding equilibrium constants defined according to the reactions
Ga3+ + HAc ⇄ GaAc2+ + H+
2Ga3+ + HAc + 2H2O ⇄ Ga2(OH)2Ac3+ + 3H+
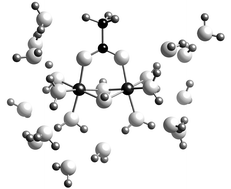
are log β−1,1,1 = −2.08 ± 0.09 and log β−3,2,1 = −5.65 ± 0.06, respectively. The latter complex has a stoichiometry identical to that previously identified in the aluminium(III)-acetate system. Also in agreement with this system, the IR and EXAFS data strongly indicates that this complex consists of two edge-sharing Ga octahedra bridged with an acetate ion, and should be formulated [Ga2(μ-OH)2(μ-O2CCH3)]3+. This interpretation is further supported by the molcular orbital calculations.

 Please wait while we load your content...
Please wait while we load your content...