 Open Access Article
Open Access ArticleSynergistic effects of microwave irradiation and CeF3 surface coating of lithium titanate for stable, high-capacity, and high-rate lithium-ion batteries
Aderemi B.
Haruna
a,
Seromo
Podile
a,
Itumeleng
Seotsanyana-Mokhosi
a,
Thapelo
Mofokeng
a,
Alexander
Roberts
b,
Jennifer
Johnstone-Hack
c,
Paul
Shearing
def and
Kenneth I.
Ozoemena
 *a
*a
aMolecular Sciences Institute, School of Chemistry, University of the Witwatersrand, Johannesburg 2050, South Africa. E-mail: Kenneth.ozoemena@wits.ac.za
bCentre for E-Mobility and Clean Growth, Coventry University, Coventry CV1 2TL, UK
cSchool of Chemical, Materials and Biological Engineering, Sir Robert Hadfield Building, The University of Sheffield, Mappin Street, Sheffield S1 3JD, UK
dDepartment of Engineering Science, University of Oxford, Parks Road, Oxford OX1 3PJ, UK
eElectrochemical Innovation Lab, Department of Chemical Engineering, University College London, London WC1E 7JE, UK
fThe ZERO Institute, University of Oxford, Oxford OX2 0ES, UK
First published on 29th December 2025
Abstract
The low theoretical specific capacity and rapid capacity degradation, especially during long cycling, have been the key challenges to the practical application of lithium titanate (Li4Ti5O12) as an anode material for stable, high-energy and high-power density lithium-ion batteries (LIBs). This study reports a novel strategy of the synergy of surface lattice plane engineering by microwave irradiation and CeF3 surface coating to synthesize a high-specific-capacity, high-rate and durable LTO–CeF3–mw anode material for LIBs. The innovative anode material shows outstanding specific capacity, rate capability and long-term cycle stability. It is of interest to note that the LTO–CeF3–mw anode material has a specific capacity of 191.1 mAh g−1 at 175 mA g−1 (1C), which is greater than the theoretical specific capacity of LTO (175 mAh g−1). Furthermore, LTO–CeF3–mw has specific capacities of 168.3 and 119.9 mAh g−1 at 875 mA g−1 (5C) and 1750 mA g−1 (10C), respectively. Remarkably, it shows a specific capacity of 166.5 mAh g−1 after 1000 cycles at 5C and a capacity retention of 98.9%. This notable electrochemical performance of LTO–CeF3–mw is attributed to the synergy of surface lattice plane engineering by microwave irradiation and CeF3 surface coating that transformed the surface lattice plane (111) of LTO to (220) and (310) in LTO–CeF3–mw, resulting in high surface area which significantly improved mass transport. Therefore, the unique structure of the LTO–CeF3–mw anode material is a key development for achieving safe, durable, high-energy and high-power lithium-ion batteries, with potential applications in large-scale energy storage.
1. Introduction
Spinel lithium titanate, Li4Ti5O12 (LTO), has been considered the preferred alternative to the current state-of-the-art graphite anode material for lithium-ion batteries (LIBs). This is because of its great advantages, which include its flat high working voltage (∼1.55 V versus Li/Li+), which is above the reduction potential of most organic electrolytes. Therefore, the formation of a solid electrolyte interphase (SEI) and lithium dendrites can be suppressed, enhancing the safety of lithium-ion batteries.1,2 Furthermore, LTO shows negligible volume change (zero strain) during lithium insertion and extraction, which leads to excellent cyclability and safety.3 Unfortunately, LTO has not been widely used in practical applications because it has a low theoretical capacity (175 mA h g−1),3 poor capacity at high C-rates because of its inherent low electronic conductivity and poor Li+ diffusion coefficient,4–6 and capacity degradation during long cycling charge–discharge process.7 There have been several strategies reported to mitigate these drawbacks,6,8–13 and of these strategies, the introduction of Ti3+ is the most reported.14–26 The LTO (Ti4+) has poor electronic conductivity because of its empty 3d electronic orbital (3d0) and poor ionic conductivity because of its small ionic size. The partial reduction of Ti4+ to Ti3+ to form Ti3+–O–Ti4+ pairs in LTO has been reported to enhance the electron conductivity of LTO via the Ti3+ having electronic configuration [Ar]3d1.16,17,22 However, Aurbach et al.27 reported that Ti4+ is the most stable oxidation state of Ti. Ti4+ is very stable because of its completely filled 3p orbital, while Ti3+ is less stable because it has one electron in its 3d orbital (3d1). Therefore, this research work used simple microwave irradiation and CeF3 surface modification to strategically modify the commercial LTO to retain its more stable Ti4+ without the introduction of the conventional conductive Ti3+. The uniqueness of this study is the synergistic modification strategy that retained the Ti4+ and still enhanced the specific capacity and capacity retention of the modified microwave-coated sample.In this work, CeF3 was used to surface-modify LTO because of its high ionic conductivity and structural stability.28 CeF3 surface-modified LTO has a better stability in LiPF6 electrolyte compared to metal oxides that have lattice oxygen that can produce water which reacts with the LiPF6 salt to produce HF that leads to side reactions. CeF3 was first used to surface-modify LTO (to obtain a product denoted as LTO–CeF3) and subsequently subjected to microwave irradiation (to obtain a product denoted as LTO–CeF3–mw). Both processes were aimed at strategically maintaining the Ti4+ and introducing substitutional (point) defects into the commercial LTO.
This study showed that the strategy introduced led to significant surface lattice plane transformation and point defects. The LTO–CeF3–mw exhibited larger d-spacing than its counterparts investigated in this work. Such lattice expansion is advantageous for smooth Li+ diffusion and thus improves the ionic conductivity. To the best of our knowledge, this is the first time that microwave irradiation and CeF3 have been used to fine-tune the physico-chemical properties of the LTO anode for improved electrochemical performance (in terms of cycling stability, increased specific capacity, and high rate capability).
2. Experimental section
2.1 Synthesis of LTO–CeF3, LTO–m and LTO–CeF3–m
Ammonium fluoride NH3F and cerium nitrate hexahydrate Ce(NO3)3·6H2O were each dissolved in distilled water. Thereafter, LTO powder (Shandong Gelon LIB Co., Ltd China) was submerged and stirred inside the cerium nitrate hexahydrate solution. The ammonium fluoride solution was steadily added to the resulting LTO and cerium nitrate hexahydrate solution. After a constant stir at 80 °C for 5 h, the resulting powder was filtered using a centrifuge. The filtered powder was then calcinated at 400 °C for 5 h in an argon atmosphere. This synthesis method is illustrated in Fig. 1a. The ammonium fluoride and cerium nitrate hexahydrate made a stoichiometric molar ratio of Ce3+/F− = 1/3, with the amount of CeF3 corresponding to ∼2 wt% of the LTO powders (based on previous reports of wt% coat of 1 and 2).28–30 To synthesize microwaved materials, the LTO-p and CeF3-coated LTO were subjected to microwave radiation at 600 watts for 20 min31 as shown in Fig. 1b.2.2 Physical characterizations
Bruker D2 phaser is used to determine the XRD of the materials, and the instrument was operated using Cu-Kα radiation at 30 kV and 10 mA. A Micromeritics Tristar 3000 BET instrument was used to determine the pore sizes of the materials. The samples were degassed at 150 °C for 4 h, with gas adsorption performed in liquid nitrogen (−195 °C). A thermogravimetric analyzer (PerkinElmer 6000) was used to determine how stable and versatile the materials were. The morphological and structural properties of the synthesized materials were determined using HRTEM (JEOL JEM 2100) at 200 Kv; samples were dissolved in ethanol, sonicated for about 20 min and dispersed on a carbon-coated copper grid for analysis. XPS measurements were studied with ESCAlab 250Xi XPS having a monochromatic Al kα radiation source (1486.7 eV). Survey spectra were taken at a pass energy of 100 eV and a high-resolution pass energy of 20 eV. The 514.5 nm line of an argon-ion laser and a Horiba Jobin-Yvon LabRAM HR Raman spectrometer equipped with an Olympus BX41 microscope attachment were used to obtain the Raman spectra. The LabSpec v5 software was used to capture the data.2.3 Electrochemical characterization
The LTO anodes were constructed with 85 wt% active material, 10 wt% carbon black (Super C45), 2.5 wt% CMC and 2.5 wt% SBR. Mixtures of an active material and carbon black were ground into a fine powder and slowly added to a liquid thickening agent in piles. A liquid thickening agent was made by stirring CMC and SBR in distilled water heated to 60 °C. The resulting well-mixed slurry was coated on a copper foil and dried at 105 °C for 12 h in a vacuum oven. The active mass loading of the 4 electrode materials is ∼2.0 mg on a 12 mm diameter disc. The electrochemical tests of all the samples were tested using a coin-type cell (CR 2032) assembled inside an argon-filled glove box. The cell comprised of the as-prepared working electrode, lithium foil as a counter electrode and grade GD 1UM, 47 mm Whatman paper as a separator. The electrolyte used was a 1 M LiPF6 solution in ethylene carbonate (EC)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) diethyl carbonate (DEC)
diethyl carbonate (DEC)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl methylcarbonate (EMC) with a volume ratio of 1
ethyl methylcarbonate (EMC) with a volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The coin cells remained in the glovebox overnight before they were taken out for electrochemical measurements (CV: 1.0–2.5 V and galvanometry discharge: 1.0–2.5 V) using the BioLogic system (BCS-800 series) instrument.
