 Open Access Article
Open Access ArticleLithium-ion uptake on lithium manganese oxide soft and bare electrodes
S. Orozco-Barrera†
 a,
J. A. Lirio Piñar†
a,
J. A. Lirio Piñar† a,
C. Kök
a,
C. Kök bc,
G. R. Iglesias
bc,
G. R. Iglesias ad,
A. V. Delgado
ad,
A. V. Delgado a,
V. Presser
a,
V. Presser *bce and
S. Ahualli
*bce and
S. Ahualli *a
*a
aDepartment of Applied Physics, School of Sciences, University of Granada, 18071 Granada, Spain. E-mail: sahualli@ugr.es
bINM - Leibniz Institute for New Materials, Campus D2 2, 66123 Saarbrücken, Germany. E-mail: Volker.presser@leibniz-inm.de
cDepartment of Materials Science & Engineering, Saarland University, Campus D2 2, 66123 Saarbrücken, Germany
dNanoMag Lab Department of Applied Physics Edificio I+D Josefina Castro, University of Granada, Spain
eSaarene - Saarland Center for Energy Materials and Sustainability, Campus C4 2, 66123 Saarbrücken, Germany
First published on 27th April 2026
Abstract
Lithium is a critical resource for high-energy batteries and emerging energy storage technologies. Conventional extraction methods, such as solar evaporation of brines, are energy-intensive and environmentally harmful, highlighting the need for sustainable alternatives. Here, we present an electrochemical strategy for selective lithium recovery using a hybrid intercalation-based electrochemical cell that exploits the unique properties of lithium manganese oxide (LMO). Unlike traditional carbon-based electrodes, LMO features a spinel crystal structure that enables reversible Li+ intercalation under controlled potentials, providing intrinsic ion selectivity. To improve durability and performance, the LMO electrode was functionalized with the anionic polyelectrolyte poly(sodium 4-styrenesulfonate) (PSS). At the same time, the activated carbon counter-electrode was coated with the cationic polyelectrolyte poly(diallyldimethylammonium chloride) (PDADMAC). Experiments in single-salt (LiCl) and mixed-salt (LiCl + NaCl) systems, including highly asymmetric brines, reveal that Li+ uptake strongly depends on the applied cell voltage, reaching values above 40 mg g−1 at 1.2 V. The PSS coating is expected to mitigate Mn dissolution and alters desorption dynamics, favoring Na+ release while maintaining Li+ selectivity. These results demonstrate the potential of LMO-based hybrid electrodes for energy-efficient lithium recovery from complex saline environments.
1. Introduction
Lithium is a crucial element in industry, particularly notable for its significance in the production of high-energy-density rechargeable batteries and other electronic devices.1–4 The demand for lithium has grown exponentially in recent decades, driven by the rise of electromobility and the increasing need for renewable energy storage. The primary sources of lithium are brines found in salt flats5–7 and minerals such as spodumene.8 The most significant lithium-rich brine deposits are located in the region known as the lithium triangle in South America, which encompasses Argentina, Bolivia, and Chile. The most common conventional extraction process is based on solar evaporation, a technique that involves the evaporation of large volumes of water to concentrate the salts and extract the lithium.9–11 In these large-scale operations, the high aqueous solubility of lithium chloride, 830.5 g L−1 at 20 °C,12 allows lithium cations to remain in solution while other salts precipitate. Once the brine is sufficiently concentrated, chemical precipitation is induced using carbonate precursors, such as adding Na2CO3 to precipitate Li2CO3, taking advantage of its lower solubility limit.13Despite its widespread use, this carbonate-based recovery is ineffective for dilute sources, as the solubility limit of Li2CO3 cannot be reached without prior and extensive concentration of LiCl, often requiring 12 to 24 months to complete.14 Furthermore, this method is associated with significant environmental challenges, including intensive water usage in arid regions and the generation of saline waste, which can have detrimental effects on local biodiversity.9,15
Electrochemical methods have emerged as promising technologies for more efficient and sustainable lithium extraction from low-concentration aqueous streams in a matter of hours.16–18 As an example, capacitive deionization, employing ion electrosorption as the ion removal mechanism,19 may enable the selective recovery of ions from aqueous solutions or brines.20–22 Compared to conventional methods, capacitive deionization can operate more efficiently at a smaller scale. This makes it a competitive option for lithium recovery, particularly in cases where traditional techniques are either ineffective or harmful to the environment.23–26 However, without proper functionalization of the electrodes, this technique is not efficient in selectively separating monovalent cations such as Na+ and Li+. In this context, hybrid electrochemical ion separation cells, where one of the electrodes is constructed with intercalation materials, provide a more accurate description of the system studied here, as ion trapping occurs via redox intercalation rather than double-layer adsorption.27
Using LiMn2O4 (LMO) as the primary active material provides a unique ability to selectively intercalate Li+.24,28–30 LMO has a spinel-like crystal structure, which facilitates efficient ion mobility and enables high reversibility in the intercalation and deintercalation of Li+. This property is crucial to maintain the structural integrity of the material over multiple charge–discharge cycles.31 Also, its relatively low production cost and low environmental impact compared to other materials, which reinforces its feasibility for electrode synthesis, make LMO suitable for electrochemical systems.32
A key aspect of this process is how the active electrode material participates in the transfer of electrons during the redox reaction (eqn (1)):
| LiMn2O4 ↔ Li1−xMn2O4 + xLi+ + xe− | (1) |
Unlike LMO's smaller surface area (<2 m2 g−1), activated carbons are known for their large specific surface area and the ion uptake is governed by ion electrosorption.33–35 Recent studies have demonstrated that the coating of carbon electrodes with polyelectrolytes can substantially augment their ion removal capacity and overall desalination performance.36–39 This enhancement is attributed to the promotion of counterion transport and the suppression of co-ion release, resulting in improved charge efficiency and stability.36–38,40–43
This study focuses on elucidating the influence of polyelectrolyte coating on the performance of hybrid electrochemical ion separation cells employing LMO as the active electrode material. Poly(sodium 4-styrenesulfonate) PSS is utilized as an anionic coating for the LMO electrode, while poly(diallyldimethylammonium chloride) (PDADMAC) is employed as a cationic coating for the AC counter-electrode. The selection of these polymers was predicated on their water-solubility, commercial availability, and established role in the literature concerning the enhancement of charge efficiency and electrode stability in capacitive deionization systems.36,44 PSS has been reported to enhance lithium-ion transport44–47 and to mitigate the detrimental effects of the Jahn–Teller distortion, which can lead to Mn dissolution during electrochemical cycling.48,49 This modification is expected to enhance the material's durability and maintain its lithium selectivity over prolonged operation. PDADMAC has demonstrated benefits in stabilizing and improving the capacitive performance of carbon electrodes.39 However, the influence of such coatings on intercalation-type electrodes, such as LMO, has not yet been systematically explored.
