Joanna Wolowska, Graham R. Eastham, Brian T. Heaton, Jonathan A. Iggo, Chacko Jacob and Robin Whyman
Chem. Commun., 2002, 2784-2785
DOI:
10.1039/B208450C,
Communication
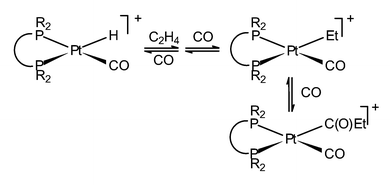
All the intermediates involved in the platinum catalysed methoxycarbonylation of