1. The coin cells remained in the glovebox overnight before they were taken out for electrochemical measurements (CV: 1.0–2.5 V and galvanometry discharge: 1.0–2.5 V) using the BioLogic system (BCS-800 series) instrument.
3. Results and discussion
Fig. 2 compares the TEM images of the four materials. The images suggest that the pristine LTO is somewhat aggregated upon irradiation with microwaves (see LTO–mw). However, upon coating with the CeF3, microwave irradiation leads to well-dispersed CeF3 nanoparticles on the LTO. This is consistent with the SEM-EDS mapping (Fig. S1). The SEM-EDS revealed that the atomic % of Ce in LTO–CeF3 and LTO–CeF3–mw are 1.4 and 1.3%, respectively. The enhanced dispersion of CeF3 nanoparticles on the LTO–CeF3–mw suggests that it will give better Li-ion diffusion/transport kinetics and better protection of the LTO–CeF3–mw electrode surface than others.The HRTEM shows that LTO-p has lattice fringes with spacings of 0.486 nm and 0.493 nm in regions (I) and (II), respectively, as shown in Fig. S2. The observed spacing in these regions is attributed to the lattice plane (111) of the spinel Li4Ti5O12 phase.32 This confirms the existence of only Li4Ti5O12 in the LTO-p sample. The selected area electron diffraction (SAED) patterns of regions (I) and (II), with all these regions being mono-crystalline, and this is seen through their discrete bright spots that are orderly arranged in a grid pattern.
It is pertinent to note that microwave irradiation introduced the LTO surface lattice planes (220), (310) and (222) in the microwave-irradiated sample LTO–mw, as shown in Fig. 3A. Rikarte et al.33 reported a similar occurrence where the LTO surface plane (111) is transformed to surface plane (222) and anatase TiO2 plane (101). The LTO–mw lattice planes (220), (222) and (310) have lattice fringe spacings of 0.280, (0.238 and 0240) and 0.267 nm, respectively. These lattice fringe spacings are assigned to the lattice planes using the “crystallographic information file (cif)” of Li4Ti5O12. The SAED images of all the regions indicate the single crystalline nature of the cubic spinel.34
Fig. 3B shows that LTO–CeF3 contains anatase TiO2, CeF3 and LTO, as demonstrated by lattice fringe spacings: 0.334 nm corresponding to the TiO2 anatase (101) plane in region (I), 0.312 and 0.321 nm corresponding to the CeF3 (111) plane in regions (II) and (III), respectively,35 and 0.416 nm corresponding to the LTO (111) plane in region (IV).36 The presence of TiO2 in sample LTO–CeF3 shows that CeF3 might have not only physically covered the surface of the LTO but has chemically reacted with the LTO to produce the TiO2, as in the chemical reaction in eqn (1). This implies that CeF3 leads to both surface coating and bulk doping, which is a normal behaviour for its cousin CeO2.
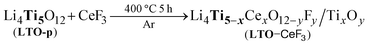 | (1) |
Surface coating and doping have been common strategies for defect engineering in electrode materials such as LTO to improve their structure and surface properties for enhanced electrochemical performances. In this research work, the CeF3 surface modification is used to introduce cation and anion lattice defects (Ce and F, respectively) into the LTO lattice structure. These lattice defects in the coated samples LTO–CeF3 and LTO–CeF3–mw are revealed in the HRTEM and XPS analysis results below. The CeF3-coated electrode samples are expected to give faster Li+ kinetics in electrochemistry.
Furthermore, it is interesting to note that the LTO lattice plane (111) in the LTO–CeF3 sample has a decreased lattice spacing of 0.416 nm compared to 0.486 nm in the LTO-p lattice plane (111) (Table 1). The observed SAED images indicate that anatase TiO2 and CeF3 have a mono-crystalline nature. Fig. 3C indicates that LTO–CeF3–mw has LTO lattice planes (310) in region (I) and (220) in region (II) having lattice fringe spacings of 0.267 and 0.299 nm, respectively, while region (III) has the TiO2 anatase lattice plane (101) with lattice fringe spacings of 0.349 nm and region (IV) has the CeF3 lattice plane (111) with lattice fringe spacing of 0.317 nm. The SAED images of the regions on this sample are single crystalline. It can be concluded that the coated sample LTO–CeF3 and microwave-irradiated coated sample LTO–CeF3–mw have a dual-phase LTO–TiO2 composite. LTO–CeF3–mw also shows grain boundary (GB).
| Lattice plane | LTO (111) | LTO (111) | LTO (220) | LTO (222) | LTO (222) | LTO (310) | CeF3 (111) | CeF3 (111) | TiO2 |
|---|---|---|---|---|---|---|---|---|---|
| LTO-p (nm) | 0.486 | 0.493 | |||||||
| LTO–mw (nm) | 0.280 | 0.240 | 0.238 | 0.267 | |||||
| LTO–CeF3 (nm) | 0.416 | 0.312 | 0.321 | 0.334 | |||||
| LTO–CeF3–mw (nm) | 0.299 | 0.267 | 0.317 | 0.349 |
Table 1 shows that LTO–CeF3–mw has lattice fringe spacing of 0.299 nm for lattice plane LTO (220) which is greater than that of LTO–mw: 0.280 nm. Furthermore, LTO–CeF3–mw has larger lattice fringe spacing of 0.317 and 0.349 nm, respectively, for lattice planes CeF3 (111) and TiO2 (101) than that of LTO–CeF3: CeF3 (111) and TiO2 (101) that has 0.312 and 0.334 nm, respectively. These lattice expansions of surface lattice planes LTO (220), CeF3 (111) and TiO2 are present because of the synergy between microwave irradiation and CeF3 surface modification of the LTO-p.37 These improved properties of the microwave-irradiated coated sample LTO–CeF3–mw are because of the presence of the point defects that have better microwave irradiation absorption and thus resulted in higher heating rate and temperature than the stoichiometry Ti5 of LTO–mw in the same microwave irradiation operation parameters.38
Fig. 4A shows Raman spectra of the four samples. All the materials contain Raman vibration modes at 236, 343, 429, 678, and 770 cm−1 which conform with the A1g + Eg + 3F2g mode of the spinel LTO structure.39 The vibration bands at 678 and 770 cm−1 resulted from the Ti–O stretch vibration of TiO6 octahedra. The bands at 343 and 429 cm−1 are linked to the Li–O stretch vibration of LiO4 or LiO6 tetrahedral, and the 236 cm−1 band originates from O–Ti–O.40,41 It is interesting to note that relative to the pristine sample (LTO-p), both the Ti–O and O–Ti–O vibration bands in LTO–mw, LTO–CeF3 and LTO–CeF3–mw did not show any shift. This shows that there is no change in the oxidation state of the Ti in the modified samples (all the samples have the oxidation state of Ti as +4, and there is no partial reduction of the Ti4+ to Ti3+. Furthermore, it is pertinent to note that the coated samples (LTO–CeF3 and LTO–CeF3–mw) show an additional vibration peak at 143 cm−1, which is attributed to anatase TiO2.42 Raman spectra show that the coated samples LTO–CeF3 and LTO–CeF3–mw have both anatase TiO2 and spinel Li4Ti5O12 vibration peaks, which confirm that LTO–CeF3 and LTO–CeF3–mw are dual-phase spinel Li4Ti5O12–TiO2 composites. This is consistent with the HRTEM results.