A series of systematic experiments was conducted in which key operational parameters, such as the applied voltage, LMO-to-AC mixing ratio, and electrode connection mode during the discharge step, were varied. The goal was to assess how these variations influence the selective uptake and release of Li+ and Na+. A distinctive feature of this study is the real-time visualization of the desalination cycles through conductivity monitoring, which enables in situ evaluation of ion-removal dynamics and kinetics. This approach provides novel insights into the role of polymer coatings in enhancing both the selectivity and the structural stability of LMO-based electrodes for Li+ recovery from mixed brines.37,39,41
2. Experimental
2.1. Materials
LiCl (99.98% purity, Sigma Aldrich) and NaCl (99.73% purity, Fisher Chemicals) solutions were prepared in Milli-Q water (Milli-Q Academic, Millipore). LiMn2O4 (>98% purity, Sigma Aldrich), YP-80F activated carbon (AC) (supplied by Kuraray), and carbon black (>99% purity, Thermo Fisher) were used as materials in the preparation of the electrodes. Poly-diallyldimethylammonium chloride (PDADMAC) (average molecular weight above 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, Sigma Aldrich) and PSS (average molecular weight of 70
000, Sigma Aldrich) and PSS (average molecular weight of 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, Sigma Aldrich) were used as cationic and anionic polyelectrolytes, respectively, to coat the electrodes where appropriate. In addition, a commercial anion exchange membrane (AMX, Tokuyama) from Neosepta, with a thickness of 140 µm, was placed in front of the AC electrode.
000, Sigma Aldrich) were used as cationic and anionic polyelectrolytes, respectively, to coat the electrodes where appropriate. In addition, a commercial anion exchange membrane (AMX, Tokuyama) from Neosepta, with a thickness of 140 µm, was placed in front of the AC electrode.
2.2. Particle characterization
The particle morphology was obtained from high-resolution electron microscope (Thermo Fisher Scientific TALOS F200X microscope) pictures. The particle size distributions in water were determined by dynamic light scattering (Malvern Zeta Sizer Nano-ZS, Malvern Pananalytical). Nitrogen sorption measurements at −196 °C were performed using a Quadrasorb IQ system (formerly Quantachrome; now Anton Paar) after degassing the samples under vacuum at 100 °C for 12 h. The specific surface area was calculated using the Brunauer–Emmett–Teller method.50–52 Quenched solid density functional theory was used to calculate the pore size distribution, assuming slit-shaped pores.The structural properties of the particles were evaluated by powder X-ray diffraction using a Bruker D8 Discover diffractometer equipped with a Pilatus3R 100K-A detector. The experiments were conducted at 25 °C, employing Cu Kα radiation (λ = 1.5406 Å) with an operating voltage of 50 kV and a current of 1 mA. Data were collected over a 2θ range of 5° to 85°, with a step size of 0.02° and a counting time of 40 s per step, and compared with diffractograms generated by the PowderCell 2.4 software (developed by the Federal Institute for Materials Research and Testing).
Chemical analysis of the materials was performed by Fourier-Transform infrared spectroscopy in an FT/IR-6200 spectrometer, JASCO, with a resolution of 0.25 cm−1.
2.3. Electrode preparation
The electrochemical cell employed in this study consists of two flat electrodes arranged in a parallel-plate configuration. The working electrode was made of LMO or its PSS-coated counterpart (LMO-soft), while the counter-electrode was prepared from AC or PDADMAC-functionalized AC (AC-soft). Both electrodes were assembled on graphite current collectors and separated by a 400 µm insulating spacer, with an anion-exchange membrane (AMX) positioned adjacent to the carbon electrode.For the preparation of the active electrode material, 0.23 g of active material (a mixture of AC and LMO, hereafter referred as AC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LMO) and carbon black powder mixture (95
LMO) and carbon black powder mixture (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 ratio by mass) was added to 0.77 g of a 33 g L−1 suspension of poly-vinylidene-fluoride (PVDF), manufactured by Arkema as Kynar HSV 900, with molecular weight approximately 1
5 ratio by mass) was added to 0.77 g of a 33 g L−1 suspension of poly-vinylidene-fluoride (PVDF), manufactured by Arkema as Kynar HSV 900, with molecular weight approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 in 1-methyl 2-pyrrolidone (Sigma Aldrich). The AC
000 in 1-methyl 2-pyrrolidone (Sigma Aldrich). The AC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LMO mass/mass ratios employed in this study are 0
LMO mass/mass ratios employed in this study are 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 (hereafter referred to as LMO), 20
100 (hereafter referred to as LMO), 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80, 40
80, 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60, and 60
60, and 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40. The suspension was magnetically stirred until a homogeneous slurry was obtained and spread on the surface of a graphite plate (Mersen) serving as the current collector. The electrode was dried overnight at 70 °C, yielding 0.04 ± 0.01 g of active material with a characteristic bluish-gray appearance, as shown in SI, Fig. S1A.
40. The suspension was magnetically stirred until a homogeneous slurry was obtained and spread on the surface of a graphite plate (Mersen) serving as the current collector. The electrode was dried overnight at 70 °C, yielding 0.04 ± 0.01 g of active material with a characteristic bluish-gray appearance, as shown in SI, Fig. S1A.
Prior to the assembly of the LMO electrode in a measuring cell, it was subjected to a pre-treatment process to ensure the release of Li+ from the LMO structure itself, thus allowing the electrode to trap-release Li+. For this, the electrode was immersed in a 30 mM LiCl solution, and 1 V was applied for 120 min.28 After this process, the electrode becomes reddish, which is characteristic of the delithiated LMO (SI, Fig. S1B).
The polymeric coating of the LMO electrode was carried out by immersing the already formed electrode in a stirred 200 mM aqueous solution of PSS overnight. After this stage, the electrode was rinsed thrice with deionized water (Milli-Q) and dried at room temperature.53
For the coating of the AC counter-electrode, the carbon powder was dispersed in 200 mM PDADMAC solution, stirred for 12 h, centrifuged, and redispersed in water at least three times or as needed for complete removal of the non-adsorbed polyelectrolyte. Finally, the carbon particles were dried at 65 °C and used for the preparation of the electrode as described above.54
The desalination cell was described in detail in ref. 40. The active area of the electrodes is 50 cm2. The LMO (or LMO-soft) electrode is placed facing the AC (or AC-soft) counter-electrode, separated by a 400 µm mesh insulating spacer. The AMX membrane was always placed next to the latter electrode.
