Open Access Article
Open Access ArticleNon-thermal plasma upgrading of humidified CO2 into syngas in a dielectric barrier discharge reactor: tuning H2/CO ratios via specific energy input and gas flow rate
Maxwell
Klein
and
Joshua
Jack
 *
*
Department of Civil and Environmental Engineering, University of Michigan, Ann Arbor, MI 48109, USA. E-mail: jdjack@umich.edu
First published on 2nd February 2026
Abstract
This study investigates the non-thermal plasma (NTP)-driven conversion of humidified CO2 into syngas (H2:CO:CO2) using a dielectric barrier discharge (DBD) reactor. NTP enables CO2 and H2O dissociation at near-ambient conditions through high-energy electron collisions, eliminating the need for thermal input or expensive catalysts. While prior studies have examined gas composition and humidity effects, the influence of specific energy input (SEI) and gas flow rate on syngas yield and energy efficiency remains underexplored. Here, we systematically evaluate the relationship between SEI, flow rate, energy efficiency, and H2/CO composition. Experiments conducted in a coaxial DBD reactor demonstrate syngas generation with tunable H2/CO ratios ranging from 0.1 to 0.2 with energy efficiencies ranging from 4.5 to 23.3%. Results indicate that higher flow rates enhance energy efficiency, while increasing SEI leads to a plateau in CO and H2 production, suggesting limits in energy transfer efficiency. Overall, these findings can inform the design of next-generation NTP systems towards a circular carbon economy.
1. Introduction
The Intergovernmental Panel on Climate Change (IPCC) highlights carbon capture and utilization (CCU) technologies as critical components of a net-zero emissions future.1 This is particularly relevant for industrial sectors such as cement and steel manufacturing, where CO2 emissions are generated by design (e.g., calcination) and cannot be eliminated by electrification alone.2 In addition, hard-to-electrify sectors, including aviation and maritime transportation, are likely to rely on carbon-based fuels for the foreseeable future, which could be synthesized from CO2, closing the carbon loop.Non-thermal plasma (NTP) has emerged as a promising method for low-temperature CO2 conversion into value-added products, offering an electrified route to carbon circularity. In NTP conversion, a high-voltage electric field accelerates free electrons, which can catalyze bond dissociation through electronic excitation, molecular collisions, ionization, or vibrational activation.3 These systems are classified as “non-thermal” due to the large disparity between electron temperature and bulk gas temperature, enabling highly endothermic reactions such as CO2 and H2O dissociation to occur near ambient conditions with minimal bulk gas heating.
One promising application of NTP is syngas production from H2O and CO2. NTP-driven syngas generation can eliminate the need for solid catalysts, reducing system complexity and cost while avoiding issues such as catalyst deactivation, poisoning, or regeneration. Notably, syngas (H2:CO:CO2) can serve as a versatile feedstock for Fischer–Tropsch synthesis of many high-value fuels and chemicals.4 As such, NTP-driven syngas production could potentially revolutionize industrial chemical manufacturing.
The syngas ratio (i.e., H2/CO molar ratio) can be a crucial parameter for efficient downstream chemical synthesis.5 Net CO formation via NTP typically proceeds through electron impact dissociation of CO2, in a complex reaction network that can be cyclic.4 Many industrial CO2 streams (e.g., flue gas, cement plant off-gas) also contain water vapor that can not only supply H+ but influence plasma chemistry by introducing additional reaction pathways that produce H2. Notably, net H2 formation occurs by electron impact dissociations of H2O that create OH and H−, the latter of which collides with H2O to produce OH− and H2.4 Altogether, electron impact dissociations are the primary drivers of syngas formation in NTP. However, many other reaction pathways may also occur, leading to a wide variety of intermediates.4 In addition, subsequent gas-phase (or surface reactions) can recombine plasma intermediates or unstable gas products like H2O2 and HO2 back into more stable products such as H2, CO2, or H2O, forming complex reaction clusters.4
Overall, the use of NTP to convert humidified CO2 gas into value-added products has only been studied in a handful of studies using dielectric barrier discharge (DBD) reactors.4,6–11 Accordingly, the underlying mechanisms of combined H2O and CO2 conversion are not well understood.4 A review conducted by Snoeckx et al. finds that CO2 conversion increases with energy input while H2:CO decreases with energy input.4 The study also finds that CO2 conversion decreases in the presence of H2O vapor, possibly as a result of destabilization of the plasma discharge4 or the quenching of vibrational levels of CO2.4,12,13 For instance, converting CO2 with a water content of 2.05% into syngas at a specific energy input (SEI) of 32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 J L−1, yielded an efficiency of only 7.2%.4 Still, energy efficiencies remain low (<25%) even when using pure CO2 gas feeds due to a lack of optimization.14 Besides energy efficiencies, controlling the H2/CO ratio of the syngas is critical for real-world applications, but only a limited range has been demonstrated. Two important operational parameters for achieving both high syngas energy efficiencies and tunable H2/CO ratios could be SEI and gas flow rate. However, no previous studies have systematically studied the impact of these parameters on CO2 and H2O conversion, presenting a critical gap in research.
000 J L−1, yielded an efficiency of only 7.2%.4 Still, energy efficiencies remain low (<25%) even when using pure CO2 gas feeds due to a lack of optimization.14 Besides energy efficiencies, controlling the H2/CO ratio of the syngas is critical for real-world applications, but only a limited range has been demonstrated. Two important operational parameters for achieving both high syngas energy efficiencies and tunable H2/CO ratios could be SEI and gas flow rate. However, no previous studies have systematically studied the impact of these parameters on CO2 and H2O conversion, presenting a critical gap in research.
To our knowledge, this study is the first to methodically explore the relationship between SEI, energy efficiency, and H2/CO production across a range of fixed gas flow rates in a DBD reactor for syngas production. A key contribution of this work lies in isolating the influence of SEI on syngas composition under otherwise constant operating conditions. In this study, we aimed to demonstrate the feasibility of producing tunable syngas ratios (H2:CO) using a simple, low-cost reactor architecture. From an application standpoint, the ability to control syngas composition by adjusting voltage and flow rate offers a pathway to demand-driven syngas generation from realistic humidified CO2 waste streams. Accordingly, this study holds significant translational potential to improve the efficiency and tunability of humidified CO2 conversion into value-added products, presenting a promising pathway to decarbonizing the chemical and fuel industries.