XPS surface composition analysis (Fig. 4B) shows that all the samples have two characteristic peaks at ≈458.0 and 463.8 eV which relate to the binding energy of Ti 2p3/2 and Ti 2p1/2 of Ti4+ in LTO.43 It is intriguing to see that there is no obvious change in the Ti 2p spectra in the modified samples, and this provides evidence that there was no formation of Ti3+. Furthermore, it is of interest to note that the spin–orbit splitting, i.e., the change in energy between the Ti 2p3/2 and Ti 2p1/2 peaks (ΔETi![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2p = E2p1/2 − E2p3/2) is 5.8 eV, which shows that Ti oxidation state is +4 (Ti4+). These findings correlate with the Raman results. Fig. 4C shows the Ce 3d peaks of LTO–CeF3 and LTO–CeF3–mw in the range of 870 to 920 eV which confirms the presence of Ce in the CeF3 surface-modified samples.
2p = E2p1/2 − E2p3/2) is 5.8 eV, which shows that Ti oxidation state is +4 (Ti4+). These findings correlate with the Raman results. Fig. 4C shows the Ce 3d peaks of LTO–CeF3 and LTO–CeF3–mw in the range of 870 to 920 eV which confirms the presence of Ce in the CeF3 surface-modified samples.
Crystallographic structures of the materials were evaluated using X-ray powder diffraction, as shown in Fig. 4E. All the diffraction peaks of the materials can be indexed to (111), (311), (400), (331), (333), (440), (531) and (533) of standard cubic spinel Li4Ti5O12 (JCPDS Card No. 49-0207) with the Fd3m space group.44,45 No additional reflection peaks are observed, indicating the absence of impurities and diffraction peaks associated with CeF3 in the coated samples (LTO–CeF3 and LTO–CeF3–m) are not detected, which may be due to relatively low CeF3 content (∼2 wt%).46 Also, this indicates that the microwave irradiation and CeF3 surface modification do not change the spinel structure of the LTO. However, the lattice plane (111) is the most intense peak in the samples’ diffraction patterns, which signifies that all four samples have normal spinel structures. The expanded (111) peak region (Fig. 4F) shows some interesting findings that should be emphasized. First, in the LTO–CeF3, the peak (111) shifted to the higher 2θ degree position relative to that of the LTO-p. This indicates that there is lattice contraction in the coated sample LTO–CeF3. This is consistent with the results of the HRTEM which show that there is a decrease in lattice spacing from 0.486 nm in LTO (111) of LTO-p to 0.416 nm in that of LTO–CeF3. It is interesting to note that the lattice contracted instead of expanding, despite the introduction of Ce3+, which has a larger ionic radius of 1.07 Å than that of Ti4+ (0.605 Å). The lattice contraction in LTO–CeF3 may have occurred because the bond strength of Ce–O is greater than that of Ti–O in the octahedral site, and the Ce–O has higher octahedral site preference energies (OPE) than Ti–O which results in a decrease in the bond length.47,48
Second, in LTO–mw, the peak (111) shifted to the higher 2θ degree position relative to that of the LTO-p. Again, this indicates that there is lattice contraction in the microwave-irradiated sample LTO–mw. The shift to the higher angle may have resulted from the microwave irradiation-induced lattice distortion, as shown in the HRTEM image (Fig. 3A, region I). Nozariasbmarz et al.49 proposed that the fast heating and high electric field of microwave irradiation can distort the atoms from their lattice sites and thus cause lattice strain28 which resulted in the shift to the higher 2θ degree position. The lattice contraction is corroborated by the HRTEM lattice d-spacing.
Third, in LTO–CeF3–mw, the peak (111) shifted to the lower 2θ degree position relative to the LTO-p. This indicates that there is lattice expansion in the LTO–CeF3–mw. It is interesting to note that unlike the microwave sample (LTO–mw) and non-microwave-irradiated coated sample (LTO–CeF3) that show lattice contraction, the microwave-irradiated coated sample (LTO–CeF3–mw) shows lattice expansion. The lattice expansion of the microwave-irradiated coated sample is caused by the synergetic effect of the presence of point defects and electromagnetic irradiation. Though the coated sample LTO–CeF3 has point defects but experienced lattice contraction, this may be because of the absence of microwave irradiation. Furthermore, the microwave sample LTO–mw also experienced lattice contraction because of the absence of the point defects. Hence, the lattice expansion occurs when there is a combined use of point defects and microwave irradiation. Yu et al.50 reported that point defects are better microwave absorbers. Thus, it can be proposed that the point defects in LTO–CeF3–mw sample enabled better microwave absorption and thus caused its lattice expansion relative to the lattice contraction in LTO–mw that has no point defects. It may therefore be concluded that the lattice expansion in the LTO–CeF3–mw sample resulted from the microwave irradiation via the point defects. Hence, it is expected that LTO–CeF3–mw electrode material will have the smoothest Li+ diffusion. These results are also in agreement with the calculated lattice constants of the four samples using the Bragg's law (nλ = 2d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin
sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ). Table S1 shows the calculated lattice constants of the LTO samples using the 2θ degree position of the most intense peak (111). The calculated lattice constants for LTO-p, LTO–mw, LTO–CeF3 and LTO–CeF3–mw are 8.3699, 8.3609, 8.3635 and 8.3762 Å, respectively. These results are comparable with the literature reports.51,52 The lattice constants of the LTO–mw (8.3609 Å) and LTO–CeF3 (8.3635 Å) are less than that of the LTO-p (8.3699 Å), which confirms the lattice contraction as evidenced in Fig. 4F by the shift of the LTO–mw and LTO–CeF3 peaks (111) to the higher 2θ degree position relative to that of LTO-p. The lattice constant of the LTO–CeF3–mw (8.3762 Å) is more than that of the LTO-p (8.3699 Å), which confirms the lattice expansion as evidenced in Fig. 4F by the shift of the LTO–CeF3–mw peak (111) to the lower 2θ degree position relative to that of LTO-p.
θ). Table S1 shows the calculated lattice constants of the LTO samples using the 2θ degree position of the most intense peak (111). The calculated lattice constants for LTO-p, LTO–mw, LTO–CeF3 and LTO–CeF3–mw are 8.3699, 8.3609, 8.3635 and 8.3762 Å, respectively. These results are comparable with the literature reports.51,52 The lattice constants of the LTO–mw (8.3609 Å) and LTO–CeF3 (8.3635 Å) are less than that of the LTO-p (8.3699 Å), which confirms the lattice contraction as evidenced in Fig. 4F by the shift of the LTO–mw and LTO–CeF3 peaks (111) to the higher 2θ degree position relative to that of LTO-p. The lattice constant of the LTO–CeF3–mw (8.3762 Å) is more than that of the LTO-p (8.3699 Å), which confirms the lattice expansion as evidenced in Fig. 4F by the shift of the LTO–CeF3–mw peak (111) to the lower 2θ degree position relative to that of LTO-p.