2.4. Electrochemical Li+ recovery
The experimental method described in ref. 36 was followed to perform electrochemical Li+ recovery. Briefly, in the uptake step, the LMO electrode was set at −0.4 V, −0.9 V, or −1.2 V with respect to the AC-soft electrode by means of an Ivium-Stat potentiostat (Ivium Technologies) in a two-electrode configuration. Because of this setup without a standard reference electrode, it is important to clarify that the reported voltage values throughout this work represent the total cell voltage (i.e., the potential difference applied between the LMO working electrode and the AC counter electrode). For release, an opposite cell voltage (reverse voltage or RV) was applied to reach the potential for controlled deintercalation of the ions from the LMO structure. The electrolyte solutions investigated were LiCl 10 mM, NaCl 5 mM + LiCl 5 mM, and NaCl 9.5 mM + LiCl 0.5 mM. The flow rate was 0.7 mL s−1, and each cycle duration was 10 min.2.5. Determination of the ionic composition of the solutions
A follow-up of the ion uptake and release processes was carried out by in-line measurements of electrical conductivity using a 529670-conductivity probe connected to a Leybold 524D10 Cassy Lab interface (Leybold). To evaluate the concentrations of individual ions, two types of measurements were conducted. Ion chromatography was employed for online determinations, whereby 2 mL samples were taken at various times during the cycle to identify the type and concentration of the ionic species of interest at each step of the process. The analysis of the samples was performed with a 940 Professional IC Vario ion chromatograph (Metrohm).Real-time measurements were performed to determine ionic concentrations of the target species, lithium, and sodium, at the outlet of the desalination cell.55 This was carried out by inductively coupled plasma optical emission spectroscopy (ICP-OES) using a SPECTRO ARCOS ICP-OES analyzer (Spectro Ametek) with a flow rate of 1 mL min−1.
3. Results and discussion
3.1. Particle characterization
Fig. 1C and E show electron micrographs of the LMO, AC, and carbon black samples. The appearance of the former is plate-like with an average size of 45 ± 14 nm. The AC particles exhibit pores and have a size of 2.9 µm (d50), while the carbon black particles were about 32 ± 15 nm in size. The nitrogen adsorption isotherms (SI, Fig. S2) demonstrate a very high specific surface area for AC (2029 m2 g−1) as compared to the other samples (15 m2 g−1 for LMO and 56 m2 g−1 for carbon black).X-ray diffractograms are plotted in Fig. 1F. As can be observed, the peaks obtained for LMO coincide with those in the theoretical diffractogram considering a unit cell with the space group Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m and 8.2476 Å cell size, with Li+, Mn3+/Mn4+, and O2− ions occupying Wyckoff positions 8a, 16d, and 32e, respectively (Fig. 1A and B).56,57 The diffractograms obtained for LMO with and without PSS coating are practically identical, indicating that the crystal structure remains unaffected by the functionalization with the polyelectrolyte. This is advantageous due to the selective properties of LMO towards Li+, which are derived from its spinel-like structure.24,28–31
m and 8.2476 Å cell size, with Li+, Mn3+/Mn4+, and O2− ions occupying Wyckoff positions 8a, 16d, and 32e, respectively (Fig. 1A and B).56,57 The diffractograms obtained for LMO with and without PSS coating are practically identical, indicating that the crystal structure remains unaffected by the functionalization with the polyelectrolyte. This is advantageous due to the selective properties of LMO towards Li+, which are derived from its spinel-like structure.24,28–31
As evident in the diffractogram of activated carbon AC (SI, Fig. S3), the absence of peaks indicates that it is a non-crystalline, amorphous solid.56 A broad peak can be observed at approximately 2θ = 23.5°, which can be attributed to the short-range order of the hexagonal arrangement of the carbon atoms, which is analogous to that observed in graphite.57 The presence of partially ordered mesopores can be inferred from the peak that appears at small angles, 2θ < 5°.58 This appears to be attenuated by the PDADMAC coating, which may be indicative of pore occlusion resulting from polyelectrolyte deposition.59–61
Infrared spectra were obtained for LMO and AC, and their functionalized variants, as displayed in Fig. 1G and SI, Fig. S4. As demonstrated in Fig. 1G, LMO is characterized by a series of pronounced peaks, which are indicative of two distinct vibrational modes of the manganese–oxygen bonds appearing at 500–580 cm−1 and 620–650 cm−1, respectively.62 Furthermore, the spectrum also exhibits traces of environmental water and carbon dioxide. By contrast, PSS-functionalized LMO exhibited bands characteristic of PSS.63 For instance, the following vibrational modes have been identified: (C–H) sp3 (aliphatic) stretches at 2850–2960 cm−1;63,64 aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretches at 1500–1600 cm−1,65 symmetric (1180–1250 cm−1) and asymmetric (1030–1040 cm−1) stretches of the sulfonate groups.66,67 Furthermore, a peak at approximately 1130 cm−1 was identified in the functionalized LMO spectrum, which is most likely attributable to vibrations of the sulfonate group of PSS, displaced by electrostatic or coordinative interactions with the LMO surface.68,69 These results confirm the presence of PSS on LMO particles, as well as the interaction of the polyelectrolyte with the surface of the particles.
C stretches at 1500–1600 cm−1,65 symmetric (1180–1250 cm−1) and asymmetric (1030–1040 cm−1) stretches of the sulfonate groups.66,67 Furthermore, a peak at approximately 1130 cm−1 was identified in the functionalized LMO spectrum, which is most likely attributable to vibrations of the sulfonate group of PSS, displaced by electrostatic or coordinative interactions with the LMO surface.68,69 These results confirm the presence of PSS on LMO particles, as well as the interaction of the polyelectrolyte with the surface of the particles.
The results for the AC and its PDADMAC functionalization are displayed in SI, Fig. S4. The analysis of the spectra revealed that both substances exhibit peaks at 1000–1100 cm−1, which correspond to the C–O stretching modes of surface oxygenated groups.70 Additionally, the spectra demonstrate peaks at 1600–1620 cm−1, which correspond to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching of the graphitic skeleton of the AC.71 Further analysis indicates peaks at 2850–2920 cm−1, which are indicative of aliphatic C–H stretching and suggest that the material is not fully graphitized.72 Finally, the spectra show peaks at 3200–3600 cm−1, which correspond to adsorbed water and hydroxyl groups.73 Additionally, traces of environmental carbon dioxide (CO2) were detected. Other bands observed in AC-soft can be ascribed to the presence of PDADMAC, as reported in ref. 74. In particular, bands at 950–1050 cm−1 C–N+ vibrations, and at 1380–1400 cm−1 methyl symmetric bending of the CH3 group.75,76 A peak at 1620–1640 cm−1 is indicative of water adsorbed on a hydrophilic cationic polymer.77
C stretching of the graphitic skeleton of the AC.71 Further analysis indicates peaks at 2850–2920 cm−1, which are indicative of aliphatic C–H stretching and suggest that the material is not fully graphitized.72 Finally, the spectra show peaks at 3200–3600 cm−1, which correspond to adsorbed water and hydroxyl groups.73 Additionally, traces of environmental carbon dioxide (CO2) were detected. Other bands observed in AC-soft can be ascribed to the presence of PDADMAC, as reported in ref. 74. In particular, bands at 950–1050 cm−1 C–N+ vibrations, and at 1380–1400 cm−1 methyl symmetric bending of the CH3 group.75,76 A peak at 1620–1640 cm−1 is indicative of water adsorbed on a hydrophilic cationic polymer.77
3.2. Influence of the applied cell voltage on ion uptake and release
Before evaluating the influence of the applied cell voltage, it is important to recall that the extraction process of Li+ ions from LMO requires activation of the material by applying a positive voltage to overcome the lithium diffusion barrier.78,79 It has been reported that for undoped LMO, the barrier is approximately 0.35 eV, and it is presumed to be higher at the material surface.80 Based on experimental cyclic voltammetry data, a cell voltage of 0.9 V is sufficient for activation, highlighting the importance of precise voltage control to enhance sorption efficiency.81,82To examine how the cell voltage affects ion uptake once the activation threshold is reached, experiments were conducted at three cell voltages: 0.4 V, 0.9 V, and 1.2 V. To evaluate the influence of the applied voltage, two characteristic parameters were considered: the specific uptake capacity (SAC) and the charge efficiency (ε). The SAC is defined as (eqn (2))
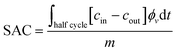 | (2) |
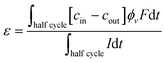 | (3) |
To provide a more fundamental electrochemical perspective, the lithium recovery capacity is not only reported in terms of mass (SAC, mg g−1) but also as the lithium molar insertion fraction (y). This metric represents the moles of Li+ intercalated per mol of LMO (LiMn2O4) and is calculated from the SAC using the respective molar masses of Li and LMO (MLi and MLMO, respectively) eqn (4):
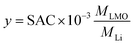 | (4) |
Expressing the Li content in terms of molar composition clarifies the specific electrochemical mechanism governing the ion uptake process. Note that if y is below 1, Li is inserted in tetrahedral sites, corresponding to the so-called 4 V process, which retains the crystal structure of LMO. In contrast, y > 1 would force the lithium ions partially into octahedral sites, producing Jahn–Teller distortions and degrading the crystal structure.83,84 For the sake of clarity and to facilitate comparison with theoretical limits, all SAC values reported hereafter are accompanied by their corresponding lithium molar insertion fraction (y) in parentheses.