2. Experimental methods
2.1. Dielectric barrier discharge reactor setup
A DBD reactor was built, which consisted of a sealed Pyrex tube (outer diameter: 32.4 ± 0.2 mm; inner diameter: 25.5 ± 0.2 mm), functioning as the dielectric barrier, wrapped with a nickel foam electrode (80–120 PPI, 1 mm thick, 114.5 ± 0.2 mm long) connected to ground. Nickel foam was selected for its malleability and ability to maintain a stable electrode morphology after cleaning. Stainless steel mesh, tested initially, was impacted by cleaning and produced non-uniform electric fields that promoted destructive macro-filament formation, compromising discharge stability and reproducibility. A copper rod (22.0 ± 0.2 mm diameter) served as the inner high-voltage electrode, yielding a reaction volume of 32.1 ± 0.3 mL. The inner electrode was connected to a plasma generator that applied a high-frequency oscillating voltage to sustain the plasma discharge (Fig. S1 and Section S1.1).During each experiment, humidified CO2 was introduced via a mass flow controller into the DBD reactor at flow rates of 1.00, 0.20, or 0.04 L min−1. High-purity CO2 (bone-dry, >99.9% v/v, Airgas Suppliers) was passed through a humidification vessel (Dioxide Materials) using a mass flow controller (Bronkhorst EL-Flow Select) before entering the reactor. The gas flow rate was continuously monitored upstream of the reactor, with negligible variation over each experiment. Before experimentation, the humidity of the gas was quantified across different flow conditions using a humidity and temperature sensor (DwyerOmega RH-USB series). A detailed description of the humidified gas preparation and measurement procedures is provided in the SI (Section S1.2).
2.2. Operation and gas sampling procedure
The plasma discharge was driven by a high-voltage pulsed power supply operating at 500 Hz with a 50% duty cycle, while the pulse oscillation frequency was maintained at 36.5 ± 0.5 kHz. The specific energy input was controlled by adjusting the applied voltage and calculated post-experiment from oscilloscope measurements. The entire setup was enclosed within a custom-built Faraday cage to minimize electromagnetic interference with nearby instrumentation. All experiments began with the reactor equilibrated to ambient room temperature (22 °C). During operation, the humidified CO2 traversed the plasma region and exited from the opposite side of the reactor, where it was collected in a gas sampling bag. Humidified CO2 was flowed for ≥5 minutes to flush the system and stabilize humidity and gas conditions. Prior to plasma ignition, all equipment, including a gas chromatograph (GC), gas bag, thermal imager, and oscilloscope, was prepared for immediate data collection. The plasma generator was initiated at 50 V, and the frequency knob was tuned to maximize current. Voltage was then adjusted to the target value. Once the voltage stabilized, the timer was reset, and data collection proceeded according to a standardized timeline. After five minutes, a thermal image was captured; gas was collected for two minutes, and the oscilloscope recorded current and voltage traces over ≥10 pulses (0.02 s). Resistance and capacitance measurements followed. After seven minutes, final thermal images were taken, and the gas bag was sealed. A 150 ± 1 µL gas sample was then extracted from the gas bag using a Hamilton syringe and injected into an Agilent 8890 GC with a Shincarbon ST column and thermal conductivity detector. Additional details related to the reactor operation and sampling procedure are provided in Section S1.1–1.3.2.3. Gas chromatography
Gas chromatographer (GC) analyses targeted H2, CO, CO2, N2, O2, and CH4. A custom method employed a 20 mL min−1 flow rate and a temperature ramp from 200 °C to 135 °C (75 °C min−1) to resolve closely eluting N2/O2 peaks. Additional details related to gas analysis are provided in Section S1.3. Calibration was performed using certified gas standards for all species. Species concentrations (mol%) were converted to molar flow rates using the ideal gas law. These values were used to compute CO2 and H2O conversion, H2/CO product ratios, and energy efficiency. Propagated errors included GC calibration uncertainty, syringe sampling error, and fit uncertainty.2.4. Specific energy input and energy efficiency calculations
Electrical data were collected using a digital oscilloscope at 1 GS s−1 over 10 plasma pulses. Current and voltage signals were phase-aligned using Python, and instantaneous power was computed pointwise (P = IV). To manage data size, 1 in every 1000 points was sampled. Error was estimated by calculating the variance obtained from voltage and current arrays. Additional details regarding the energy data collection are provided in Section S1.4.During each experiment, dissipated power was computed as follows:
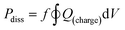 | (1) |
Using the dissipated power values, SEI values were calculated by dividing the dissipated power by the flow rate using eqn (2), where Qflowrate represents the flow rate in liters per second.
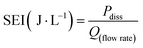 | (2) |
Energy efficiency values were then calculated using eqn (3), where m represents the molar flow rates (moles per second), ΔH represents the enthalpy of molecules (eV mol−1), and Psupplied is the external power input (eV s−1). The detailed procedure for calculating the molar flow rates, enthalpies, and external power is provided in Section S1.5.
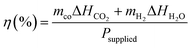 | (3) |
2.5. Targeted reaction pathways
The conversion of CO2 and H2O in non-thermal plasmas is thought to be initiated by electron-impact excitation and dissociation processes,14 wherein energetic electrons selectively activate molecular vibrations or directly cleave chemical bonds. These interactions generate radicals and excited species15 (e.g., OH, O, H, CO2(V)) that can recombine under non-equilibrium conditions to form stable products such as CO and H2. As a result, the electron energy distribution function (EEDF) plays a central role in determining dominant reaction pathways and product selectivity.In CO2 conversion, low average electron energies preferentially promote vibrational excitation, enabling vibrational ladder climbing and eventual dissociation into CO and atomic O. At higher electron energies, direct electron-impact dissociation becomes accessible but can also enhance back-reactions that limit net CO formation. In contrast, H2O conversion proceeds primarily through direct electron-impact dissociation, producing hydrogen radicals that readily recombine to form H2. Meanwhile, OH radicals produced from water dissociation can participate in secondary pathways or recombine to form water. The reaction pathways for H2O and CO2 conversion are detailed in the SI (Section S1.6). When CO2 and H2O are processed simultaneously, their chemistries become coupled through a shared radical pool. However, the extent to which SEI and flow rate jointly control CO and H2 selectivity and energy efficiency in these systems remains underexplored, motivating the present study.
