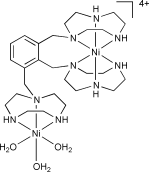
A structural and electrochemical study of the nickel(II) complexes, [Ni(tacn)2](ClO4)2 (1), [NiLeth](ClO4)2 (2), [NiLox](ClO4)2·H2O (3), [Ni2Ldur](ClO4)4·2H2O (4), [Ni3Ldur(H2O)3](ClO4)6·9H2O (5) and [Ni2Lisomes(H2O)3](ClO4)4·7H2O (6) [tacn = 1,4,7-triazacyclononane, Leth = 1,2-bis(1,4,7-triazacyclonon-1-yl)ethane, Lox = 1,2-bis(1,4,7-triazacyclonon-1-ylmethyl)benzene,
Ldur = 1,2,4,5-tetrakis(1,4,7-triazacyclonon-1-ylmethyl)benzene, Lisomes = 1,2,3-tris(1,4,7-triazacyclonon-1-ylmethyl)benzene] is reported. Single-crystal X-ray crystallography has established that 2 and 6 feature nickel(II) centres in a distorted octahedral geometry, which are sandwiched between pairs of tacn rings. In 6, a second nickel(II) centre coordinates the third tacn ring of Lisomes and three water molecules complete the distorted octahedral coordination sphere. For the sandwiched Ni(II) centres, steric contraints introduced by the ethane and ortho-oriented tacn rings in Lisomes cause significant distortions to the Ni(II) coordination spheres. Cyclic, square-wave and near steady-state voltammetric studies on 1–6 indicate that the sandwiched nickel(II) centres may be reversibly oxidised to the nickel(III) state, with the potential for the nickel(II)/(III) couple showing a dependence on the nature of the tether linking the sandwiching tacn rings. Complex 4 undergoes oxidation in two overlapping one-electron processes [i.e., Ni(II)Ni(II) → Ni(III)Ni(II) → Ni(III)Ni(III)], indicating that the two nickel centres within the complex behave in an essentially independent fashion.

 Please wait while we load your content...
Please wait while we load your content...