Experiments performed at a cell voltage of 0.4 V, presented in Fig. 2A, show negligible differences between the deionization of LiCl and LiCl/NaCl mixture solutions. This is expected because the applied voltage is insufficient to significantly activate the intercalation of Li+ in the LMO electrode, with the maximum SAC values being (7.2 ± 0.1) mg g−1 (0.1876 ± 0.0026). In contrast, at a cell voltage of 0.9 V (Fig. 2B), oxidation of the LMO is anticipated, facilitating the intercalation of Li+ into its structure. As shown in the Figure, the system demonstrates considerable extraction capacity for Li+ in pure solution, reaching SAC values of (21.9 ± 0.1) mg g−1 (0.5706 ± 0.0026), with near-maximum efficiencies (Table 1). For a mixed solution, we see a less pronounced ion uptake/release curve, which has an amplitude lower than that of LiCl.
| Solution | Cell voltage (V) | SAC (mg g−1) | Lithium molar insertion fraction y | Charge efficiency ε | |
|---|---|---|---|---|---|
| LMO electrode | 10 mM LiCl | 0.4 | 7.2 ± 0.1 | 0.1876 ± 0.0026 | 0.96 ± 0.02 |
| 0.9 | 21.9 ± 0.1 | 0.5706 ± 0.0026 | 0.94 ± 0.02 | ||
| 1.2 | 44.5 ± 0.1 | 1.1594 ± 0.0026 | 0.91 ± 0.02 | ||
| LMO-soft electrode | 10 mM LiCl | 0.4 | 10.7 ± 0.1 | 0.2788 ± 0.0026 | 0.81 ± 0.02 |
| 0.9 | 12.5 ± 0.1 | 0.3257 ± 0.0026 | 0.78 ± 0.02 | ||
| 1.2 | 34.1 ± 0.1 | 0.8884 ± 0.0026 | 0.76 ± 0.02 |
Finally, to evaluate whether increasing the cell voltage enhances the selectivity of the process or induces side effects like undesired oxidation, additional experiments were conducted at 1.2 V. As shown in Fig. 2C, the application of this cell voltage reveals a complex deionization behavior that, to our knowledge, has not been previously reported. One might expect that in a mixed solution containing both Li+ and Na+, the LMO electrode would only capture the Li+, remaining indifferent to the Na+. Conductivity measurements demonstrate that ion release occurs even during the ion-capture step in mixed solutions. This suggests that the elevated cell voltage not only intensifies the intercalation of Li+ into the LMO but could also be causing the release of previously adsorbed Na+, which is likely to be expelled from the electrode because of the preferential uptake of Li+. To confirm this hypothesis, we carried out real-time ICP-OES measurements of ion concentration using a 5 mM NaCl + 5 mM LiCl mixed solution (SI, Fig. S5). Initial uptake occurs at a similar rate for both Na+ and Li+ ions; however, Na+ uptake is less sustained, and a net ion release is observed at the end of the process. Conversely, Li+ incorporation into LMO continues throughout the ion uptake step. Obviously, ion replacement occurs when Li+ is intercalated into the LMO lattice, causing the release of previously uptaken Na+.
3.3. Polymeric coating on LMO
During the cell operation, LMO experiences manganese dissolution into the aqueous electrolyte, resulting in the gradual degradation of the active material. This process becomes particularly significant in environments involving electrochemical potential variations, such as those present in the desalination cycles investigated. Several factors, including the pH of the electrolyte, the operating temperature, and the applied charge/discharge cycles, govern the extent of Mn dissolution. Such effects compromise the durability of LMO and restrict its applicability in long-life systems.25,85–88 To address this limitation, several strategies have been proposed, notably structural modification through metal doping89–91 to stabilize its spinel structure.On the one hand, coating LMO with polymers has been posited as an effective strategy to enhance its stability.86,92 This polymeric coating functions as a physical barrier, thereby mitigating the structural degradation of LMO caused by manganese dissolution in aqueous media. On the other hand, PSS coating AC enhances the electrical conductivity in proximity to the interfaces, thereby increasing cation uptake efficiency while impeding the co-ion penetration into the electrode during reverse-voltage operation.44–47 For this reason, LMO has been combined with PSS, and a direct comparison between the LMO-soft electrode and its uncoated version is presented in Fig. 2, where the experiments were repeated for the three different cell voltages with PSS-coated LMO (LMO-soft). It yields ion uptake/release profiles (Fig. 2D–F) for both the pure and mixed solutions that are qualitatively similar to those obtained with bare LMO (Fig. 2A–C). Consequently, we used a cell voltage of 1.2 V for all subsequent analyses.
The concentration profiles are depicted in Fig. 3. These measurements were taken for a 1.2 V cell voltage; the release was performed in RV mode. Polyelectrolyte-coated electrodes exhibit a more distinct conductivity minimum that occurs slightly earlier than in uncoated LMO. Subsequent to this minimum, there is a more rapid return of the conductivity to the inlet value. Despite a faster ion uptake, this phenomenon results in a lower overall uptake capacity compared to uncoated electrodes. Table 1 shows that the SAC decreases from (44.5 ± 0.1) mg g−1 (1.1593 ± 0.0026) for the uncoated electrode to (34.1 ± 0.1) mg g−1 (0.8884 ± 0.0026) for the LMO-soft electrode, while the charge efficiency decreases from 0.91 ± 0.02 to 0.76 ± 0.02. For the uncoated LMO at 1.2 V, the molar insertion fraction exceeds the theoretical maximum of the 4 V plateau (y > 1). This over-lithiation explains the complex deionization behavior observed at this elevated voltage. In contrast, the LMO-soft electrode under identical conditions remains in the stable 4 V region, suggesting an interesting effect of the polyelectrolyte coating, even if the amount of retained Li+ (SAC) is slightly reduced.