3. Results and discussion
3.1. Maximum energy conversion efficiencies yield the lowest syngas ratio
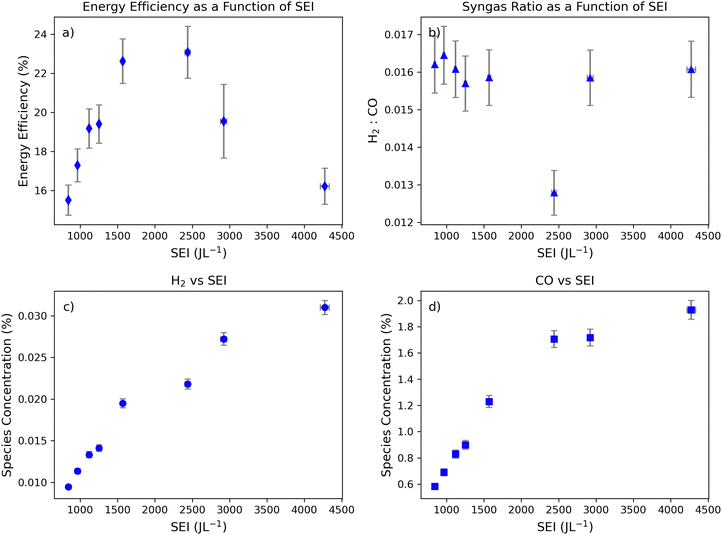
The effect of SEI on syngas conversion was initially investigated in a high CO2 flow regime of 1.0 L min−1. In this flow regime, energy efficiencies increased to a maximum of ∼23% at an SEI of 2440 J L−1 before gradually decreasing at higher SEI values (Fig. 1a). Consistent with expected energy utilization behavior, energy efficiencies exhibited a non-monotonic trend with SEI, peaking at intermediate values, and declining at both low and high SEI (Fig. 1a). Notably, the highest energy efficiency in this flow regime also generated the lowest syngas ratio due to high CO production (Fig. 1b). Overall, the relationship between syngas ratio and energy input did not demonstrate a declining linear trend as described in related literature.4At the SEI that achieved the peak energy efficiency (2440 J L−1), the CO concentration increased sharply (∼+40% increase), whereas the H2 concentration increased only marginally relative to the preceding condition (Fig. 1c and d). This divergence likely reflects a transition in the EEDF that increased the population of electrons energetic enough to dissociate CO2, thereby enhancing CO formation and atomic oxygen production in the plasma. Although the population of electrons capable of dissociating H2O into H2 would have also increased at this electron temperature, the observed increase in H2 concentration was smaller than that for CO (Fig. 1c). This was likely because the excess atomic oxygen produced from CO2 dissociation favored H2 recombination pathways that reform H2O, thereby suppressing net H2 production. In contrast, CO was likely more stable due to its higher bond dissociation energy, allowing a larger fraction of CO molecules to persist as the gas exits the plasma zone. Consequently, the effluent gas exhibited an elevated CO concentration with only a modest increase in H2, producing a local minimum in the syngas ratio. These phenomena are further discussed using simulation data in Section 3.6.
As SEI increases further beyond the point of the lowest syngas ratio, CO formation approaches a plateau (Fig. 1b and d). Beyond this point, additional increases in SEI no longer preferentially enhance CO formation, and H2-producing pathways regain relative importance, leading to a subsequent increase in the syngas ratio (Fig. 1b–d). These observations may reflect the onset of CO dissociation at high SEI, reducing net CO production.
Interestingly, this saturation effect indicates that plasma ionization becomes less efficient beyond a critical SEI, with excess energy contributing to further bond dissociation. This interpretation is supported by the decline in energy efficiency observed beyond moderate SEI in this flow regime (Fig. 1a). However, the SEI range necessary to confirm the full extent of this plateau would require more complex testing equipment outside the scope of this study.
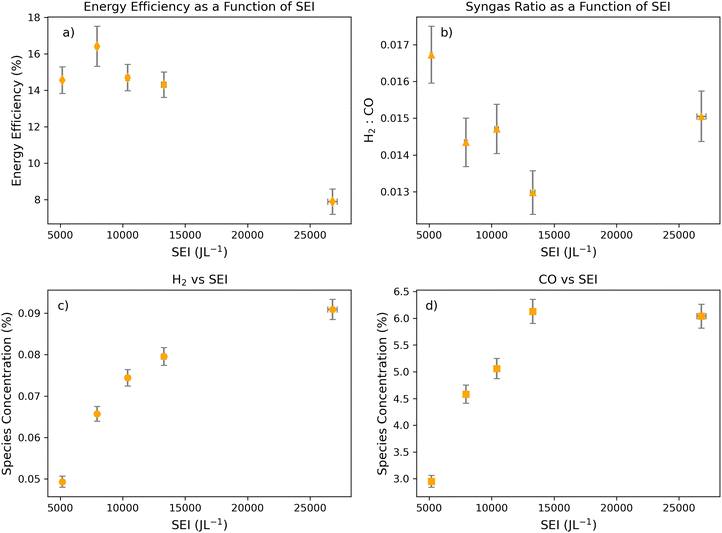
3.2. CO formation plateaus after the lowest syngas ratios are achieved
To further examine the effect of SEI on syngas formation, lower gas flow rates of 0.2 L min−1 were applied to the DBD module (Fig. 2). The general trends in this moderate flow regime mirror those in the high-flow regime. Namely, the lowest syngas ratio (H2/CO) occurs near the peak energy efficiency of the system (Fig. 1a and b). In addition, CO and H2 production plateau at high SEI levels (Fig. 1c and d). As expected, the lowest syngas ratio (0.122 ± 0.005) occurs around 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 J L−1, before the maximum SEI value in this regime.
000 J L−1, before the maximum SEI value in this regime.
The trends in SEI versus energy efficiency and H2/CO ratio share some commonalities but differ in important ways (Fig. 2a and b). In general, both energy efficiency and the H2/CO ratio decrease with increasing SEI. At high SEI, a larger fraction of the supplied energy was likely dissipated as heat or consumed in CO2 or H2O recombination reactions, resulting in reduced energy efficiency (Fig. 2a). These observations are reflected in the relationship between SEI and syngas composition. At moderate SEI, elevated average electron temperatures promoted CO2 dissociation to CO, resulting in a lower H2/CO ratio (Fig. 2b). At the highest SEI (∼27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 J L−1), however, enhanced H2O conversion to H2 yields moderate H2/CO ratios and reduced energy efficiencies despite CO formation having reached a plateau (Fig. 2b–d). These trends are consistent with those observed at higher flow rates and may reflect CO decomposition or a maximum in atomic oxygen generated during CO2 dissociation, which favors H2 reformation to H2O.
500 J L−1), however, enhanced H2O conversion to H2 yields moderate H2/CO ratios and reduced energy efficiencies despite CO formation having reached a plateau (Fig. 2b–d). These trends are consistent with those observed at higher flow rates and may reflect CO decomposition or a maximum in atomic oxygen generated during CO2 dissociation, which favors H2 reformation to H2O.
Interestingly, the lowest SEI (∼5000 J L−1) also resulted in relatively low energy efficiency (∼14%), likely because electron temperatures were insufficient to effectively dissociate either H2O or CO2 during molecule-electron collisions. This interpretation is consistent with the low concentrations of both H2 and CO measured under these conditions (Fig. 2c and d). However, as a higher fraction of electrons was likely capable of dissociating water, a high H2/CO ratio was observed.
Beyond the lowest SEI, CO formation increases more rapidly than H2 formation over the SEI range of ∼5000–15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 J L−1. Although H2O is more readily dissociated than CO2, its much lower concentration limits the relative increase in H2 production. As a result, CO formation dominates, leading to a decrease in the H2/CO ratio with increasing SEI. At an SEI of ∼7900 J L−1, energy efficiency reaches a maximum, coinciding with enhanced CO formation and a concurrent decline in the syngas ratio.
000 J L−1. Although H2O is more readily dissociated than CO2, its much lower concentration limits the relative increase in H2 production. As a result, CO formation dominates, leading to a decrease in the H2/CO ratio with increasing SEI. At an SEI of ∼7900 J L−1, energy efficiency reaches a maximum, coinciding with enhanced CO formation and a concurrent decline in the syngas ratio.
Proceeding to higher SEIs, CO and H2 outputs increase only marginally despite substantial additional energy input (Fig. 2c and d), while overall energy efficiency declines, indicating that a growing fraction of input energy is diverted away from productive H2/CO synthesis pathways. One plausible mechanism for this, beyond product dissociation, is that increased vibrational excitation (e.g., OH stretch modes in H2O) absorbs input energy without facilitating bond scission or productive recombination. However, advanced in situ or operando characterization techniques would be needed to verify such effects.
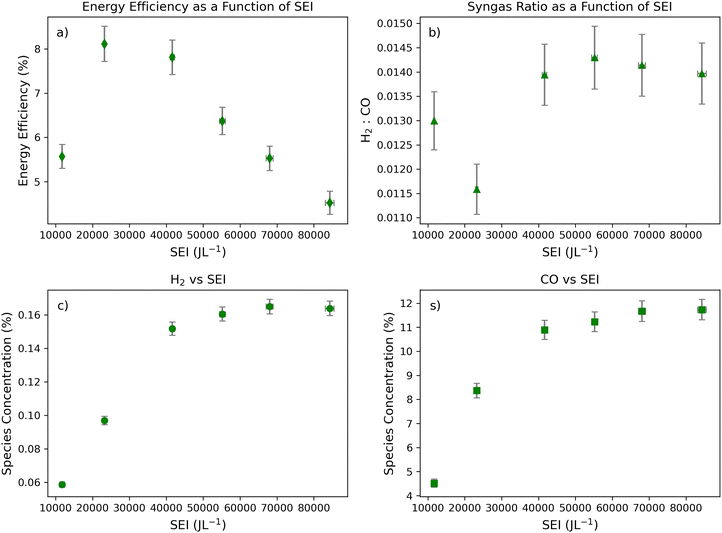
Accompanying tests at high and moderate gas flow rates, the effect of SEI on syngas formation was examined at ultra-low gas flow rates of 0.04 L min−1 (Fig. 3). In general, this gas flow regime generated some of the clearest relationships between SEI, energy efficiencies, and product formation. This behavior may arise because the low flow rate enables the highest SEI to be reached, resulting in elevated electron temperatures and extended residence times that increase the likelihood of electron–molecule collisions.
As in previous flow regimes, the minimum syngas ratio was observed well below the highest SEI. In addition, CO and H2 concentrations plateau at elevated SEI levels. Notably, an SEI of 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 ± 300 J L−1 yields the lowest syngas ratio and occurs prior to the plateau in H2 production, consistent with trends observed across other flow regimes. At this SEI, CO formation likely increases due to a rapid rise in the population of electrons capable of dissociating CO2 (Fig. 3d). This enhanced CO2 dissociation would result in a comparatively smaller increase in H2 production due to the lower H2O concentration in the feedstock, consistent with the observed syngas ratios (Fig. 3b and c).
300 ± 300 J L−1 yields the lowest syngas ratio and occurs prior to the plateau in H2 production, consistent with trends observed across other flow regimes. At this SEI, CO formation likely increases due to a rapid rise in the population of electrons capable of dissociating CO2 (Fig. 3d). This enhanced CO2 dissociation would result in a comparatively smaller increase in H2 production due to the lower H2O concentration in the feedstock, consistent with the observed syngas ratios (Fig. 3b and c).
3.3. Flow rate and specific energy input impact energy efficiency
Comparing the results from different gas flow regimes shows that energy efficiency generally decreased as flow rates decreased (Fig. 4). However, higher flow rates generally yielded superior energy efficiencies at equivalent SEI values. These results highlight that SEI alone cannot reliably predict energy efficiency.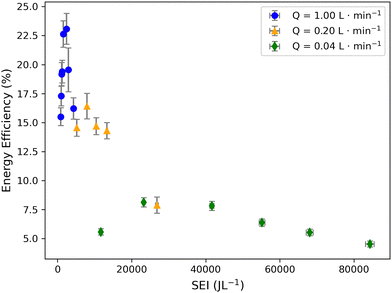 | ||
| Fig. 4 Comparison of energy efficiencies achieved for H2/CO under different flow regimes. Energy Efficiency for H2/CO production as a function of SEI and flow rate (markers). | ||
Interestingly, higher energy efficiencies were consistently observed at higher flow rates. For example, the maximum energy efficiency at 1.0 L min−1 (∼23%) was approximately 30% higher than that achieved at 0.2 L min−1 (∼16%), while the maximum efficiency at 0.2 L min−1 was ∼66% higher than that observed at 0.04 L min−1 (Fig. 4). We attribute this trend to residence-time effects. At lower flow rates, residence times increase substantially (up to ∼48 s at 0.04 L min−1), allowing gas-phase species to undergo many more plasma–molecule and electron–molecule collisions. Although this extended exposure enhances overall CO2 conversion and product formation, it also increases the likelihood of non-productive secondary processes. In particular, species that have already formed (e.g., H2, CO) may undergo repeated dissociation, leading to wasted energy and reduced energy efficiency. As a result, low flow rates exhibit higher SEI and conversion but lower energy efficiency, whereas higher flow rates benefit from shorter residence times, fewer unnecessary collisions, and consequently higher energy efficiency. These phenomena likely led to the observed trend where energy efficiency generally decreases with increasing SEI across all flow regimes (Fig. 4). While additional flow rates (e.g., 0.5 or 1.0 L min−1) could have provided supplementary insights, other intermediate flow rates would have likely produced values closely clustered to the moderate flow rate tested. Furthermore, higher flow rates (>1.0 L min−1) were constrained by the humidity control limitations and gas sampling requirements in our protocol. Because identical SEI values achieved via reduced flow or increased voltage can yield different plasma behaviors, SEI must be interpreted in the context of flow rate, residence time, gas composition, and pulse conditions. Future work could work to further decouple these effects by comparing identical SEI values reached through different operational pathways.
3.4. Feedstock conversion and syngas ratio are influenced by flow rate and specific energy input
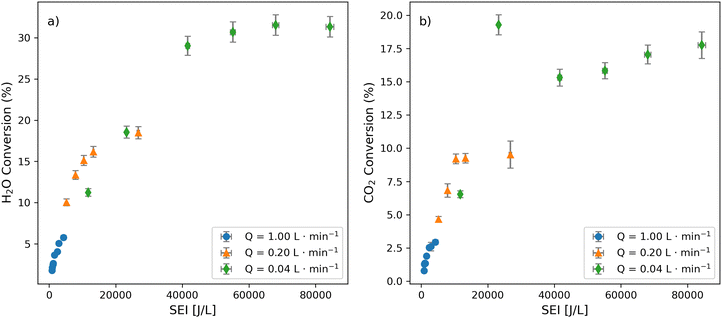
Building on the observation that SEI and flow rate impact energy efficiency, the role of these parameters on feedstock (i.e., H2O, CO2) conversion and syngas ratio was analyzed (Fig. 5 and 6). Notably, feedstock conversion exhibits a clear and consistent dependence on SEI across all flow rates. H2O conversion increases with SEI before reaching a plateau (Fig. 5a), consistent with a logistic increase in the population of electrons with enough energy to dissociate H2O. CO2 conversion shows a similarly monotonic increase with SEI across flow regimes, followed by saturation at high SEI (Fig. 5b). This behavior likely reflects the increasing fraction of electrons capable of dissociating CO2 as energy density rises, coupled with residence-time effects. Namely, high flow rates possibly yielded lower conversion due to shorter residence times and fewer productive molecule-electron collisions, whereas low flow rates seemed to promote higher conversion by extending plasma exposure (Fig. 5). A single deviation is observed under the lowest flow rate (0.04 L min−1) and an SEI of 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 ± 300 J L−1. This point coincides with the lowest syngas ratio and the highest carbon deposition (Fig. S5), suggesting the involvement of additional pathways such as CO dissociation or surface-mediated processes. Resolving this behavior will require advanced spectroscopic investigation outside the scope of the current study. Overall, this analysis of feedstock conversion reinforces that increasing SEI (via higher power and/or longer residence time) raises electron temperature and conversion until a plateau is reached, beyond which additional energy no longer yields proportional gains. This interpretation is supported by the simulations discussed in Section 3.6 and underpins the observed relationships between SEI and syngas ratio (Fig. 6).
300 ± 300 J L−1. This point coincides with the lowest syngas ratio and the highest carbon deposition (Fig. S5), suggesting the involvement of additional pathways such as CO dissociation or surface-mediated processes. Resolving this behavior will require advanced spectroscopic investigation outside the scope of the current study. Overall, this analysis of feedstock conversion reinforces that increasing SEI (via higher power and/or longer residence time) raises electron temperature and conversion until a plateau is reached, beyond which additional energy no longer yields proportional gains. This interpretation is supported by the simulations discussed in Section 3.6 and underpins the observed relationships between SEI and syngas ratio (Fig. 6).
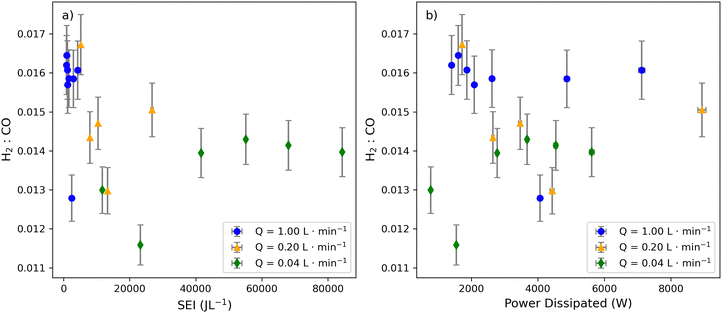 | ||
| Fig. 6 Comparison of syngas ratios across flow regimes. Syngas ratios as a function of SEI (A) syngas ratio as a function of dissipated power (B), with influent gas flow rates as markers. | ||
The results of this study demonstrate the ability to tune syngas ratios from around 0.011 to 0.016 by adjusting SEI and flow rate (Fig. 6a). In general, there is no clear difference between the syngas ratios achievable at different flow rates. While the lowest flow rate demonstrates the lowest syngas ratio of 0.011, this condition seemed to be an outlier. Altogether, this study draws different conclusions about the relationship between increased power applied and syngas ratios than the study conducted by Snoeck et al. 2017, which noted that the syngas ratio (H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO) decreases with energy input.4 This could be due to differences in the DBD reactor architecture between our study and past work. Notably, this study also observed that the minimum syngas ratio occurs well below the maximum applied power and plateau behavior of CO (Fig. 1–3). Similarly, all but these outlying minimum syngas experiments have overlapping error bars, meaning that there is not a clear linear relationship between the syngas ratio and energy input as might be expected based on the activation energies of CO2 and H2O.
CO) decreases with energy input.4 This could be due to differences in the DBD reactor architecture between our study and past work. Notably, this study also observed that the minimum syngas ratio occurs well below the maximum applied power and plateau behavior of CO (Fig. 1–3). Similarly, all but these outlying minimum syngas experiments have overlapping error bars, meaning that there is not a clear linear relationship between the syngas ratio and energy input as might be expected based on the activation energies of CO2 and H2O.
Notably, while many operating conditions can yield similar syngas ratios (H2/CO), the absolute amounts of H2 and CO produced in the plasma can differ substantially. Consequently, experiments with comparable syngas ratios may nonetheless exhibit distinct overall gas compositions. This behavior reflects the fact that the syngas ratio primarily captures reaction pathway selectivity rather than absolute product yields. Changes in flow rate mainly affect residence time and, therefore, the extent of reaction, allowing H2 and CO production to scale together under similar discharge conditions while maintaining a nearly constant ratio. As a result, comparable H2/CO ratios can be obtained at different flow rates despite large differences in absolute H2 and CO concentrations. This effect can be illustrated by plotting the syngas ratio as a function of H2 and CO concentration (SI Section and Fig. S6, S7), which demonstrates that similar syngas ratios can arise from markedly different product CO and H2 yields under distinct operating conditions.
Plotting syngas ratio with respect to the dissipated power does not support a negative trend as might be expected based on SEI trends (Fig. 5b). Instead, similar dissipated power applied under different flow rates can yield either comparable or distinct syngas ratios. One explanation is that similar electron temperatures may be achieved under different operating conditions, leading to comparable reaction selectivity. For example, when electron temperatures are sufficient to maximize H2O dissociation, both high- and low-flow regimes can generate similar hydrogen radical concentrations and thus comparable recombination behavior upon exiting the plasma. However, differences in residence time can truncate reaction pathways at higher flow rates, resulting in divergent product distributions even at similar applied power. Consistent with this interpretation, at comparable power the 1.00 L min−1 condition exhibits significantly higher syngas ratios than the 0.04 L min−1 condition, reflecting its ∼25-fold shorter residence time (Fig. 6b). These results indicate that syngas formation is governed not only by electron temperature but also by residence time. Together, they establish a more nuanced relationship between syngas ratio, residence time, and energy input, providing a refined framework for interpreting plasma-driven syngas formation and guiding future studies.
3.5. Simplistic computational modeling and reasoning
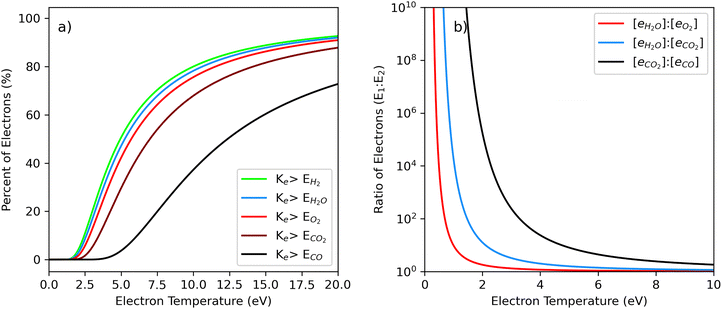
To better understand the underlying reactions of the DBD plasma system, modeling of simplistic electron energy distribution functions was performed and compared to the relevant bond dissociation energies of molecules in the humidified CO2 feed gas (Fig. 7 and Table 1). A Druyvesteyn distribution was used to depict the plasma, which has more electrons with energies near the average kinetic energy and a less voluminous high-energy tail than a Maxwell–Boltzmann distribution.3 As such, the Druyvesteyn distribution has been found to more accurately describe plasmas that are not in local thermodynamic equilibrium.3,15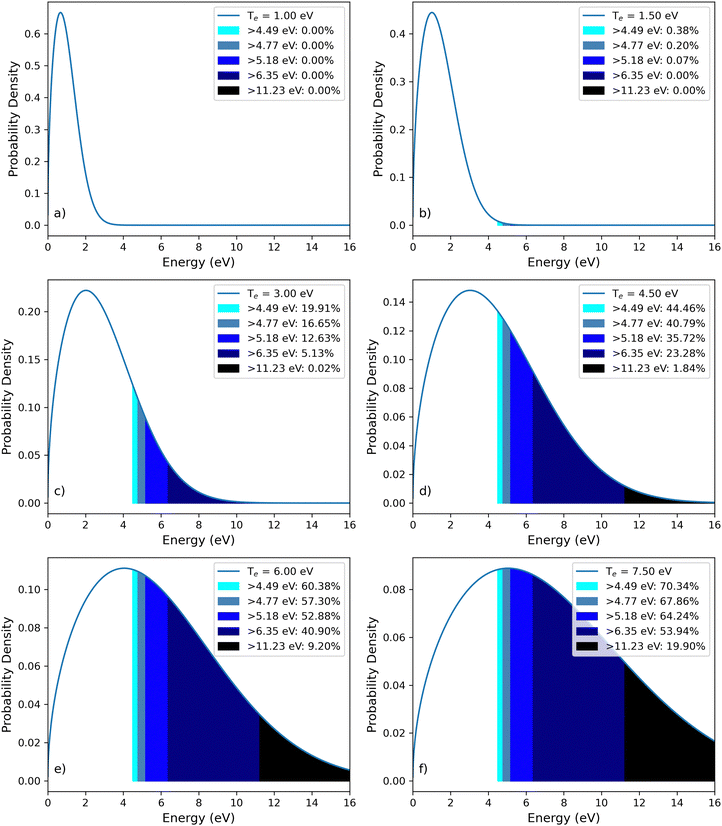 | ||
Fig. 7 Druyvesteyn electron energy distributions during syngas formation. Predicted electron density or fraction (y-axis) versus electron energy (eV). Electron energy and percentage distribution for each bond activation zone are shown in each legend. The lightest blue shade in the legend represents the percentage of electrons with kinetic energy above the bond energy of H2. The next-lightest shade of blue represents electrons with kinetic energy greater than O–H bonds in OH and H2O. The dark blue area represents electrons with kinetic energy greater than the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond strength of CO2. The purple shade represents electrons with kinetic energy greater than the C O bond strength of CO2. The purple shade represents electrons with kinetic energy greater than the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) O bond strength of CO. The activation energies16 of H2 (4.49 eV), H2O (4.77 eV), OH (4.77 eV), O2 (5.18 eV), CO2 (6.35 eV), and CO (11.23 eV) define each shaded region. The effective electron temperature increases in each plot from 1.00 eV in Panel a to 7.5 eV in Panel f. O bond strength of CO. The activation energies16 of H2 (4.49 eV), H2O (4.77 eV), OH (4.77 eV), O2 (5.18 eV), CO2 (6.35 eV), and CO (11.23 eV) define each shaded region. The effective electron temperature increases in each plot from 1.00 eV in Panel a to 7.5 eV in Panel f. | ||
| Bond | Energy (eV) | Parent molecule | Products |
|---|---|---|---|
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
6.35 | CO2 | CO, O |
| O–H | 4.77 | H2O4 | OH, H |
| O–H | 4.77 | OH | O, H |
| H–H | 4.49 | H2 | H, H |
C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) O O |
11.23 | CO | C, O |
O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
5.18 | O2 | O, O |
As expected, the EEDF model predicts that greater electron temperatures yield higher energy electrons (Fig. 6). When electrons with energy greater than a bond energy collide with a heavy molecule like CO2, there is a chance that the collision causes the molecule to dissociate into quasi-stable products (e.g., CO, O). When the electron temperature is sufficiently high, almost all electrons are well above the dissociation energy of a molecule like H2O, leading to a high probability of H2O dissociation alongside CO2 conversion. Conversely, at low electron temperatures where very few electrons have enough energy to dissociate a molecule like CO2, as a consequence, fewer CO2 dissociation reactions are expected. Accordingly, EEDF models combined with an understanding of molecular structure can help interpret many of the results from this study.
A common trend across all gas flow rate regimes was an increase in H2 and CO formation up until a plateau at high SEIs (Fig. 1–3). Based on bond dissociation energies of the feedstock molecules, it would be expected that the first reaction to occur at low electron temperatures would be H2O dissociation to H2 (4.77 eV, Table 1). As electron temperatures increase, it would then be expected that H2 decomposition (5.18 eV) could occur, followed by CO2 decomposition into CO (6.35 eV), and finally CO decomposition (11.23 eV). Accordingly, it is expected that increasing the average electron temperature by increasing SEI would enhance H2 and CO formation up until the decomposition reactions of these molecules take place, leading to a semi-equilibrium and plateau of their concentrations in the effluent plasma. This logic aligns the trends observed across all the flow regimes tested (Fig. 1–3) and is consistent with the EEDF simulation results (Fig. 7 and 8). For example, at a low electron temperature (Te = 3.00 eV), approximately 16% of electrons can drive H2 formation, ∼12% can dissociate H2, ∼5% can convert CO2 to CO, and virtually none can dissociate CO. In contrast, at a higher electron temperature (Te = 7.50 eV), these fractions increase to ∼67%, ∼64%, ∼53%, and ∼20%, respectively (Fig. 7). Thus, increasing electron temperature beyond a critical threshold is expected to inhibit H2 and CO formation, as observed in this study with increasing SEI (Fig. 1–3).
Overall, these mechanisms likely contributed to the relationship between SEI and energy efficiency across all flow regimes analyzed in this study. For each flow rate, energy efficiency initially increases with SEI, reaches a maximum, and then decreases at higher SEI values (Fig. 4). However, H2O and CO2 conversion were found to rise with increasing SEI before reaching a plateau at elevated SEI (Fig. 5). These observations suggest that after a significant population of electrons becomes capable of dissociating H2 and CO, increasing SEI promotes further H2O and CO2 conversion while decreasing energy efficiency due to increased electron-impact destruction of the products. This rationale tracks with trends observed in the EEDF simulation data. As an example, at an electron temperature of 4.5 eV, ∼35% of electrons have sufficient energy to dissociate H2, and ∼2% can dissociate CO. However, increasing the electron temperature to 6.0 eV nearly doubles the fraction capable of dissociating H2 and increases the fraction capable of dissociating CO by a factor of five (Fig. 8a). Accordingly, at high SEI, we would expect reduced net H2 and CO yields even if H2O and CO2 dissociation increase.
Product decomposition informed by EEDF also helps explain the observed dependence of the syngas ratio on SEI. Across all flow regimes tested, the minimum syngas ratio was found to occur at intermediate SEI, well below the maximum SEI (Fig. 1–4). Notably, the minimum syngas ratio is observed before H2 production plateaus, and well before the maximum syngas ratio in each regime. This minimum often coincides with peak energy efficiency, reflecting preferential enhancement of CO formation relative to H2. These phenomena are predicted by EEDFs as a result of an increase in CO2 dissociation reactions that occurs at a slightly higher electron temperature than the dissociation of H2O. This results in O radicals, which can consume H atoms. When an H-containing molecule combines with an O-containing molecule, the stoichiometric balance is shifted towards less CO2, leading to more CO and less H2. Based on previous studies, it is expected that minimum syngas ratios would occur at SEI values before a plateau in CO formation. This notion is reinforced by comparing the number of electrons capable of dissociating one bond relative to another at different electron temperatures (Table 1 and Fig. 7).
Accordingly, combining experimental and modeling data can predict ideal operating conditions for target syngas ratios. For example, EEDF simulations indicate there is an electron temperature window where the ratio of electrons with energy levels greater than the bond energy of H2O far exceeds those with energy levels greater than the bond energy of CO2 (Fig. 8b). Therefore, targeting this electron temperature is predicted to produce high syngas ratios under low SEI conditions. These conditions could be ideal for some real-world operations and are supported by the results of this study across operating flow rates (Fig. 1–4). Conversely, these models can also predict electron temperatures where both H2O and CO2 dissociation are high, leading to a large number of free O radicals that capture H to form OH and H2O. This mechanism could limit the reformation of H2 and lead to a minimum syngas ratio, which could be ideal in some industrial scenarios. As such, beyond helping to interpret the results of this study, these computational models can help guide future research and plasma applications.
4. Conclusions
This study investigated syngas production under different flow regimes using a coaxial DBD reactor and humified CO2 gas. This work aimed to clarify the relationship between syngas ratios and SEI under different flow rates to provide a foothold for future studies to expand upon. Notably, experiments showed that contrary to other works,4 there was no clear decrease in produced syngas ratios with increasing energy input. Instead, minimum syngas ratios in each flow regime were produced before the maximum applied SEI.To better understand the mechanisms behind these results, this study employed a simplistic computational model for predicting and understanding syngas production achieved under different SEIs and gas flows. The EEDF model supports that conditions can be found that heavily favor the dissociation of some molecules and not others, leading to fluctuations in produced syngas ratios. Furthermore, combining experiment and computational results reveals a more complicated relationship between syngas ratio and SEI than previously described.4 While other studies have explored the relationship between SEI and product gases in a DBD reactor,18 this study is among the first to use SEI as the independent variable for syngas production and energy efficiency in a silent DBD reactor for the reduction of CO2 and H2O.
Overall, this study provides a roadmap of syngas ratios and associated energy efficiencies under different operational conditions. We expect that this data can be used in future studies to help tune syngas ratios to match market-specific needs4 and downstream applications such as Fischer–Tropsch synthesis of sustainable fuels. Although it was not the core focus of this study, this work also achieved an impressive maximum energy efficiency of 23 ± 1%. Building on this study, future studies could work toward highly efficient reactors capable of creating desired syngas blends. As such, this work sets the stage for NTP to be used in multiple sectors of the energy economy to achieve net-zero emissions through efficient CO2 valorization.
Author contributions
Conceptualization: JJ. Formal analysis: MK. Funding acquisition: JJ. Investigation: MK and JJ. Methodology: MK and JJ. Project administration: JJ. Resources: JJ. Supervision: JJ. Visualization: MK. Writing – original draft: MK and JJ. Writing – review & editing: MK and JJ.Conflicts of interest
There are no conflicts to declare.Data availability
The datasets supporting this article have been included as part of the supplementary information (SI). The SI includes additional details on the reactor setup as well as supplemental calculations and analyses. Further information that is not provided can be obtained from the authors upon request. Supplementary information is available. See DOI: https://doi.org/10.1039/d5ya00293a.Acknowledgements
JJ and MK acknowledge financial support provided by the University of Michigan's College of Engineering and Michigan Institute for Plasma Science and Engineering.References
- Intergovernmental Panel on Climate Change, Climate Change 2022: Mitigation of Climate Change, https://www.ipcc.ch/report/ar6/wg3/chapter/technical-summary/, ( 2022).
- S. Barbhuiya, F. Kanavaris, B. B. Das and M. Idrees, Decarbonising cement and concrete production: Strategies, challenges and pathways for sustainable development, J. Build. Eng., 2024, 86, 108861 Search PubMed
.
-
A. Grill, Fundamentals of Plasma, Cold Plasma Materials Fabrication: From Fundamentals to Applications, IEEE, 1994, pp. 1–23 DOI:10.1109/9780470544273.ch1
.
- R. Snoeckx, A. Ozkan, F. Reniers and A. Bogaerts, The Quest for Value-Added Products from Carbon Dioxide and Water in a Dielectric Barrier Discharge: A Chemical Kinetics Study, ChemSusChem, 2017, 10, 409–424 Search PubMed
.
- P. L. Spath and D. C. Dayton, Preliminary Screening – Technical and Economic Assessment of Synthesis Gas to Fuels and Chemicals with Emphasis on the Potential for Biomass-Derived Syngas, ( 2003).
- Z. Cui,
et al., Plasma-Catalytic Methanol Synthesis from CO2 Hydrogenation over a Supported Cu Cluster Catalyst: Insights into the Reaction Mechanism, ACS Catal., 2022, 12, 1326–1337 CrossRef CAS
.
-
S. Futamura and H. Kabashima, Synthesis Gas Production from CO2 and H2O with Nonthermal Plasma, ed. Park, S.-E., Chang, J.-S. & Lee, K.-W., in Studies in Surface Science and Catalysis, Elsevier, vol. 153, 2004, pp. 119–124 Search PubMed
.
- Nonthermal Plasma-Assisted CO2-H2O Conversion over NiO and Co3O4 Supported on CeO2 - Bajpai - 2023 - Chemical Engineering & Technology - Wiley Online Library. https://onlinelibrary.wiley.com/doi/10.1002/ceat.202200481.
- X. Yao, Y. Zhang, Z. Wei, M. Chen and W. Shangguan, Plasma-Catalytic Conversion of CO2 and H2O into H2, CO, and Traces of CH4 over NiO/Cordierite Catalysts, Ind. Eng. Chem. Res., 2020, 59, 19133–19144 Search PubMed
.
- Anonymous. Plasma-Assisted Catalytic Reverse Water Gas Shift Reactor. Tech Briefs41, ( 2017).
- S. Mahammadunnisa, E. L. Reddy, D. Ray, C. Subrahmanyam and J. C. Whitehead, CO2 reduction to syngas and carbon nanofibres by plasma-assisted in situ decomposition of water, Int. J. Greenhouse Gas Control, 2013, 16, 361–363 Search PubMed
.
- A. Indarto, D. R. Yang, J.-W. Choi, H. Lee and H. K. Song, Gliding arc plasma processing of CO2 conversion, J. Hazard. Mater., 2007, 146, 309–315 CrossRef CAS PubMed
.
- G. Chen,
et al., Simultaneous dissociation of CO2 and H2O to syngas in a surface-wave microwave discharge, Int. J. Hydrogen Energy, 2015, 40, 3789–3796 CrossRef CAS
.
- R. Aerts, W. Somers and A. Bogaerts, Carbon dioxide splitting in a dielectric barrier discharge plasma: a combined experimental and computational study, ChemSusChem, 2015, 8, 702–716 Search PubMed
.
- X. Ma,
et al., Plasma Assisted Catalytic Conversion of CO2 and H2O Over Ni/Al2O3 in a DBD Reactor, Plasma Chem. Plasma Process., 2019, 39, 109–124 CrossRef CAS
.
- R. J. Carman and R. P. Mildren, Electron energy distribution functions for modelling the plasma kinetics in dielectric barrier discharges, J. Phys. Appl. Phys., 2000, 33, L99 CrossRef CAS
.
- P. Flower, K. Theopold, R. Langley and A. Christianson 9.4: Bond Strength and Energy. Chemistry LibreTextshttps://chem.libretexts.org/Courses/Bellarmine_University/BU%3A_Chem_103_(Christianson)/Phase_3%3A_Atoms_and_Molecules_-_the_Underlying_Reality/9%3A_Chemical_Bonding/9.4%3A_Bond_Strength_and_Energy ( 2018).
- S. Müller,
et al., Investigation of Partial Oxidation of Methane in a Cold Plasma Reactor with Detailed Product Analysis, Plasma Chem. Plasma Process., 2023, 43, 513–532 Search PubMed
.
| This journal is © The Royal Society of Chemistry 2026 |