Vibrational properties and matrix photochemistry of trimethyldioxorhenium(vii), (CH3)3ReO2†
Abstract
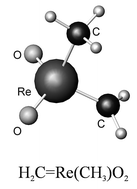
Gaseous, liquid and matrix-isolated trimethyldioxorhenium(VII), 1, has been characterised (i) by the vibrational spectra of the three isotopomers, (CH3)3ReO2, 1-h9, (CD3)3ReO2, 1-d9, and (CHD2)3ReO2, 1-d6, recorded between 4000 and 400 cm−1, and (ii) by density functional theory (DFT) calculations. The ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Re(CH3)O2, 2, but a secondary change becomes evident on continued photolysis, probably resulting in the formation of the bis(methylidene) derivative (H2C)2Re(O)OH, 3. The results are discussed in relation to the photochemistries of other alkyloxorhenium compounds.
Re(CH3)O2, 2, but a secondary change becomes evident on continued photolysis, probably resulting in the formation of the bis(methylidene) derivative (H2C)2Re(O)OH, 3. The results are discussed in relation to the photochemistries of other alkyloxorhenium compounds.

 Please wait while we load your content...
Please wait while we load your content...