When a cell voltage of 0.9 V or 0.4 V is applied, the insertion coefficient y remains below 1 for both electrode configurations, with SAC values lower for LMO-soft than for LMO electrodes. In the case of 0.4 V, SAC increases slightly when using LMO-soft electrodes, although it remains much lower than when higher cell voltages are applied. These differences indicate that the polymer coating facilitates the initial accessibility of Li+ ions to the active LMO substrate, attributed to the negatively charged sulfonate groups in PSS.93 However, this phenomenon also hinders the subsequent intercalation of Li+ ions, resulting in diminished retention and accelerated, thus less prolonged, uptake kinetics. In mixed solutions (Fig. 3B), a similar trend is observed for both electrode configurations. Initial uptake and subsequent release (minimum and maximum in conductivity, respectively, when 1.2 V is applied) are accentuated in the case of LMO-soft, consistent with observations in pure solutions.
Considering the differences in ion affinity between AC and LMO, it is expected that the ratio of LMO to AC will be crucial for selectively capturing cations. This becomes particularly important in the desalination of LiCl or LiCl/NaCl solutions. Results for the AC:LMO-soft electrodes show that the amount of Li+ removed decreases as the ratio AC:LMO-soft is raised (Fig. 3C). This confirms that the LMO component plays a relevant role in Li+ uptake, indicating that intercalation is more favorable than capacitive electrosorption within AC. Conductivity measurements in LiCl/NaCl mixed solutions often display a secondary maximum after the initial minimum (Fig. 2C and F), attributable to Na+ release and specifically associated with the properties of the LMO component (Fig. 3D).
Although the spinel framework is capable of accommodating Na+ ions, as previously reported for non-aqueous sodium cells,94 this process induces significant lattice strain. It triggers a phase transition where the original spinel structure partially converts into a layered phase (NazMnO2). In our aqueous mixed system, the high thermodynamic preference of the LMO structure for Li+ ions leads to the rapid and competitive displacement of any transiently inserted Na+, thereby preserving the spinel framework. This ion-exchange-like mechanism results in the observed Na+ release shortly after the beginning of the uptake stage, manifesting as a secondary conductivity maximum (SI, Fig. S5). When the AC fraction exceeds 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 (AC
60 (AC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LMO), this maximum disappears, demonstrating that AC contributes only through a non-selective capacitive process, where both Na+ and Li+ ions participate in building the EDLs within the carbon pores.
LMO), this maximum disappears, demonstrating that AC contributes only through a non-selective capacitive process, where both Na+ and Li+ ions participate in building the EDLs within the carbon pores.
3.4. Tuning the desorption stage for cation selectivity
In electrochemical systems based on LMO, selectivity is influenced by the preferential intercalation capacity of lithium in the material structure, which favors the uptake of Li+ over Na+. In addition, the release of these ions may also exhibit selective behavior. To investigate this phenomenon, ion release was performed in two stages. In the first stage, the short-circuit mode (zero-cell voltage, ZV) was used. This is intended to allow ions less strongly bound to the electrodes to migrate out of them so that in this step, Na+ or just adsorbed (not intercalated) Li+ will be released into the solution. This observation is consistent with previous studies showing that Na+ preferentially adsorbs at the electrode surface without significant intercalation, whereas Li+ is more prone to intercalate into the LMO lattice.78Measurements were performed with LMO and LMO-soft electrodes bathed in a LiCl and NaCl mixed solution (Fig. 4A–D). Samples were taken for ion chromatography analysis at different times during the cycle to identify the ionic species present in the effluent solution at each step of the process. During the uptake phase (steps 1–2), Na+ and Li+ exhibit distinct behaviors: after approximately 50 s (step 2), Na+ is partially released while Li+ remains retained within the electrode (Fig. 4A–C). Meanwhile, Cl− continues to adsorb on both electrode types. In the subsequent zero-voltage ion release (step 3, ∼330 s), both cations are released in small and nearly equal amounts (∼0.15 mM each), regardless of whether LMO or LMO-soft electrodes are used (Fig. 4B and D).
During the reverse-voltage stage (step 4), a stronger release of Li+ occurs (0.62 mM for LMO and 0.52 mM for LMO-soft). In comparison, Na+ release remains limited (0.21 mM for LMO) and within the experimental uncertainty for LMO-soft, consistent with the absence of residual Na+ in the electrode. This stage also causes a higher conductivity increase, confirming that it is primarily associated with Li+ release from the LMO structure, with minimal Na+ contribution.
A transient Na+ release is detected during the early uptake stages (steps 1–2), which reduces the amount of Na+ captured for subsequent release. However, small residual amounts of Na+ are still measurable after steps 3 and 4 for bare LMO. In contrast, the contribution of Na+ during release is almost negligible for LMO-soft electrodes.
In this context, differential release of Na+ and Li+ is a key factor. During the initial ZV ion release step, Na+ is preferentially released, whereas Li+ remains bound due to its stronger interaction with the LMO lattice, consistent with our observations in mixed LiCl/NaCl solutions. When the cell voltage is reversed in the second step, Li+ release becomes predominant. This differential behavior suggests that the polymer coating not only protects the electrode material but also enhances the selectivity by favoring Li+-release over Na+-release.
3.5. Performance of LMO electrodes with asymmetric Li+/Na+ solutions
To evaluate the performance of the LMO and LMO-soft electrodes when employed under highly asymmetric conditions that closely resemble the challenges associated with lithium recovery, the experiments were conducted using a LiCl/NaCl mixture with a composition of 0.5 mM Li+ + 9.5 mM Na+. This composition represents laboratory simulations designed to mimic the low lithium fractions typically encountered in natural brines, where the proportion of Li+ in the total salt content varies between 0.1% and 2%.9The corresponding results are shown in Fig. 5 as relative concentration changes (instead of absolute differences) for improving data visualization. Compared with the results in Fig. 4, LMO-soft electrodes consistently remove about 6% of Li+ during step 1 across all initial concentrations (Fig. 5C), suggesting that the LMO-soft electrode architecture preserves its effectiveness for Li+ separation, even under the highly asymmetric initial conditions. In contrast, bare LMO captures a negligible amount of Li+. The response of the system to Na+ is similar in both cases, with an initial ion uptake followed by a pronounced release (Fig. 4 and 5).
 | ||
| Fig. 5 (A) Relative conductivity in the uptake stage when desalting a 0.5 mM LiCl + 9.5 mM NaCl mixed solution for electrodes with LMO and LMO-soft. Relative sodium and lithium concentration changes in steps 1 and 2 of Fig. 5A for LMO (B) and LMO-soft (C) electrodes. The cell voltage was 1.2 V. | ||
4. Conclusions
This study investigates the use of LMO as a promising active electrode material for selective lithium recovery with hybrid electrochemical ion separation cells, and its combination with a polyelectrolyte layer (LMO-soft electrodes). By exploiting the intrinsic intercalation capabilities of the spinel lattice, LMO successfully overcomes a significant constraint of conventional capacitive deionization using AC electrodes: the limited ability to differentiate between monovalent cations.Voltage-dependent experiments confirmed that Li+ uptake increases markedly with applied potential, reaching >40 mg g−1 at 1.2 V with efficiencies above 90%. The intercalation of Li+ drives the release of Na+, which initially migrated to the LMO electrode, thereby unveiling a replacement mechanism that directly addresses the challenge of selectivity in mixed-ion environments. The adjustment of electrode composition led to a reinforcement of the interplay between capacitive adsorption associated with AC and intercalation mechanisms relative to LMO. Concurrently, low levels of activated carbon to lithium manganese oxide (AC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LMO) ratios have been observed to preserve Li+ intercalation and the associated Na+ displacement.
LMO) ratios have been observed to preserve Li+ intercalation and the associated Na+ displacement.
Moreover, a side result we have found by using combined conductivity and ion chromatography measurements is a sequential release, whereby Na+ is expelled during the zero-voltage step and Li+ during reverse-voltage operation.
The introduction of a polyelectrolyte achieves a dual enhancement in electrode stability and ion separation. While LMO-soft demonstrated diminished uptake capacities, the coating may improve long-term durability and reshapes ion release profiles. Notably, these behaviors persisted under asymmetric feed compositions (0.5 mM Li+ + 9.5 mM Na+), selected as a laboratory simulation for the low-lithium conditions characteristic of brines. Despite the challenging conditions, LMO-soft electrodes demonstrated a consistent ability to recover Li+ while facilitating Na+ release. This finding underscores the robustness of the intercalation–desorption sequence when Li+ is present as a minor component within the salt mixture. This finding directly addresses the practical motivation of developing systems suited for realistic brine environments.
Overall, the results demonstrate the efficacy of combining intercalation-type electrodes with functional polymer coatings to create a versatile platform for selective lithium extraction. The study establishes a linkage between fundamental electrochemical mechanisms and the pressing need for sustainable alternatives to conventional lithium extraction. This connection is achieved by integrating voltage-driven intercalation, ion replacement, polymer-modulated ion release, and compositional tuning. This integrated approach delineates a pathway toward stable, efficient, and scalable electrochemical technologies for lithium recovery from saline resources.
Author contributions
S. Orozco-Barrera: investigation, data curation. J. A. Lirio-Piñar: investigation, data curation. C. Kök: data curation, writing – review & editing. G. R. Iglesias: supervision, methodology, writing – review & editing. A. V. Delgado: methodology, writing – review & editing. V. Presser: supervision, methodology, writing – review & editing. S. Ahualli: supervision, methodology, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
All the data, including raw data, will be shared via the Open Science Framework at Digibug at https://digibug.ugr.es/handle/10481/25025.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ya00009f.
Acknowledgements
Financial support of this investigation by the grant PID2023-151881OB-I00 (AEI/10.13039/501100011033/Unión Europea Next Generation EU/PRTR) is gratefully acknowledged. J. A. L. P. acknowledges FPU2023 (ref. FPU23/03086) funding by MICIU (España). We acknowledge support for the eLiFlow project by the European Union from the European Regional Development Fund (EFRE) and the State of Saarland, Germany.References
- L. Kavanagh, J. Keohane, G. Cabellos, A. Lloyd and J. Cleary, Resources, 2018, 7, 57 CrossRef.
- C. Tabelin, J. Dallas, S. Casanova, T. Pelech, G. Bournival, S. Saydam and I. Canbulat, Miner. Eng., 2021, 163, 106743 CrossRef CAS.
- W. Chen, J. Liang, Z. Yang and G. Li, Innovative Solutions for Energy Transitions, 2019, vol. 158, pp. 4363–4368 Search PubMed.
- X. Zhang, A. Han and Y. Yang, J. Mater. Chem. A, 2020, 8, 22455–22466 RSC.
- P. Choubey, M. Kim, R. Srivastava, J. Lee and J. Lee, Miner. Eng., 2016, 89, 119–137 CrossRef CAS.
- P. Meshram, B. Pandey and T. Mankhand, Hydrometallurgy, 2014, 150, 192–208 CrossRef CAS.
- P. Ji, Z. Ji, Q. Chen, J. Liu, Y. Zhao, S. Wang, F. Li and J. Yuan, Sep. Purif. Technol., 2018, 207, 1–11 CrossRef CAS.
- F. Brandt and R. Haus, Miner. Eng., 2010, 23, 659–661 CrossRef CAS.
- M. Vera, W. Torres, C. Galli, A. Chagnes and V. Flexer, Nat. Rev. Earth Environ., 2023, 4, 149–165 CrossRef CAS.
- Y. Zhang, Y. Hu, L. Wang and W. Sun, Miner. Eng., 2019, 139, 105868 CrossRef CAS.
- M. Floyd, Master’s thesis, Utrecht University, 2021.
- J. R. Rumble, CRC Handbook of Chemistry and Physics, CRC press Boca Raton, FL, 102 edn, 2017 Search PubMed.
- F. R. Spellman, The science of lithium, CRC Press, 2023 Search PubMed.
- D. Torres, K. Pérez, F. Galleguillos Madrid, W. Leiva, E. Gálvez, E. Salinas-Rodríguez, S. Gallegos, I. Jamett, J. Castillo, M. Saldana and N. Toro, Metals, 2024, 14, 1095 CrossRef CAS.
- S. Mousavinezhad, S. Nili, A. Fahimi and E. Vahidi, Resour., Conserv. Recycl., 2024, 205, 107583 CrossRef CAS.
- M. A. Alkhadra, X. Su, M. E. Suss, H. Tian, E. N. Guyes, A. N. Shocron, K. M. Conforti, J. P. de Souza, N. Kim, M. Tedesco, K. Khoiruddin, I. G. Wenten, J. G. Santiago, T. A. Hatton and M. Z. Bazant, Chem. Rev., 2022, 122, 13547–13635 CrossRef CAS PubMed.
- P. Srimuk, X. Su, J. Yoon, D. Aurbach and V. Presser, Nat. Rev. Mater., 2020, 5, 517–538 CrossRef CAS.
- C. Kök, L. Wang, J. Ruthes, A. Quade, M. Suss and V. Presser, Energy Environ. Mater., 2024, 7, 517–538 Search PubMed.
- S. Porada, R. Zhao, A. van der Waals, V. Presser and P. M. Biesheuvel, Prog. Mater. Sci., 2013, 58, 1388–1442 CrossRef CAS.
- P. M. Biesheuvel, H. V. M. Hamelers and M. E. Suss, Colloid Interface Sci. Commun., 2015, 9, 1–5 CrossRef CAS.
- B. P. Jia and W. Zhang, Nanoscale Res. Lett., 2016, 11, 64 CrossRef PubMed.
- J. Oladunni, J. H. Zain, A. Hai, F. Banat, G. Bharath and E. Alhseinat, Sep. Purif. Technol., 2018, 207, 291–320 CrossRef CAS.
- A. Khalil, S. Mohammed, R. Hashaikeh and N. Hilal, Desalination, 2022, 528, 115611 CrossRef CAS.
- J. Farahbakhsh, F. Arshadi, Z. Mofidi, M. Mohseni-Dargah, C. Kök, M. Assefi, A. Soozanipour, M. Zargar, M. Asadnia, Y. Boroumand, V. Presser and A. Razmjou, Desalination, 2024, 575, 117249 CrossRef CAS.
- A. Siekierka, M. Bryjak, A. Razmjou, W. Kujawski, A. Nikoloski and L. Dumée, Membranes, 2022, 12, 343 CrossRef CAS PubMed.
- L. Wang, K. Frisella, P. Srimuk, O. Janka, G. Kickelbick and V. Presser, Sustainable Energy Fuels, 2021, 5, 3124–3133 RSC.
- S. Arnold, L. Wang, R. Mertens, S. Wieczorek and V. Presser, Sep. Purif. Technol., 2025, 367, 132770 CrossRef CAS.
- S. Kim, J. Lee, J. S. Kang, K. Jo, Y. E. Sung and J. Yoon, Chemosphere, 2015, 125, 50–56 CrossRef CAS PubMed.
- R. Trócoli, C. Erinmwingbovo and F. La Mantia, ChemElectroChem, 2017, 4, 143–149 CrossRef.
- D. Weng, H. Duan, Y. Hou, J. Huo, L. Chen, F. Zhang and J. Wang, Prog. Nat. Sci.: Mater. Int., 2020, 30, 139–152 CrossRef CAS.
- T. Zhang, D. Li, Z. Tao and J. Chen, Prog. Nat. Sci.: Mater. Int., 2013, 23, 256–272 CrossRef.
- R. Pitchai, V. Thavasi, S. Mhaisalkar and S. Ramakrishna, J. Mater. Chem., 2011, 21, 11040–11051 RSC.
- A. Hashem, S. Abbas, X. Hou, A. Eid and A. Abdel-Ghany, Heliyon, 2019, 5, e02027 CrossRef CAS PubMed.
- T. Eriksson, Acta Univ. Ups., 2001, 53 Search PubMed.
- K. Nakajima, F. Souza, A. Freitas, A. Thron and R. Castro, Chem. Mater., 2021, 33, 3915–3925 CrossRef CAS.
- S. Ahualli, S. Orozco-Barrera, M. D. Fernandez, A. V. Delgado and G. R. Iglesias, Polymers, 2019, 11, 1556 CrossRef CAS PubMed.
- L. Wang, Y. Liang and L. Zhang, Environ. Sci. Technol., 2020, 54, 5874–5883 CrossRef CAS PubMed.
- J. A. Lirio-Piñar, J. Calvo and S. Ahualli, Phys. Rev. E, 2024, 110, 034610 CrossRef PubMed.
- P. A. Fritz, R. M. Boom and K. Schroen, Sep. Purif. Technol., 2019, 220, 145–151 CrossRef CAS.
- S. Ahualli, G. R. Iglesias, M. M. Fernandez, M. L. Jimenez and A. V. Delgado, Environ. Sci. Technol., 2017, 51, 5326–5333 CrossRef CAS.
- G. R. Iglesias, S. Ahualli, M. M. Fernandez, M. L. Jimenez and A. V. Delgado, Environ. Sci.: Water Res. Technol., 2019, 5, 873–883 RSC.
- G. R. Iglesias, S. Ahualli, A. V. Delgado, P. M. Arenas-Fernández and M. M. Fernández, J. Power Sources, 2020, 453, 227840 CrossRef CAS.
- Y. Liu, Z. Li, X. Liu, Z. Chen, D. Fu, F. Fan, H. Xu and X. Wang, Sep. Purif. Technol., 2025, 364, 132343 CrossRef CAS.
- X. Gao, A. Omosebi, J. Landon and K. L. Liu, Energy Environ. Sci., 2015, 8, 897–909 RSC.
- X. Gao, A. Omosebi, N. Holubowitch, A. Liu, K. Ruh, J. Landon and K. Liu, Desalination, 2016, 399, 16–20 CrossRef CAS.
- S. Ahualli, G. R. Iglesias and A. V. Delgado, in Charge and Energy Storage in Electrical Double Layers, ed S. Ahualli and A. V. Delgado, Elsevier, London, Ch. 9, 2018, 169–192 Search PubMed.
- K. Jo, Y. Baek, S. Kim, S. Hong and J. Yoon, Korean J. Chem. Eng., 2020, 37, 1199–1205 CrossRef CAS.
- Z. Li, Y. You, Z. Zhu, L. Wang, S. Ou, J. Xu and M. Yuan, Appl. Surf. Sci., 2025, 682, 161689 CrossRef CAS.
- T. Liu, A. Dai, J. Lu, Y. Yuan, Y. Xiao, L. Yu, M. Li, J. Gim, L. Ma, J. Liu, C. Zhan, L. Li, J. Zheng, Y. Ren, T. Wu, R. Shahbazian-Yassar, J. Wen, F. Pan and K. Amine, Nat. Commun., 2019, 10, 4721 CrossRef CAS PubMed.
- S. Brunauer, L. Deming, W. Deming and E. Teller, J. Am. Chem. Soc., 1940, 62, 1723–1732 CrossRef CAS.
- D. Siderius, J. Evans, P. Iacomi, L. Vanduyfhuys, V. Van Speybroeck, V. Bon and S. Kaskel, Angew. Chem., Int. Ed., 2025, 64, e202513606 CrossRef CAS PubMed.
- M. Thommes, K. Kaneko, A. Neimark, J. Olivier, F. Rodriguez-Reinoso, J. Rouquerol and K. Sing, Pure Appl. Chem., 2015, 87, 1051–1069 CrossRef CAS.
- S. Ahualli, S. Bermudez, F. Carrique, M. L. Jimenez and A. V. Delgado, Polymers, 2020, 12, 2097 CrossRef CAS PubMed.
- S. Orozco-Barrera, K. Wakabayashi, T. Yoshii, H. Nishihara, G. Iglesias, A. Delgado and S. Ahualli, Sep. Purif. Technol., 2025, 354, 129314 CrossRef CAS.
- M. Torkamanzadeh, C. Kök, P. R. Burger, P. Ren, Y. Zhang, J. Lee, C. Kim and V. Presser, Cell Rep. Phys. Sci., 2023, 4, 101661 CrossRef CAS.
- J. Rouquerol, F. Rouquerol, P. Llewellyn, G. Maurin and K. Sing, Adsorption by Powders and Porous Solids: Principles, Methodology and Applications, Academic Press, Amsterdam, 2 edn, 2013 Search PubMed.
- H. Marsh and F. Rodríguez-Reinoso, in Activated Carbon, ed. H. Marsh and F. Rodríguez-Reinoso, Elsevier, Oxford, 2006, pp. 322–365 Search PubMed.
- L. Wang, W. Ding and Y. Sun, Mater. Res. Bull., 2016, 83, 230–249 CrossRef CAS.
- J. Stumme, O. Ashokkumar, S. Dillmann, R. Niestroj-Pahl and M. Ernst, Membranes, 2021, 11, 106 CrossRef CAS PubMed.
- A. Bhat, E. Reale, M. del Cerro, K. Smith and R. Cusick, Water Res.: X, 2019, 3, 100027 CAS.
- A. Kalde, J. Kamp, E. Evdochenko, J. Linkhorst and M. Wessling, Membranes, 2021, 11, 671 CrossRef CAS PubMed.
- T. Richardson, S. Wen, K. Striebel, P. Ross and E. Cairns, Mater. Res. Bull., 1997, 32, 609–618 CrossRef CAS.
- Y. Wang, Y. Shen, Y. Zhang, B. Yue and C. Wu, J. Macromol. Sci., Part B: Phys., 2006, 45, 563–571 CrossRef CAS.
- K. Ali, H. Raza, M. Malik, S. Ibn Shamsah, R. Amna and A. Sarfraz, J. New Mater. Electrochem. Syst., 2020, 23, 1–6 CrossRef.
- H. G. Shurvell, Handbook of Vibrational Spectroscopy, John Wiley & Sons, Ltd., Hoboken, 2006 Search PubMed.
- E. Jarek, Z. Krasinska-Krawet, T. Kruk, L. Lamch, S. Ronka, K. Wilk and P. Warszynski, Colloids Interfaces, 2021, 5, 3 CrossRef CAS.
- Z. Xu and P. Braterman, J. Mater. Chem., 2003, 13, 268–273 RSC.
- X. Wang, G. Feng and M. Ge, J. Mater. Sci., 2017, 52, 6917–6927 CrossRef CAS.
- D. Rana, J. Biswakarma and S. Lustig, ACS Omega, 2024, 9, 38998–39003 CrossRef CAS PubMed.
- V. Siipola, T. Tamminen, A. Källi, R. Lahti, H. Romar, K. Rasa, R. Keskinen, J. Hyväluoma, M. Hannula and H. Wikberg, BioResources, 2018, 13, 5976–6002 CAS.
- I. Janekarn, A. Hunt, Y. Ngernyen, S. Youngme and N. Supanchaiyamat, R. Soc. Open Sci., 2020, 7, 200438 CrossRef CAS PubMed.
- J. Serafin, B. Dziejarski, O. Fonseca-Bermúdez, L. Giraldo, R. Sierra-Ramírez, M. Bonillo, G. Farid and J. Moreno-Piraján, Int. J. Hydrogen Energy, 2024, 86, 662–676 CrossRef CAS.
- E. Salama, M. Samy, H. S. Hassan, S. Mohamed, K. Mensah and M. F. Elkady, Environ. Sci. Pollut. Res., 2024, 31, 44863–44884 CrossRef CAS PubMed.
- SpectraBase, Spectrum: PDADMAC (LdlrNETyCBW), https://spectrabase.com/spectrum/LdlrNETyCBW, (accessed 23 September 2025, 2025) Search PubMed.
- S. Jareansin, P. Sukaam and B. Kusuktham, Polym. Bull., 2019, 76, 4507–4520 CrossRef CAS.
- J. Chen, M. Liu, H. Liu, L. Ma, C. Gao, S. Zhu and S. Zhang, Chem. Eng. J., 2010, 159, 247–256 CrossRef CAS.
- A. B. D. Nandiyanto, R. Oktiani and R. Ragadhita, Indones. J. Sci. Technol., 2019, 4, 97–118 CrossRef.
- F. Marchini, D. Rubi, M. del Pozo, F. Williams and E. Calvo, J. Phys. Chem. C, 2016, 120, 15875–15883 CrossRef CAS.
- N. Kuwata, G. Hasegawa, D. Maeda, N. Ishigaki, T. Miyazaki and J. Kawamura, J. Phys. Chem. C, 2020, 124, 22981–22992 CrossRef CAS.
- B. Xu and S. Meng, J. Power Sources, 2010, 195, 4971–4976 CrossRef CAS.
- J. Zhang, J. Shen, T. Wang, C. Wei, Y. Ma, C. Zhu and Y. Yue, Electrochem. Acta, 2013, 111, 691–697 CrossRef CAS.
- E. Kim, B. Kim and J. Lee, Environ. Eng. Res., 2024, 29, 230677 CrossRef.
- J. Goodenough and K. S. Park, J. Am. Chem. Soc., 2013, 135, 1167–1176 CrossRef CAS PubMed.
- F. Yu, Y. Wang, C. Guo, H. Liu, W. Bao, J. Li, P. Zhang and F. Wang, Crystals, 2022, 12, 317 CrossRef CAS.
- X. Zhu, F. Meng, Q. Zhang, L. Xue, H. Zhu, S. Lan, Q. Liu, J. Zhao, Y. Zhuang, Q. Guo, B. Liu, L. Gu, X. Lu, Y. Ren and H. Xia, Nat. Sustainability, 2021, 4, 392–401 CrossRef.
- Z. I. Radzi, K. H. Arifin, M. Z. Kufian, V. Balakrishnan, S. R. S. Raihan, N. Abd Rahim and R. Subramaniam, J. Electroanal. Chem., 2022, 920, 116623 CrossRef.
- G. Tan, S. Wan, J. Chen, H. Yu and Y. Yu, Adv. Mater., 2024, 36, 2310657 CrossRef CAS PubMed.
- Q. Xia, X. Lei, F. Yue, W. Liu, C. Xu, J. Xu, M. Yang, X. Huang, D. Su and H. Xia, Nano Lett., 2025, 25, 7892–7899 CrossRef CAS PubMed.
- A. Siekierka, Sep. Purif. Technol., 2020, 236, 116234 CrossRef CAS.
- A. Siekierka, Desalination, 2022, 527, 115569 CrossRef CAS.
- Y. Wu, P. Shi, Y. Zhong and R. Cai, Energy Fuels, 2023, 4083–4093 CrossRef CAS.
- K. Lin, S. Yang, Z. Shi, Q. Fan, Z. Liu and L. Liu, J. Power Sources, 2022, 520, 230768 CrossRef CAS.
- H. Liu, K. Chen, C. Fang and C. Chiu, Electrochim. Acta, 2021, 375, 137915 CrossRef CAS.
- N. Yabuuchi, M. Yano, S. Kuze and S. Komaba, Electrochim. Acta, 2012, 82, 296–301 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |




