Niobocene hydrides with functionalised silyl ligands. Tuning of interligand hypervalent interactions (IHI) M–H → Si–X
Abstract
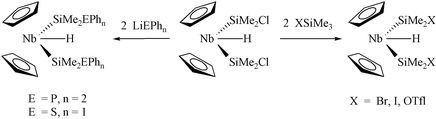
This paper presents syntheses and X-ray structure determinations of a variety of mono- and bis-(silyl) niobocene hydride complexes with functionalised silyl ligands. Complexes of the type [NbCp2(SiR3)H2] (SiR3 = SiMe2Cl (3), Si(OEt)3, SiMe2OEt (4)) can be prepared conveniently by heating the precursor [NbCp2H3] in the presence of the corresponding silane HSiR3. As the electrondonating ability of the R group increases, the corresponding mono(silyl) derivatives [NbCp2(SiR3)H2] are either unstable (SiR3 = SiMe3) or do not exist at all (SiR3 = SiEt3, Si(OCH2CH2)3N). The previously described bis(silyl) complex [NbCp2(SiMe2H)2H] easily reacts with half an equivalent of iodine, giving the functionalised derivative [NbCp2(SiMe2I)2H] (10). A labelling experiment for the analogous reaction with bromine establishes that an electrophile attacks the more accessible Si–H bond rather than the more electron-rich Nb–H bond. Bis(silyl) complexes [NbCp2(SiMe2X)2H] (X = Br (8), I (10), OTfl (11)) can be conveniently prepared by the reaction of [NbCp2(SiMe2Cl)2H] (2) with the corresponding silane XSiMe3. Reactions of 2 with fluorinating reagents such as [CPh3][BF4], BF3·OEt2 and AgBF4 do not work well for the synthesis of the compound [NbCp2(SiMe2F)2H]. Nucleophilic chlorine substitution in 2 under the action of lithium amides and alkoxides does not occur, whereas reaction with soft reagents such as MeLi, LiPPh2 and LiSPh leads to the corresponding derivatives [NbCp2(SiMe3)2H] and [NbCp2(SiMe2EPhn)2H] (E = S, n = 1 (14); E = P, n = 2 (15)). Methanolysis of 10 gives high yields of the derivative [NbCp2(SiMe2OMe)2H] (18). The mono(silyl) precursor [NbCp2(SiMe2Cl)H2] (3) reacts with LiOEt affording a mixture of [NbCp2(SiMe2OEt)H2] and [NbCp2H3]. Reaction of 3 with LiPPh2 gives high yields of the compound [NbCp2(SiMe2PPh2)H2] (17). Reduction of 3 with LiAlH4 results in the dimeric compound [NbCp2H2Al(μ-H)2]2 (13). Carrying out this reaction in the presence of an amine leads to the formation of the complex [NbCp2(SiMe2H)H2]. X-Ray structure determinations were performed for the compounds (10), (13), (17), and (18). Complex (10) has 5 centre–6 electron interligand hypervalent interactions (IHIs) I–Si⋯H⋯Si–I; the analogous IHI O–Si⋯H⋯Si–O in 18 is significantly weaker, whereas 17 has a classical structure. Complex 13 has a cenrosymmetric dimeric structure, with two five-co-ordinate aluminium centres bridged by hydride ligands.

 Please wait while we load your content...
Please wait while we load your content...