The adsorption isotherms (Fig. S3) of all the samples show a type III profile. The BET parameters for the LTO samples are presented in Table S2. Nitrogen gas adsorption results reveal that the surface area of LTO-p, LTO–mw, LTO–CeF3 and LTO–CeF3–mw are 4.18, 2.29, 3.89 and 5.28 (m2 g−1), respectively. The microwave-irradiated coated sample LTO–CeF3–mw has the largest BET surface area compared with the surface-coated LTO–CeF3 sample and microwave-irradiated sample LTO–mw. LTO–CeF3–mw has a higher surface area than LTO–mw because the LTO–CeF3–mw sample has defects that enhanced the absorption of microwave irradiation, allowing it to absorb more energy (heat), which resulted in smaller particle sizes and, hence, a higher surface area than the other 3 samples. Consequently, the LTO–CeF3–mw electrode has the largest surface area and it is expected to have more Li+ insertion sites and effective electrolyte contact for faster Li+ kinetics.
3.1 Electrochemical measurements
| Li4Ti5O12 + 3Li+ + 3e− → Li7Ti5O12 | (2) |
The redox peaks of the coated electrode materials LTO–CeF3 and LTO–CeF3–mw are narrower (Fig. 5B) and have higher current responses than those of the uncoated electrode materials LTO-p and LTO–mw (Fig. 5A). These narrower redox peaks indicate the fast kinetics and remarkable efficiency of the insertion/extraction process of Li+ in the spinel lattice, which may be attributed to the presence of point defects in these two samples. Furthermore, the CV for the coated samples did not show additional redox peaks around 1.70/2.05 V for the Li+ insertion/extraction within the anatase TiO2 lattices, unlike some previous reports.4,53,54 This suggests a very weak electrochemical activity of TiO2 because of the low amount of TiO2 in these coated LTO samples.
Table 2 shows (i) the peak-to-peak separation voltage (Epp/V), which indicates the electron transfer kinetics electrode materials; (ii) the peak heights of the anodic (Ipa/mA) and cathodic (Ipc/mA) curves, which confirm the mass transport and redox dynamics, and (iii) the ratio of the peak current (Ipa/Ipc), which shows the reversibility. The coated electrode materials LTO–CeF3 and LTO–CeF3–mw have the lowest Epp of 0.15 V compared with the uncoated materials LTO (0.20 V) and LTO–m (0.22 V). This means that the CeF3 coating improves the electron transfer kinetics of LTO compared to uncoated LTO materials. This electrochemical improvement may be attributed to the presence of point defects in these two samples. These results show that microwave irradiation does not affect the electron transfer kinetics of the materials. Furthermore, the point defects improve the mass transport in the coated materials as shown by the higher current response of the coated materials. However, the microwave-irradiated coated (LTO–CeF3–mw) electrode has the highest values of Ipa and Ipc (mass transport) because of the synergic effect of the CeF3 coating and microwave irradiation that led to better CeF3 surface coat, as established by HRTEM above (Fig. 2D). Despite this, the Ipa/Ipc ratio suggests that neither the microwave treatment nor the point defects significantly affect the reversibility of the redox processes (i.e., Coulombic efficiency). Nevertheless, the LTO–mw shows a larger/broader profile which signifies larger Li+ storage. Furthermore, the CV is used to quantify the surface capacitance of (220) and (310) planes by the empirical power-law relationship between peak current and scan rate in the cyclic voltammograms (CVs) of the LTO-p, LTO–mw, LTO–CeF3 and LTO–CeF3–mw at 0.1, 0.3, 0.5 and 1.0 mV s−1 (Fig. S5). Therefore, using power-law equation:
| i = avb | (3) |
Log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i = b i = b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v + log v + log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a | (4) |
| i (V) = k1v + k2v1/2 | (5) |
| Electrode | E pa/V | E pc/V | ΔE/V | I pa/mA | I pc/mA | I pa/Ipc |
|---|---|---|---|---|---|---|
| LTO-p | 1.67 | 1.47 | 0.20 | 1.91 | 1.57 | 1.22 |
| LTO–mw | 1.68 | 1.46 | 0.22 | 2.28 | 1.58 | 1.44 |
| LTO–CeF3 | 1.65 | 1.50 | 0.15 | 2.53 | 1.94 | 1.30 |
| LTO–CeF3–mw | 1.65 | 1.50 | 0.15 | 2.59 | 1.98 | 1.31 |
Rearranging eqn (5)
| i (V)/v1/2 = k1v1/2 + k2 | (6) |
Hence, the values of k1 and k2 may be used to separate the fraction of the current result from diffusion-controlled and non-diffusion-controlled, respectively, at fixed voltages. Therefore, Fig. S7 shows that the CV at 0.1 mV m−2 of LTO–mw has 35% surface capacitance (non-diffusion-controlled), while LTO–CeF3–mw has 28%. It may thus be proposed that the surface capacitance of the surface lattice planes (220) and (310) of the LTO–mw (35%) is greater than that of LTO–CeF3–mw (28%).
Therefore, the long cyclic stability performance of LTO–mw, LTO–CeF3 and LTO–CeF3–mw electrode materials was carried out at 875 mA g−1 (5C) for 1000 cycles (Fig. 5D). The percentage capacity retention, which is used to assess the cyclic stability, can be calculated from nth cycle (final cycle) discharge capacity divided by the initial discharge capacity multiply by 100. LTO–CeF3–mw delivers the highest initial specific capacity of 168 mA h g−1 and has a high capacity retention of 98.9% (166.1 mA h g−1) after 1000 cycles. While LTO–mw and LTO–CeF3 cells have initial capacities of 148.1 and 145.4 mA h g−1 and capacity retentions of 5.2% (7.7 mA h g−1) and 1.8% (2.6 mA h g−1), respectively, after 1000 cycles. The LTO-mw electrode exhibits poor capacity retention due to its exposed, uncoated surface, which is susceptible to electrolyte attack. The LTO–CeF3 shows lower specific capacity and poor capacity retention because it has only the surface lattice plane (111). Wei57 emphasized the significance of surface orientation to the electrochemical performance of spinel electrode materials and reported similar poor electrochemical performance for the exposed crystal plane (111). The LTO–CeF3–mw has the highest capacity retention of 98.9% because of its higher surface area and increased lattice spacing. It is very important to note this remarkable property of LTO–CeF3–mw in exhibiting significantly low capacity fading during the long-term charge–discharge cycles. Also, Fig. S4 shows that the LTO–CeF3–mw electrode has an outstanding Coulombic efficiency of ∼100%. In addition, the LTO–CeF3–mw electrode has an outstanding Coulombic efficiency of ∼100% for both the 1st and the 1000th cycles. In contrast, LTO–mw has Coulombic efficiency of ∼101% for the 1st and 113% for the 1000th cycle, and LTO–CeF3 has ∼100% Coulombic efficiency for the 1st cycle and 93% for the 1000th cycle. These remarkable performances of LTO–CeF3–mw electrode material could be attributed to the lattice expansion, large surface that has more active sites for Li+ storage and enhanced the effective electrolyte contact for faster Li+ kinetics.
Fig. 6A shows the capacity-rate performances of the LTO–mw, LTO–CeF3 and LTO–CeF3–mw cells at different current densities of 87.5 mA g−1 (0.5C), 175 mA g−1 (1C), 525 mA g−1 (3C), 1750 mA g−1 (10C) and 87.5 mA g−1 (0.5C). The two microwave-irradiated samples show the highest discharge capacities from 0.5C to 10C. This performance could be attributed to the presence of the newly tuned surface lattice planes LTO (220) and (330) introduced by microwave irradiation. The microwave-irradiated non-coated sample LTO–mw has a specific capacity of 196.6, 192.5 and 120.9 mAh g−1 at 87.5 mA g−1 (0.5C), 175 mA g−1 (1C), 1750 mA g−1 (10C), respectively, and the microwave-irradiated coated sample LTO–CeF3–mw has specific capacities of 195.2, 191.1 and 119.9 mAh g−1, respectively. This slightly improved performance of LTO–mw, despite its lower BET surface area, may be related to it having more exposed surface lattice planes (220), (222) and (310) than the sample LTO–CeF3–mw that has only (220) and (310). The surface lattice plane is distinctly different from the BET surface area. The surface lattice plane relates to the geometrical arrangement of atoms in the crystal structure, and their orientation is identified by Miller indices. The orientation is of great importance as it affects the crystal's properties such as surface reactivity. While the BET surface area is the total accessible area and is usually measured by the amount of nitrogen gas physically absorbed at a specific temperature and pressure. At the high charge-rate (C-rate) of 1750 mA g−1 (10C), the LTO–CeF3, LTO–mw and LTO–CeF3–mw have specific capacities of 99.9, 120.9 and 119.9 mAh g−1, respectively. The uncoated microwave-irradiated sample LTO–mw has a higher specific capacity (120.9 mAh g−1) than the coated sample LTO–CeF3 (99.9 mAh g−1). This confirms that the CeF3 surface modification (TiO2 and point defects) is not the cause of the increase in specific capacities of the LTO–mw and LTO–CeF3–mw electrode materials, but because of the tuned surface lattice planes (220), (222) and (310). This finding suggests that the LTO surface lattice planes (220), (222) and (310) are more electrochemically active sites than the lattice plane (111).
It is of interest to note that Fig. 6A shows that the microwave-irradiated samples LTO–mw, and LTO–CeF3–mw have high specific capacities of 192.5 and 191.1 mAh g−1 at 1C which are greater than the theoretical specific capacity of LTO (175 mAh g−1) in the voltage window of 2.5 to 1.0 V.
These higher values, greater than the theoretical specific capacity of 175 mAh g−1 at 1C for LTO specific capacity, are consistent with previous reports, but most of these reports are in the voltage window of 2.5 to 0.01 V58,59 or have dual-phase Li4Ti5O12–TiO2 composites.4,56
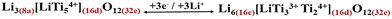 | (7) |
Conventionally, it is thought that the specific capacity of LTO is limited by the number of octahedral sites (16d) that can accommodate Li (i.e. Cth ≈ 175 mAh g−1; eqn (7)).
However, it has been recognized that the tetrahedral sites (8a) can accommodate extra Li (considering that 16c is fully occupied) from the available number of tetravalent Ti (the 2 moles of Ti4+ in the octahedral site (16d) in eqn (3)) (i.e., Cth ≈ 292 mAh g−1; eqn (8)).
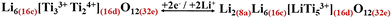 | (8) |
But it should perhaps not be surprising that the tetrahedral sites (8a) may simply accommodate just an extra 0.5 mole of Li instead of 2 moles of Li from the available 2 moles of Ti4+ as observed in our study: microwave treatment (i.e., Cth ≈ 204 mAh g−1; eqn (9)).
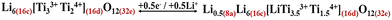 | (9) |
This study is different in that the voltage window used for all the electrochemical measurements is 2.5 to 1.0 V and the LTO–mw anode material that has the highest capacity of 192.5 mAh g−1 at 175 mA g−1 (1C) does not have the dual phase. It is important to note that though LTO–mw has the highest specific capacity at 1C, Fig. 6A and Table 2 show that the conventional limiting factors (Li+ diffusion, Li+ transport and electron transport) are not applicable nor responsible for the high capacity. This is because the LTO–mw has the highest CV peak-to-peak separation voltage (Epp/V), which signifies the highest polarization effect that translates to low kinetics. Amazingly, this is the electrode material that has the highest specific capacity of all the samples and has a specific capacity higher than the theoretical specific capacity. Consequently, this outstanding performance may be related to the LTO–mw having large Li+ storage as shown in the CV profile area. This agrees with the LTO–mw's CV analysis above and Fig. S7 that show its surface capacitance has a non-faradaic (non-diffusion) process of 35% which is greater than that of the LTO–CeF3–mw (28%). This confirms that the surface capacitive of the non-diffusion process’ specific capacity share of LTO–mw is greater than that of LTO–CeF3–mw. This may suggest the reason why the LTO–mw has the highest specific capacity of 192.5 mAh g−1 at 175 mA g−1 (1C) relative to the other samples. Cunha et al.60 have attributed similar higher specific capacity than the theoretical capacity within the voltage window of 2.5 to 1.0 V to a surface-related phenomenon, where the surface of the spinel LTO having different surface lattice planes played a significant role in the Li+ storage properties. That is, the higher specific capacity than the theoretical capacity of LTO within the voltage window of 2.5 to 1.0 V is due to significant surface contributions in Li+ storage. Hence, this higher discharge capacity of LTO–mw can be suggested to be because of the surface lattice planes (220), (222) and (310) having significant contributions in lithium storage.
On the other hand, it is also pertinent to point out that the LTO–CeF3–mw sample has a specific capacity of 191.1 mAh g−1 at 175 mA g−1 (1C) within the voltage window of 2.5 to 1.0 V. This is higher than the theoretical specific capacity. Even though the Raman and HRTEM show the presence of TiO2 in the LTO–CeF3–mw, it can be suggested that the TiO2 is not responsible for the higher specific capacity, as evident by the absence of the CV redox peaks and GCD voltage plateau at 1.70 V/2.08 V that are synonymous with TiO2 contribution to specific capacity.4 This also confirms that the higher discharge capacity may have resulted from Li+ storage at the surface lattice planes (220) and (310). It is impressive to emphasize that all three samples recover to their initial discharge capacities when the rate returns to 87.5 mA g−1 (0.5C). This shows the good reversibility of the modified LTO samples. Furthermore, the performance characteristics (rate capability and cycling performance) of the LTO–CeF3–mw anode material surpass most of the summarized reports categorized as “high performance” by Kazemi et al.61
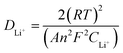 | (10) |
| Z′ = Rs + Rct + σω−1/2 | (11) |
Fig. 7A shows the comparison of rate capacities of LTO–CeF3–mw anode material in this work with LTO anode materials previously reported, while Fig. 7B shows the comparison of capacity retention at 875 mA g−1 (5C) of LTO–CeF3–mw anode material in this work with LTO anode materials.63–69 The compared parameters show that relative to these previous studies, the electrochemical performance of microwave-coated sample, LTO–CeF3–mw, is remarkable.
4. Conclusion
This research work strategically used microwave irradiation and CeF3 surface coating to synthesize the LTO–CeF3–mw anode material for LIBs that has an outstanding specific capacity of 191.1 mAh g−1 at 175 mA g−1 (1C) which is greater than the theoretical specific capacity of LTO (175 mAh g−1). Furthermore, the LTO–CeF3–mw electrode has long-term cycling stability with high-capacity retention of 98.9% after 1000 cycles, high-rate of specific capacity of 119.9 mAh g−1 at 10C and reversible capacity. These top-notch electrochemical performances of the LTO–CeF3–mw can be attributed to the tuned surface lattice planes (220) and (310) from (111), expanded lattice spacing and high surface area. The findings of this study are of great importance to the advancement towards high-energy, high-power and long-life LIBs for electric vehicles' large-scale energy storage.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). The supplementary information includes additional data supporting this study. Fig. S1 presents SEM–EDS elemental mapping of all four samples: (A) LTO-p, (B) LTO–mw, (C) LTO–CeF3, and (D) LTO–CeF3–mw. Fig. S2 shows HRTEM micrographs of LTO-p. Table S1 exhibits the lattice constants of the four samples. Fig. S3 provides N2 adsorption/desorption isotherms, while Table S2 lists the corresponding BET surface area and porosity parameters. Fig. S4 displays galvanostatic charge–discharge profiles for cycle 1 and cycle 1000 at 5C for (A) LTO–CeF3, (B) LTO–mw, and (C) LTO–CeF3-mw. Fig. S5 presents cyclic voltammograms recorded at scan rates of 0.1, 0.3, 0.5, and 1.0 mV s−1 for all samples, and Fig. S6 shows the corresponding log (peak current) versus log (scan rate) plots. Table S3 reports the calculated b-values at different scan rates. Fig. S7 compares the capacitive and diffusion-controlled contribution fractions for (A, B) LTO–mw and (C, D) LTO–CeF3–mw. Tables S4 and S5 show the electrochemical impedance spectroscopy (EIS) fitting parameters before cycling (BOL) and after 1000 cycles (EOL), respectively. See DOI: https://doi.org/10.1039/d5ya00303b.Acknowledgements
The authors are grateful for the financial support from the following: (i) UCL-Wits collaboration Seed Fund, (ii) Fellowship of the Association of the Commonwealth Universities (ACU) for partnership with the Coventry University (UK), and (iii) the DSI-NRF-Wits SARChI Chair in Materials Electrochemistry and Energy Technologies (MEET) (UID No.: 132739). JJH acknowledges funding from EPSRC (EP/T517793/1).References
- B. Yan, M. Li, X. Li, Z. Bai, J. Yang, D. Xiong and D. Li, Novel understanding of carbothermal reduction enhancing electronic and ionic conductivity of Li 4 Ti 5 O 12 anode, J. Mater. Chem. A, 2015, 3, 11773–11781 RSC.
- A. Yu, D. Gong, M. Zhang and Y. Tang, In-situ implanted carbon nanofilms into lithium titanate with 3D porous structure as fast kinetics anode for high-performance dual-ion battery, Chem. Eng. J., 2020, 401, 125834 CrossRef CAS.
- Y.-J. Bai, C. Gong, N. Lun and Y.-X. Qi, Yttrium-modified Li 4 Ti 5 O 12 as an effective anode material for lithium ion batteries with outstanding long-term cyclability and rate capabilities, J. Mater. Chem. A, 2013, 1, 89–96 Search PubMed.
- M. Akhtar, T. Majumder, J.-K. Chang and S. B. Majumder, High-Performance and Safe Hybrid Li-Ion Batteries Based on Li 4 Ti 5 O 12 –TiO 2 (A)–TiO 2 (R)@C Anode and Na 3 V 2 O 2 (PO 4) 2 F–Na 3 V 2 (PO 4) 3 @C Cathode, ACS Sustainable Chem. Eng., 2022, 10, 1390–1397 CrossRef CAS.
- C. Ho, C. V. Li, K. Chan, H. Yung and Y. Tay, Interfacing TiO 2 (B) Nanofibers with Li 4 Ti 5 O 12 Towards Highly Reversible and Durable TiO 2 -based Anode for Li−Ion Batteries, Energy Technol., 2019, 7, 107–112 CrossRef CAS.
- H. Chang, Y.-R. Wu, X. Han and T.-F. Yi, Recent developments in advanced anode materials for lithium-ion batteries, Energy Mater., 2022, 1, 100003 Search PubMed.
- X. Bai, T. Li and Y.-J. Bai, Capacity degradation of Li 4 Ti 5 O 12 during long-term cycling in terms of composition and structure, Dalton Trans., 2020, 49, 10003–10010 RSC.
- A. Lakshmi-Narayana, M. Dhananjaya, C. M. Julien, S. W. Joo and C. V. Ramana, Enhanced Electrochemical Performance of Rare-Earth Metal-Ion-Doped Nanocrystalline Li 4 Ti 5 O 12 Electrodes in High-Power Li-Ion Batteries, ACS Appl. Mater. Interfaces, 2023, 15, 20925–20945 Search PubMed.
- Z. Wang, H. Guo, D. Ning, X. Ma, L. Zheng, D. Smirnov, K. Sun, D. Chen, L. Sun and X. Liu, Tuning Fermi level and band gap in Li 4 Ti 5 O 12 by doping and vacancy for ultrafast Li+ insertion/extraction, J. Am. Ceram. Soc., 2021, 104, 5934–5945 CrossRef CAS.
- A. Sohib, S. Priyono, W. B. Widayatno, A. Subhan, S. N. Sari, A. S. Wismogroho, C. Hudaya and B. Prihandoko, Electrochemical performance of low concentration Al doped-lithium titanate anode synthesized via sol-gel for lithium ion capacitor applications, J. Energy Storage, 2020, 29, 101480 Search PubMed.
- D. Qian, Y. Gu, Y. Chen, H. Liu, J. Wang and H. Zhou, Ultra-high specific capacity of Cr3 + -doped Li4Ti5O12 at 1.55 V as anode material for lithium-ion batteries, Mater. Lett., 2019, 238, 102–106 CrossRef CAS.
- S. Repp, E. Harputlu, S. Gurgen, M. Castellano, N. Kremer, N. Pompe, J. Wörner, A. Hoffmann, R. Thomann, F. M. Emen, S. Weber, K. Ocakoglu and E. Erdem, Synergetic effects of Fe3+ doped spinel Li 4 Ti 5 O 12 nanoparticles on reduced graphene oxide for high surface electrode hybrid supercapacitors, Nanoscale, 2018, 10, 1877–1884 RSC.
- H. Zou, X. Liang, X. Feng and H. Xiang, Chromium-Modified Li 4 Ti 5 O 12 with a Synergistic Effect of Bulk Doping, Surface Coating, and Size Reducing, ACS Appl. Mater. Interfaces, 2016, 8, 21407–21416 Search PubMed.
- K. L. Salvatore, M. N. Vila, G. Renderos, W. Li, L. M. Housel, X. Tong, S. C. McGuire, J. Gan, A. Paltis, K. Lee, K. J. Takeuchi, A. C. Marschilok, E. S. Takeuchi and S. S. Wong, Probing the Physicochemical Behavior of Variously Doped Li 4 Ti 5 O 12 Nanoflowers, ACS Phys. Chem. Au, 2022, 2, 331–345 Search PubMed.
- Z. Xie, Q. Song, H. Xie, H. Yin and Z. Ning, Chemically driven synthesis of Ti3+ self-doped Li 4 Ti 5 O 12 spinel in molten salt, J. Am. Ceram. Soc., 2021, 104, 753–765 CrossRef CAS.
- K. Liang, H. He, Y. Ren, J. Luan, H. Wang, Y. Ren and X. Huang, Ti3+ self-doped Li4Ti5O12 with rich oxygen vacancies for advanced lithium-ion batteries, Ionics, 2020, 26, 1739–1747 Search PubMed.
- G. Yang and S.-J. Park, Single-step solid-state synthesis and characterization of Li 4 Ti 5−x Fe x O 12−y (0 ≤ x ≤ 0.1) as an anode for lithium-ion batteries, J. Mater. Chem. A, 2020, 8, 2627–2636 RSC.
- K. L. Salvatore, D. M. Lutz, H. Guo, S. Yue, J. Gan, X. Tong, P. Liu, E. S. Takeuchi, K. J. Takeuchi, A. C. Marschilok and S. S. Wong, Solution-Based, Anion-Doping of Li 4 Ti 5 O 12 Nanoflowers for Lithium-Ion Battery Applications, Chem. – Eur. J., 2020, 26, 9389–9402 Search PubMed.
- M. Ding, H. Liu, J. Zhu, X. Zhao, L. Pang, Y. Qin and L. Deng, Constructing of hierarchical yolk-shell structure Li4Ti5O12-SnO2 composites for high rate lithium ion batteries, Appl. Surf. Sci., 2018, 448, 389–399 CrossRef CAS.
- C. Chen, X. Liu, C. Ai and Y. Wu, Enhanced lithium storage capability of Li 4 Ti 5 O 12 anode material with low content Ce modification, J. Alloys Compd., 2017, 714, 71–78 Search PubMed.
- X. Ji, D. Li, Q. Lu, E. Guo and L. Yao, Electrospinning preparation of one-dimensional Ce3 + -doped Li4Ti5O12 sub-microbelts for high-performance lithium-ion batteries, J. Nanopart. Res., 2017, 19, 393 CrossRef.
- H. Kim, D. Kim, K. Hara, H. Shiiba, Y. Charles-Blin, E. Otal, H. Tanaka, K. Teshima, G. Sánchez-Santolino, R. Ishikawa, Y. Ikuhara and N. Zettsu, Mixed anion effects on structural and electrochemical characteristics of Li 4 Ti 5 O 12 for high-rate and durable anode materials, J. Mater. Chem. A, 2024, 12, 7107–7121 RSC.
- S. Saxena and A. Sil, Role of calcination atmosphere in vanadium doped Li 4 Ti 5 O 12 for lithium ion battery anode material, Mater. Res. Bull., 2017, 96, 449–457 CrossRef CAS.
- T. Subburaj, K. Prasanna, K. J. Kim, P. R. Ilango, Y. N. Jo and C. W. Lee, Structural and electrochemical evaluation of bismuth doped lithium titanium oxides for lithium ion batteries, J. Power Sources, 2015, 280, 23–29 CrossRef CAS.
- G. Yang and S.-J. Park, The formation mechanism of Li4Ti5O12−y solid solutions prepared by carbothermal reduction and the effect of Ti3+ on electrochemical performance, Sci. Rep., 2019, 9, 4774 CrossRef PubMed.
- A. Mahmoud, J. M. Amarilla, K. Lasri and I. Saadoune, Influence of the synthesis method on the electrochemical properties of the Li4Ti5O12 spinel in Li-half and Li-ion full-cells. A systematic comparison, Electrochim. Acta, 2013, 93, 163–172 CrossRef CAS.
- D. Aurbach, Y. Gofer, O. Chusid and H. Eshel, On nonaqueous electrochemical behavior of titanium and Ti4+ compounds, Electrochim. Acta, 2007, 52, 2097–2101 CrossRef CAS.
- X. Ji, X. Dai, F. Wu and H. Jin, Liquid-Phase Integrated Surface Modification to Construct Stable Interfaces and Superior Performance of High-Voltage LiNi0.5Mn1.5O4 Cathode Materials, ACS Sustainable Chem. Eng., 2022, 10, 9850–9859 CrossRef CAS.
- W. Li, X. Li, M. Chen, Z. Xie, J. Zhang, S. Dong and M. Qu, AlF3 modification to suppress the gas generation of Li4Ti5O12 anode battery, Electrochim. Acta, 2014, 139, 104–110 Search PubMed.
- N. Nzimande, A. Haruna, P. Mwonga, B. Rasche, F. Cummings and K. I. Ozoemena, Ceria-Spiderweb Nanosheets Unlock the Energy-Storage Properties in the “Sleeping” Triplite (Mn2(PO4)F), ACS Appl. Energy Mater., 2021, 4, 13085–13097 Search PubMed.
- A. B. Haruna, D. H. Barrett, C. B. Rodella, R. M. Erasmus, A. M. Venter, Z. N. Sentsho and K. I. Ozoemena, Microwave irradiation suppresses the Jahn-Teller distortion in Spinel LiMn2O4 cathode material for lithium-ion batteries, Electrochim. Acta, 2022, 426, 140786 CrossRef CAS.
- J. Yoon, S. Nam, H. Shim, K. Park, T. Yoon, H. Park and S. Hyun, Highly-Stable Li4Ti5O12 Anodes Obtained by Atomic-Layer-Deposited Al2O3, Materials, 2018, 11, 803 Search PubMed.
- J. Rikarte, B. Acebedo, A. Vilalta-Clemente, F. Bonilla, A. J. Wilkinson, M. Galceran, A. Lousa, J. Rubio-Zuazo and M. Á. Muñoz-Márquez, Surface Evolution of Lithium Titanate upon Electrochemical Cycling Using a Combination of Surface Specific Characterization Techniques, Adv. Mater. Interfaces, 2020, 7, 1902164 Search PubMed.
- M. Qin, Y. Li and X.-J. Lv, Preparation of Ce- and La-Doped Li4Ti5O12 Nanosheets and Their Electrochemical Performance in Li Half Cell and Li4Ti5O12/LiFePO4 Full Cell Batteries, Nanomaterials, 2017, 7, 150 CrossRef PubMed.
- S. Liu, Y. Hui, L. Zhu, X. Fan, B. Zou and X. Cao, Synthesis and luminescence properties of CeF3:Tb3+ nanodisks via ultrasound assisted ionic liquid method, J. Rare Earths, 2014, 32, 508–513 CrossRef CAS.
- H. Zhang, P. Liu, F. Li, H. Liu, Y. Wang, S. Zhang, M. Guo, H. Cheng and H. Zhao, Facile Fabrication of Anatase TiO 2 Microspheres on Solid Substrates and Surface Crystal Facet Transformation from {001} to {101}, Chem. – Eur. J., 2011, 17, 5949–5957 Search PubMed.
- S. K. Jha, N. Nakamura, S. Zhang, L. Su, P. M. Smith, X. L. Phuah, H. Wang, H. Wang, J. S. Okasinski, A. J. H. McGaughey and B. Reeja-Jayan, Defect-Mediated Anisotropic Lattice Expansion in Ceramics as Evidence for Nonthermal Coupling between Electromagnetic Fields and Matter, Adv. Eng. Mater., 2019, 21, 1900762 Search PubMed.
- B. Vaidhyanathan, A. P. Singh, D. K. Agrawal, T. R. Shrout, R. Roy and S. Ganguly, Microwave Effects in Lead Zirconium Titanate Synthesis: Enhanced Kinetics and Changed Mechanisms, J. Am. Ceram. Soc., 2001, 84, 1197–1202 Search PubMed.
- N. M. Ncube, W. T. Mhlongo, R. I. McCrindle and H. Zheng, The electrochemical effect of Al-doping on Li 4 Ti 5 O 12 as anode material for lithium-ion batteries, Mater. Today: Proc., 2018, 5, 10592–10601 CAS.
- H. A. Llaín-Jiménez, D. A. Buchberger, M. Winkowska-Struzik, M. Ratyński, M. Krajewski, M. Boczar, B. Hamankiewicz and A. Czerwiński, Correlation between Lithium Titanium Oxide Powder Morphology and High Rate Performance in Lithium-Ion Batteries, Batteries, 2022, 8, 168 Search PubMed.
- T.-F. Yi, J. Shu, Y.-R. Zhu, X.-D. Zhu, C.-B. Yue, A.-N. Zhou and R.-S. Zhu, High-performance Li4Ti5−xVxO12 (0 ≤ x ≤ 0.3) as an anode material for secondary lithium-ion battery, Electrochim. Acta, 2009, 54, 7464–7470 CrossRef CAS.
- J.-Y. Liao, V. Chabot, M. Gu, C. Wang, X. Xiao and Z. Chen, Dual phase Li4Ti5O12–TiO2 nanowire arrays as integrated anodes for high-rate lithium-ion batteries, Nano Energy, 2014, 9, 383–391 CrossRef CAS.
- J. Kim, M. Park, S. M. Hwang, Y. Heo, T. Liao, Z. Sun, J. H. Park, K. J. Kim, G. Jeong, Y. Kim, J. H. Kim and S. X. Dou, Zr4+ Doping in Li4Ti5O12 Anode for Lithium-Ion Batteries: Open Li+ Diffusion Paths through Structural Imperfection, ChemSusChem, 2014, 7, 1451–1457 CrossRef CAS PubMed.
- C. Wu, Y. Wang, G. Ma and X. Zheng, Enhanced rate capability of Li4Ti5O12 anode material by a photo-assisted sol–gel route for lithium-ion batteries, Electrochem. Commun., 2021, 131, 107119 CrossRef CAS.
- W. Qin, H. Liu, J. An and X. Wen, Enhanced Li-ion battery performance of TiO2 nanoparticle-loaded Li4Ti5O12 nanosheet anode using carbon coated copper as current collector, J. Power Sources, 2020, 479, 229090 Search PubMed.
- L. Cheng, X.-L. Li, H.-J. Liu, H.-M. Xiong, P.-W. Zhang and Y.-Y. Xia, Carbon-Coated Li[sub 4]Ti[sub 5]O[sub 12] as a High Rate Electrode Material for Li-Ion Intercalation, J. Electrochem. Soc., 2007, 154, A692 CrossRef CAS.
- D. Li, Y. Liu, W. Zhao, Y. Gao, L. Cao, Y. Liu, W. Wang, L. Yi and T. Qi, Synthesis of Ce modified Li4Ti5O12 using biomass as carbon source, J. Electroanal. Chem., 2019, 851, 113441 CrossRef CAS.
- T.-F. Yi, J.-Z. Wu, M. Li, Y.-R. Zhu, Y. Xie and R.-S. Zhu, Enhanced fast charge–discharge performance of Li4Ti5O12 as anode materials for lithium-ion batteries by Ce and CeO2 modification using a facile method, RSC Adv., 2015, 5, 37367–37376 Search PubMed.
- A. Nozariasbmarz, K. Dsouza and D. Vashaee, Field induced decrystallization of silicon: Evidence of a microwave non-thermal effect, Appl. Phys. Lett., 2018, 112, 093103 CrossRef.
- W. Yu and G. Shao, Morphology engineering of defective graphene for microwave absorption, J. Colloid Interface Sci., 2023, 640, 680–687 CrossRef CAS PubMed.
- E. Temeche, E. Buch, X. Zhang, T. Brandt, A. Hintennach and R. M. Laine, Improved Electrochemical Properties of Li4 Ti5 O12 Nanopowders (NPs) via Addition of LiAlO2 and Li6 SiON Polymer Electrolytes, Derived from Agricultural Waste, ACS Appl. Energy Mater., 2021, 4, 1894–1905 Search PubMed.
- S. U. Muzayanha, A. Purwanto, M. I. Putra and H. K. Aliwarga, The Effect of Modified LTO Synthesis using Salt Assisted Method with Various of Salt Metal Materials, IOP Conf. Ser.:Mater. Sci. Eng., 2021, 1117, 012022 CAS.
- Y. Zhang, M. Zeng, X. Wu, Y. Bai and J. Li, Solvothermal synthesis method for dual-phase Li4Ti5O12/TiO2 composites for high-stability lithium storage, Mater. Res. Express, 2020, 7, 015515 CrossRef CAS.
- L. Noerochim, W. Caesarendra, A. Habib, Widyastuti, Suwarno, Y. L. Ni’mah, A. Subhan, B. Prihandoko and B. Kosasih, Role of TiO2 Phase Composition Tuned by LiOH on The Electrochemical Performance of Dual-Phase Li4Ti5O12-TiO2 Microrod as an Anode for Lithium-Ion Battery, Energies, 2020, 13, 5251 CrossRef CAS.
- P. Ghasemiahangarani, G. Farhan, D. Del Mundo and T. Schoetz, Charge Storage Mechanisms in Batteries and Capacitors: A Perspective of the Electrochemical Interface, Adv. Energy Mater., 2025, 15, 2404704 CrossRef CAS.
- J. H. Jeong, B.-H. Park, G.-W. Lee, K. C. Roh and K.-B. Kim, Phase transformation of spinel Li4Ti5O12 to anatase TiO2 by catalytic delithiation, Energy Storage Mater., 2020, 25, 510–519 CrossRef.
- Y. Wei, K. Tuo, P. Wang, L. Yang, W. Liang, H. Ding, X. Cui and S. Li, Appropriate proportion truncated octahedron LiNi0.5Mn1.5O4 with excellent electrochemical properties for lithium-ion batteries prepared by graphite-assisted calcination method, Ionics, 2020, 26, 6003–6012 CrossRef CAS.
- C. Chen, R. Agrawal and C. Wang, High Performance Li4Ti5O12/Si Composite Anodes for Li-Ion Batteries, Nanomaterials, 2015, 5, 1469–1480 CrossRef CAS PubMed.
- C. Y. Bon, P. Isheunesu, S. Kim, M. Manasi, Y. I. Kim, Y. J. Lee and J. M. Ko, High Capacity and Fast Charge-Discharge Li4Ti5O12 Nanoflakes/TiO2 Nanotubes Composite Anode Material for Lithium Ion Batteries, Energy Technol., 2018, 6, 2461–2468 CrossRef CAS.
- D. M. Cunha, T. A. Hendriks, A. Vasileiadis, C. M. Vos, T. Verhallen, D. P. Singh, M. Wagemaker and M. Huijben, Doubling Reversible Capacities in Epitaxial Li4Ti5O12 Thin Film Anodes for Microbatteries, ACS Appl. Energy Mater., 2019, 2, 3410–3418 CrossRef CAS.
- A. Kazemi, R. Mostaani, M. K. Ravari, M. Ghorbanzadeh, M. Yavarinasab, J. Enferadi, Z. Chen, N. Iqbal, Y. Xiang, W. Liu and A. Davoodi, Enhancing high rate performance of Lithium Titanium Oxide (LTO) anodes: A comprehensive review, J. Power Sources, 2025, 630, 236051 CrossRef CAS.
- A. B. Haruna, P. Mwonga, D. Barrett, C. B. Rodella, R. P. Forbes, A. Venter, Z. Sentsho, P. J. Fletcher, F. Marken and K. I. Ozoemena, Defect-Engineered β-MnO2−δ Precursors Control the Structure–Property Relationships in High-Voltage Spinel LiMn1.5Ni0.5O4−δ, ACS Omega, 2021, 6, 25562–25573 CrossRef CAS PubMed.
- Y. Yin, X. Luo and B. Xu, In-situ self-assembly synthesis of low-cost, long-life, shape-controllable spherical Li4Ti5O12 anode material for Li-ion batteries, J. Alloys Compd., 2022, 904, 164026 CrossRef CAS.
- G. Wang, H. Wang, G. Ma, X. Du, L. Du, P. Jing, Y. Wang, K. Wu, H. Wu, Q. Wang and Y. Zhang, Investigation on process mechanism of a novel energy-saving synthesis for high performance Li4Ti5O12 anode material, J. Energy Chem., 2022, 70, 266–275 Search PubMed.
- G. Yan, X. Xu, W. Zhang, Z. Liu and W. Liu, Preparation and electrochemical performance of P5+-doped Li4Ti5O12 as anode material for lithium-ion batteries, Nanotechnology, 2020, 31, 205402 Search PubMed.
- M. Shen, J. Zhu, S. Sun, D. Chen, F. Liu and J. Chen, Theoretical Prediction and Experimentally Realizing Cathodic Doping of Sulfur in Li4Ti5O12 for Superior Lithium Storage Performance, ACS Appl. Energy Mater., 2021, 4, 5995–6004 CrossRef CAS.
- X. Xiao, L. Liu, L. Zhang, Q. Wang, H. Yan, B. Zhao, L. Wang, L. Ma, Y. Liu and H. An, Electrochemical and transport properties of Te-doped Li4Ti5O12 as anode material for lithium-ion half/full batteries, J. Alloys Compd., 2022, 897, 162744 Search PubMed.
- D. Qian, Y. Gu, S. Guo, H. Liu, Y. Chen, J. Wang, G. Ma and C. Wu, Effect of rich R-TiO2 on the rate and cycle properties of Li4Ti5O12 as anode for lithium ion batteries, J. Energy Chem., 2019, 32, 182–188 CrossRef.
- Y. Zhang, Y. Zhang, L. Huang, Z. Zhou, J. Wang, H. Liu and H. Wu, Hierarchical carambola-like Li4Ti5O12-TiO2 composites as advanced anode materials for lithium-ion batteries, Electrochim. Acta, 2016, 195, 124–133 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |







