 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Coal-assisted flotation for olive mill wastewater: remediation and resource recovery
H. Polat *a,
M. Polat
*a,
M. Polat b,
M. C. Eren
b,
M. C. Eren a,
Ç. Güngör
a,
Ç. Güngör a and
E. Özsaygı
a and
E. Özsaygı a
a
aDepartment of Chemistry, Izmir Institute of Technology, Urla, Izmir, Turkey. E-mail: hurriyetpolat@iyte.edu.tr
bDepartment of Chemical Engineering, Izmir Institute of Technology, Urla, Izmir, Turkey
First published on 1st May 2026
Abstract
Olive mill wastewater (OMWW) is difficult to manage due to its high organic load and persistent phenolic content. Here, coal-assisted flotation is evaluated as a single-step process integrating OMWW remediation with the upgrading of low-grade Soma lignite by exploiting interfacial interactions between wastewater constituents and coal surfaces. ATR-FTIR characterized coal and OMWW, while XRF, ash analysis, contact angle, and zeta potential measurements further assessed coal composition and surface behavior. Flotation tests conducted with 5–100% OMWW in the process water showed a strong dependence on wastewater fraction: the highest coal recovery together with the lowest-ash froth product was obtained at 5% OMWW, whereas increasing OMWW content beyond 10% progressively reduced recovery, consistent with partial surface hydrophilization under more concentrated wastewater matrices. The results support a dual mechanism in which OMWW organics enhance coal hydrophobicity to promote bubble–particle attachment. At the same time, phenolic compounds are removed from solution through adsorption onto coal surface sites, corroborated by post-flotation spectroscopic signatures. Overall, coal-assisted flotation defines an operational window where OMWW treatment and lignite beneficiation can be integrated, providing a scalable option for resource-oriented wastewater management.
Environmental significanceOlive mill wastewater (OMWW) is one of those streams that looks “treatable” on paper but stays stubborn in practice. The phenolic load and acidity complicate biological routes, and many high-performance options (membranes, advanced oxidation) can be difficult to justify economically—especially for seasonal, distributed producers. We approached the problem from a different angle: instead of designing yet another stand-alone treatment train, we asked whether OMWW's own organic constituents could be used as part of the separation chemistry in a process that also delivers a recoverable product. Therefore we demonstrate that coal-assisted flotation can couple phenolic removal with coal upgrading in the same unit operation. |
1. Introduction
Olive oil production has long been central to agro-industrial activity around the Mediterranean, valued not only economically but also as part of regional heritage. However, olive oil extraction generates olive mill wastewater (OMWW), a chemically complex effluent that is challenging to manage due to its high pollutant load. In three-phase extraction systems, the high demand for process water results in substantial OMWW volumes, and wastewater handling becomes a major operational and environmental constraint. OMWW is typically characterized by high chemical oxygen demand (COD), suspended solids, residual lipids, and a significant proportion of recalcitrant phenolic compounds.1 Phenolics are particularly problematic because they are toxic, oxidative, and resistant to biodegradation.1,2 In combination with organic acids, they also depress pH, which may accelerate soil deterioration and increase contamination risks for surface and groundwater resources.1,3A broad range of OMWW treatment approaches has been explored, including biological, chemical, and membrane-based processes. In practice, phenolic inhibition often limits biological performance, whereas chemical and membrane systems can be limited by high reagent demand, energy consumption, fouling, and secondary waste generation.4 These constraints have shifted attention to strategies that prioritize phenolic reduction as an initial target, aiming to improve reuse potential and enable environmentally compatible, economically realistic management routes.4,5 Recent studies have emphasized circular economic perspectives, where OMWW is not only treated but also valorized for soil improvement and resource recovery.6,7 Innovative regional analyses also highlight the urgency of scalable solutions in Mediterranean contexts.6,7 In parallel, green treatment processes such as coagulation–flocculation combined with sunlight photocatalysis have been optimized as low-impact alternatives.8
Flotation has recently attracted interest as a separation process for contaminated waters because it can remove dispersed organic and fine particulates with comparatively low chemical input.9 Comparable flotation strategies have been extended to coal gasification residues, where surface hydrophobic modification enabled effective decarbonization of fine slag and demonstrated the adaptability of flotation to complex waste matrices.33 It is defined as a separation process in which air bubbles attach to hydrophobic particles, causing them to rise to the surface of a liquid for removal. Since hydrophobic interfacial interactions govern it, it is particularly relevant for wastewaters containing oily and amphiphilic constituents. Coal offers a practical material platform in this context, serving as both a carrier particle and an adsorbent due to its partial hydrophobicity, high surface area, and surface functional groups that interact with aromatic phenolic species.10 Comparable adsorption phenomena have been reported for phenolic compounds on coal ash and activated carbons.11–13 Even so, phenolic removal from OMWW via coal-assisted flotation has not been examined in a sufficiently systematic manner, especially in studies that explicitly link treatment outcomes to coal surface chemistry and interfacial behavior.
2. Materials and methods
2.1 Materials
Coal samples utilized in this study were sourced from the Soma Lignite Basin (Turkey) and were pre-treated by crushing and grinding before their use. The average (d50) size of ground coal was determined as −300 µm using a light scattering particle size analyser, Malvern 2000.Flotation experiments were carried out using a laboratory-scale flotation cell. In the process, dodecane (C12H26) was used as a collector to enhance the floatability of coal and therefore promote the attachment of coal particles to air bubbles.14,15 Methylisobutyl carbinol (MIBC) was selected as the frothing agent to provide stable froth formation and control bubble size distribution.16,17 In addition, a nonionic polymeric surfactant, Pluronic L-64, was utilized as an emulsifier to stabilize dodecane droplets within the aqueous phase, thereby improving collector dispersion and interaction efficiency.18 Then, olive mill wastewater (OMWW) obtained from an olive oil process factory in Ayvalık/Turkey, was introduced into the flotation medium at varying proportions to simulate realistic wastewater conditions.
2.2 Methods
A schematic representation of the experimental workflow for coal-assisted flotation treatment of olive mill wastewater is given in Fig. 1. The details of the experiments conducted were discussed in the following paragraphs. | ||
| Fig. 1 Schematic representation of the experimental workflow for coal-assisted flotation treatment of olive mill wastewater. | ||
| Parameter | OMWW, % | ||||
|---|---|---|---|---|---|
| 5 | 10 | 20 | 40 | 100 | |
| Coal (g) | 25 | 25 | 25 | 25 | 25 |
| Dodecane (mL) | 10 | 10 | 10 | 10 | 10 |
| MIBC (µL) | 200 | 200 | 200 | 200 | 200 |
| Pluronic L-64 (mL) | 10 | 10 | 10 | 10 | 10 |
| Distilled water (mL) | 950 | 900 | 800 | 600 | 0 |
| OMWW (mL) | 50 | 100 | 200 | 400 | 1000 |
The froth phase was the coal-enriched product, while the remaining liquid was treated wastewater. The liquid fraction was filtered again to remove residual suspended solids. All fractions (froth, residue, and treated liquid) were characterized to identify dominant particle types and adsorption behaviour of wastewater constituents—particularly organics and phenolics—onto specific coal surface domains and adsorption sites.
X-ray fluorescence spectroscopy (XRF) was applied to determine the elemental composition of the coal and OMWW-related solid fractions, allowing both qualitative and quantitative evaluation of major and trace elements.21
The hydrophobicity of the coal surface was assessed via contact angle measurements using the sessile-drop method.16,22
Dynamic light scattering (DLS) was employed to determine particle-size distribution and zeta potential measurements to evaluate surface charge characteristics.19,23
Ash analysis was conducted to determine the inorganic content of the coal and to evaluate the effect of flotation on mineral matter distribution. Dried samples were placed in porcelain crucibles and combusted in a muffle furnace at 800 °C for 2 h to remove organic components. After cooling, the remaining residue was weighed, and the ash content was calculated gravimetrically.
The ash content of the raw coal was found to be 12.33%. Following flotation, the ash contents of the concentrate and tailing fractions were determined separately.
 | (1) |
Flotation performance was quantified using combustible recovery (CR, %), defined as the fraction of combustible (ash-free) matter in the feed that is recovered in the froth product. CR was calculated as:
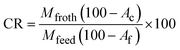 | (2) |
3. Results and discussion
3.1 Characterization of coal: structure and surface
The detailed surface, structural, and elemental characterization of the coal sample is presented and discussed to elucidate its role in the flotation-based removal of phenolic compounds.17,18 These offer valuable insights into both the quality of the coal and its adsorption performance in coal-assisted flotation processes.21,24 The details of each characterization method are discussed in the following subsections.The FTIR spectrum of freshly ground coal is shown in Fig. 3. One of the most prominent peaks observed was the –OH/–NH overlapping peak in the ∼3400 cm−1 region. An aliphatic C–H stretching peak is observed in the region around ∼2900 cm−1, indicating the representation of non-polar parts on the surface. A strong band spanning approximately 1700–1600 cm−1 is consistent with carbonyl-related vibrations (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), while the signal around ∼1600 cm−1 shows aromatic C
O), while the signal around ∼1600 cm−1 shows aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching, consistent with an aromatic framework. Various features located between 1300 and 1000 cm−1 can be attributed to C–O stretching modes of oxygen-containing groups, influencing surface polarity and adsorption. Finally, a weaker band in the 1000–800 cm−1 range shows Si–O–H vibrations and this implies a small mineral (silicate) contribution. In general, the spectrum points to a chemically diverse surface where the functional group distribution is expected to affect wettability during flotation and interaction with OMWW organic components, thus influencing adsorption and separation results.25,26
C stretching, consistent with an aromatic framework. Various features located between 1300 and 1000 cm−1 can be attributed to C–O stretching modes of oxygen-containing groups, influencing surface polarity and adsorption. Finally, a weaker band in the 1000–800 cm−1 range shows Si–O–H vibrations and this implies a small mineral (silicate) contribution. In general, the spectrum points to a chemically diverse surface where the functional group distribution is expected to affect wettability during flotation and interaction with OMWW organic components, thus influencing adsorption and separation results.25,26
Fig. 4 presents the particle size distribution and zeta potential characteristics of freshly ground coal. The top graph in the figure shows that the pulp contained a broad particle-size distribution, extending from approximately 0.2 µm to 400 µm, which can affect flotation performance.27 In the case of zeta potential measurements given in the bottom graph, there is a narrow charge distribution with a pronounced peak in the negative region. This is in accordance with the presence of functional groups that have been observed by FTIR. The functional groups of the coal surface are expected to be important, especially in adsorbing olive oil wastewater components. Then, the oil content of the wastewater should impart the coal surface partially hydrophobic, favoring effective particle–bubble attachments during flotation.
The degree of hydrophobicity (wettability) was evaluated through contact angle measurements, and the results are presented in Fig. 5. It is seen that the soma coal sample exhibits predominantly moderate contact angle values, with a significant proportion of measurements clustered around approximately 40°–60°. This behavior confirms the inherent partial hydrophobic nature of the lignite Soma coal, which is a key requirement for effective particle–bubble attachment in flotation processes.16,28 The presence of lower contact angle values, however, suggests the presence of oxygen-containing functional groups and mineral matter, contributing to surface heterogeneity. Overall, the surface characteristics of soma coal indicate that the surface supports its suitability as both an adsorbent and carriers floating particles in the coal-assisted flotation process designed in this study.
The elemental and oxide composition of the coal sample was obtained by X-ray fluorescence (XRF), and the results are given in Table 2. As expected for a low-rank coal, carbon represents the main component of the sample (25.96%), reflecting the organic/combustible matrix that dominates coal structure.29 The remaining signal is largely associated with mineral matter. In this regard, calcium, aluminum, iron, and magnesium appear as the principal inorganic elements, which is consistent with the mineral assemblages commonly reported for lignitic and sub-bituminous coals.30 Although present at lower levels, sulfur and phosphorus are also detected and are relevant from an environmental perspective because they may contribute to emissions and ash chemistry during thermal utilization. Additional minor contributions from sodium, potassium, and strontium further point to a compositionally heterogeneous mineral fraction.
| Soma coal (elemental) | Soma coal oxidation (oxide) | ||
|---|---|---|---|
| C | 25.9632 | CO2 | 95.1405 |
| Na | 0.0763 | Na2O | 0.1028 |
| Mg | 0.0858 | Mg2O | 0.1423 |
| Al | 0.2405 | Al2O3 | 0.4546 |
| Si | 0.3504 | SiO2 | 0.7495 |
| P | 0.3439 | P2O5 | 0.0115 |
| S | 0.0172 | SO3 | 0.8587 |
| Ca | 1.7065 | K2O | 0.0207 |
| Fe | 0.0905 | CaO | 2.3878 |
| Sr | 0.0019 | Fe2O3 | 0.1294 |
| Boric acid | 0.3521 | SrO | 0.0022 |
| Total | 29.2333 | Boric acid | 0.3521 |
| Total | 100.3521 | ||
The oxide breakdown supports the same picture. CO2 is the dominant oxide fraction, consistent with the carbon-rich nature of the sample and indicating the potential for CO2 evolution during combustion. The most abundant mineral-related oxides (CaO, Al2O3, Fe2O3, and MgO) represent ash-forming phases that can shape the inorganic residue. SO3 and P2O5, in turn, reflect sulfur- and phosphorus-bearing species in oxidized form. Overall, the XRF data confirm a carbon-dominated matrix accompanied by a diverse mineral-oxide fraction, which can contribute to surface heterogeneity and, consequently, influence adsorption behavior and flotation response.24
In addition to the physicochemical and surface characterization, the ash content of the coal was analysed to estimate the inorganic matter fraction. The ash percentage was calculated using eqn (1), based on the mass ratio of the residue remaining after ashing to the initial coal mass. To evaluate repeatability, the analysis was performed in triplicate, yielding ash contents of 11.0%, 11.5%, and 14.5%. Although the third value is somewhat higher, the results remain within a narrow range overall and indicate that the procedure provides acceptable reproducibility for the purpose of this study.
The ash content around 11.0–11.5% suggests a moderate mineral matter level in the sample. From a utilization perspective, such ash levels are typically considered manageable, whereas substantially higher ash contents can negatively affect combustion efficiency and operational performance.30 In the context of flotation, the presence of a moderate inorganic fraction can also be relevant because mineral phases may contribute to surface heterogeneity and alter interfacial interactions.
Taken together, the surface, structural, and compositional results support the selection of Soma coal as a suitable material for the adsorption-integrated flotation concept evaluated here. ATR-FTIR indicates the coexistence of hydrophobic moieties and oxygen-containing functional groups on the coal surface, while contact angle measurements confirm an overall hydrophobic tendency. Zeta potential measurements further suggest that the particles can remain sufficiently dispersed in aqueous media, which is favorable for effective bubble–particle contact during flotation. Finally, XRF and ash analyses point to a moderate mineral content, implying that surface heterogeneity may play a meaningful role in governing flotation response and the partitioning of OMWW constituents. This characterization framework provides the basis for interpreting the flotation performance and phenolic removal outcomes discussed in the following section.
3.2 Characterization of olive mill wastewater (OMWW)
Before assessing phenolic removal by coal-assisted flotation, the chemical features of olive mill wastewater (OMWW) were examined to establish a baseline description of the organic matrix.1,2 In this section, ATR-FTIR was used to capture the dominant functional groups present in raw OMWW and to relate these signatures to the persistence and inhibitory nature commonly reported for this wastewater.6,7The ATR-FTIR spectrum of OMWW (Fig. 6) displays several broad and intense features, reflecting a mixture of polar and aromatic structures. The wide band centered around ∼3400 cm−1 is consistent with overlapping –OH and –NH stretching vibrations and is commonly linked to phenolic hydroxyls, alcohol-type groups, and nitrogen-containing organics in OMWW.6 The prominence of this region suggests a high density of polar functionalities capable of hydrogen bonding, a hallmark of OMWW chemistry.8 A clear contribution near ∼2900 cm−1 corresponds to aliphatic C–H stretching range, indicating the presence of hydrocarbon chains associated with residual oils and non-polar organic matter.11 In the 1700–1600 cm−1 region, the spectrum shows a strong carbonyl-related contribution attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching, consistent with oxygenated compounds such as organic acids and ketone-like structures.7 The signal around ∼1600 cm−1 further supports the presence of aromatic moieties through C
O stretching, consistent with oxygenated compounds such as organic acids and ketone-like structures.7 The signal around ∼1600 cm−1 further supports the presence of aromatic moieties through C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations, in agreement with the aromatic nature of phenolic constituents in OMWW.8
C stretching vibrations, in agreement with the aromatic nature of phenolic constituents in OMWW.8
At lower wavenumbers, multiple bands in the 1300–1000 cm−1 range are associated with C–O stretching vibrations, again pointing to a matrix dominated by oxygen-containing organic species.6 Overall, the spectrum indicates that OMWW contains abundant phenolic and oxygenated functional groups, which plausibly underlie its high organic load and the limited effectiveness of conventional treatment routes.7,8
3.3 Coal-assisted flotation and phenolic compound removal
Following the characterization of coal surface properties and the chemical features of olive mill wastewater, the phenolic removal performance of the coal-assisted flotation process was evaluated under systematically varied operating conditions. Given the tendency of phenolic compounds to associate with hydrophobic and aromatic domains, coal-assisted flotation provides a natural framework in which adsorption on coal surfaces can operate in parallel with particle–bubble separation.12,13,17 Comparable adsorption phenomena have been reported for phenolic compounds on coal ash and activated carbons, reinforcing the plausibility of coal surfaces acting as effective sinks for OMWW organics.11–13 Related mineral engineering studies have emphasized the role of interfacial adsorption and hydrodynamic conditions in governing flotation selectivity for coal residues, reinforcing the mechanistic parallels observed here with OMWW suppression at higher loadings.34The flotation experiments were conducted according to the procedure and the conditions discussed above in the Methods section, and the results were presented as recovery curves where the ash contents of concentrate and tailing fractions (Table 3) were used to calculate recoveries. As expected, a decrease in ash content was observed in the concentrate phase, while the tailings exhibited higher mineral content, indicating partial upgrading of coal during flotation. Higher OMWW concentrations reduced flotation efficiency; at 100% OMWW, practically no flotation took place, indicating suppressed particle–bubble attachment.
| Sample | Ash (%) |
|---|---|
| Raw coal | 12.33 |
| 5% OMWW – tail | 13.23 |
| 5% OMWW – concentrate | 10.34 |
| 10% OMWW – tail | 13.1 |
| 10% OMWW – concentrate | 9.82 |
| 20% OMWW – tail | 11.99 |
| 20% OMWW – concentrate | 8.43 |
| 40% OMWW – tail | 9.56 |
| 40% – concentrate | 5.67 |
| 100% OMWW – tail | 8.56 |
| 100% – OMWW concentrate | — |
The outcomes in Fig. 7 clearly show that the OMWW fraction the process water governs flotation performance. At low OMWW fractions (5–10%), flotation recoveries remained relatively high (61.28% and 47.04%, respectively), and the pH of the treated wastewater increased to near-neutral values (pH = 5.92). This shift is consistent with the removal of acidic organic species adsorbed onto coal and separated with the floated phase. Similar neutralization effects have been observed in recent OMWW treatment studies employing coagulation–flocculation and photocatalysis, where partial removal of phenolic acids led to measurable pH increases.8 The present results, therefore, confirm that flotation can achieve comparable chemical improvements with lower reagent demand, positioning it as a complementary option within the spectrum of OMWW treatment technologies. Raising the OMWW fraction to 10% led to a moderate reduction in recovery (47.04%). Beyond this range, however, flotation efficiency dropped sharply: recoveries decreased to 20.27%, 15.48%, and 14.80% at 20%, 40%, and 100% OMWW, respectively. These results indicate that limited amounts of OMWW constituents can support effective treatment while maintaining flotation functionality, whereas higher OMWW loadings progressively suppress flotation performance, consistent with an overloaded matrix that disrupts bubble–particle interactions.
At low OMWW ratios, recovery remains high, ensuring reliable bubble retention and stable flotation, as the coal surface is sufficiently hydrophobic by the collector-surfactant system.16,28 As the OMWW ratio increases, the interface is no longer “collector-dominant”. Phenolics and other polar organics increasingly occupy the coal surface, shifting the balance between hydrophobic and polar areas. This effectively screens the hydrophobic patches and shifts the surface towards a more water-sensitive character. When this shift becomes significant, bubble–particle retention weakens, selectivity collapses, and recovery rapidly decreases. Therefore, the sharp drop at higher OMWW ratios reflects a simple competition: hydrophobic activation supports flotation, while polar wastewater components progressively weaken it.
pH trends exhibit similar threshold-type behavior, helping to separate flotation effects from dilution (Fig. 8). Before flotation (top graph), the pH decreases from ∼4.26 at 5% OMWW to ∼3.95 at 100% OMWW which is expected to increase acidic organic matter content. After flotation (bottom graph), the pH rises strongly for 5–40% OMWW, reaching 5.38–5.92. This increase is consistent with the removal of acidic organic species adsorbed onto the coal and separated from the liquid phase by the foam product. Below 100% OMWW, the pH change is minimal (∼4.39), consistent with low recovery and suggesting that the high organic load suppresses the binding step. In short, the pH increase at low to medium OMWW ratios is mainly due to the removal of acidic components through flotation and becomes marginal when the OMWW is highly concentrated.
That is, the results reveal a clear dependence of pH on the proportion of OMWW in the flotation medium. At low OMWW fractions (5–10%), the pH values remained in the mildly acidic to near-neutral range, whereas a progressive decrease in pH was observed with further increases in OMWW content. When the OMWW fraction reached 100%, the pH decreased to approximately 4.39, approaching the initial acidity of raw OMWW (pH ≈ 4.09).
This trend reflects the inherently acidic nature of OMWW, which is primarily attributed to its high content of organic acids, phenolic compounds, and carboxyl functional groups. As the OMWW fraction increases, these acidic constituents increasingly dominate the aqueous phase, leading to a systematic reduction in pH. The lower pH conditions promote surface hydrophilization of coal particles, which can weaken hydrophobic interactions and negatively affect flotation performance.28,31
Interestingly, at low to moderate OMWW fractions (5–10%), a slight increase in pH compared to raw OMWW was observed. This behaviour suggests that a portion of acidic organic compounds may be adsorbed onto the coal surface32 or removed with the floated phase during flotation, resulting in partial neutralization of the medium. Such pH shifts indicate that the coal-assisted flotation process not only facilitates solid–liquid separation but also contributes to reducing the acidic load of the wastewater.
The observed pH variations are closely associated with phenolic removal during coal-assisted flotation. At low OMWW fractions, higher pH values coincide with increased phenolic recovery, indicating that the partial removal of acidic phenolic compounds creates a more favourable surface chemistry for flotation.17 In contrast, higher OMWW fractions maintain lower pH conditions due to the accumulation of acidic organic species, which promotes coal surface hydrophilization and weakens particle–bubble interactions.31 This relationship highlights the coupled influence of chemical environment and surface interactions on flotation performance.
The FTIR spectra comparing raw olive mill wastewater (OMWW) with the treated effluent (tail) obtained at 5% and 10% OMWW fractions provide further insight into the phenolic removal mechanism (Fig. 9). The spectrum of raw OMWW exhibits pronounced features associated with phenolic and oxygen-containing functional groups, particularly in the regions corresponding to aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C
C, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and C–O vibrations.6,8 After flotation, these characteristic features are noticeably attenuated in the tail samples, with a more pronounced reduction observed at lower OMWW fractions. The diminished intensity of bands related to aromatic and oxygenated functionalities indicates the partial removal of phenolic compounds from the aqueous phase during coal-assisted flotation. This spectral evolution supports the adsorption of phenolic species onto the coal surface, and their subsequent separation with the phase floated.
O, and C–O vibrations.6,8 After flotation, these characteristic features are noticeably attenuated in the tail samples, with a more pronounced reduction observed at lower OMWW fractions. The diminished intensity of bands related to aromatic and oxygenated functionalities indicates the partial removal of phenolic compounds from the aqueous phase during coal-assisted flotation. This spectral evolution supports the adsorption of phenolic species onto the coal surface, and their subsequent separation with the phase floated.
 | ||
| Fig. 9 FTIR spectra of raw olive mill wastewater (OMWW) and treated effluents (tail) obtained at 5% and 10% OMWW fractions. | ||
The phenol removal observed in the flotation experiments confirms the adsorption role of coal surfaces under realistic OMWW conditions. These findings are consistent with the quantitative adsorption parameters previously reported for Soma lignite (Polat et al.,13 2006), where adsorption capacity, kinetics, and isotherm modeling were systematically evaluated. In the present study, the flotation data provide direct evidence of phenolic removal from complex wastewater matrices, while the earlier adsorption study supplies the quantitative foundation for interpreting the mechanistic pathway. Together, these complementary datasets demonstrate that phenolic adsorption onto coal is both quantitatively established and operationally effective within the coal-assisted flotation framework.
Fig. 10 presents the phenol removal efficiency (mg g−1 coal) at OMWW fractions of 5% and 10% where a better flotation performance was observed. The actual phenol amounts in the 1 liter flotation cell for these OMWW fractions were 873 and 1633 mg, respectively. The figure shows that phenol adsorption capacities observed were 33 mg phenol per gram of coal for 5% OMWW and 62 mg phenol per gram of coal for 10% OMWW. These values are lower than the usual adsorption capacities observed with activated carbons (100–300 mg g−1), most probably due to the small surface area of the lignite sample (7.7 m2 g−1) compared to those usually associated with activated carbons (in the order of 1000 m2 g−1). However, considering that 25 grams of lignite was present in the flotation cell, these adsorption values correspond to overall adsorbed phenol amounts of 825 mg and 1550 mg, translating to phenol removal efficiencies of 94.5% and 94.9%.
Overall, the results demonstrate that coal-assisted flotation can be a very effective approach for phenolic compound removal from olive mill wastewater, at low to moderate OMWW fractions. The evaluation of all the characterization results of (1) flotation products (float, tail-solid, and tail-water) (2) initial coal surface and wastewater content, confirms that flotation recovery and the phenol removal efficiency of wastewater seem to depend on coal surface characteristics (availability of both hydrophilic and hydrophobic parts) and the amount of oil in wastewater. Since adsorption of phenolic compounds on hydrophilic parts and the use of oil to make exactly these specific parts hydrophobic happen spontaneously, there is a certain amount of oil that the system can handle. Increasing OMWW content intensifies surface hydrophilization and suppresses particle–bubble interactions, thereby limiting separation efficiency. Overall, the results suggest that coal-assisted flotation is a promising and mechanistically supported strategy for phenolic compound removal under dilute OMWW conditions, while its efficiency becomes increasingly constrained as the complexity of the wastewater matrix increases.
While OMWW treatment has been widely studied through biological, chemical, and membrane-based processes, flotation remains underexplored in this context. Recent reviews emphasize the need for scalable, resource-oriented solutions that integrate treatment with valorization.6,7 By demonstrating that coal-assisted flotation can simultaneously reduce phenolic load and upgrade lignite quality, this study addresses precisely that gap. The dual benefit—wastewater remediation and coal beneficiation—situates flotation as a practical, circular economy-aligned approach that complements existing technologies and offers industrial feasibility.
4. Conclusions
This study demonstrates that coal-assisted flotation can serve as a practical, integrated route for olive mill wastewater (OMWW) remediation. At low to moderate OMWW fractions, flotation achieved meaningful phenolic reduction and acidity mitigation, while simultaneously upgrading Soma lignite quality. The dual outcome—wastewater treatment and coal beneficiation—highlights the potential of flotation to bridge environmental management with resource recovery.The adsorption of phenolic compounds onto coal surfaces, corroborated by spectroscopic evidence, aligns with broader findings on carbonaceous materials as effective sinks for aromatic pollutants.11–13 The observed pH shifts further confirm that flotation not only separates solids but also reduces the acidic load of OMWW, echoing improvements reported in recent green treatment approaches such as coagulation–flocculation combined with photo catalysis.8 By achieving comparable chemical benefits with lower reagent demand, flotation positions itself as a complementary technology within the OMWW treatment landscape.
From a broader perspective, the integration of wastewater remediation with coal upgrading resonates strongly with circular economy principles. Recent analyses emphasize the need for scalable, resource-oriented solutions that valorize waste streams while reducing environmental burdens.6,7 Coal-assisted flotation directly addresses this challenge by coupling pollutant removal with material improvement, offering a pathway that is both operationally feasible and industrially relevant.
In summary, the results establish coal-assisted flotation as a promising option for OMWW management, particularly under conditions where conventional biological or chemical treatments face limitations. By situating flotation within the broader context of sustainable wastewater treatment and resource recovery, this work contributes to advancing practical solutions for agro-industrial effluents and underscores the importance of linking surface chemistry with process design.
Author contributions
H. Polat conceptualized the study and supervised the research. M. Polat contributed to flotation design and process optimization. M. C. Eren, Ç. Güngör and E. Özsaygı performed experiments and data analysis. H. Polat drafted the manuscript with input from all authors. All authors reviewed and approved the final version.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study are included within the article.Acknowledgements
The authors have employed the assistance of Microsoft Copilot in editing the manuscript for English and and organizing the references. The authors appreciate the assistance from Olive Oil Factory (AYVALIK-TURKEY) for providing OMWW.References
- L. M. Fleyfel, J. Matta, N. F. Sayegh and N. H. El Najjar, Olive mill wastewater treatment using coagulation/flocculation and filtration processes, Heliyon, 2024, 10, e40348, DOI:10.1016/j.heliyon.2024.e40348.
- J. Wang, L. Zhang, Y. He and R. Ji, J. Hazard. Mater., 2024, 469, 133906, DOI:10.1016/j.jhazmat.2024.133906.
- W. Issaoui, I. H. Nasr, M. H. Inoubli and I. M. Ibraheem, Water, 2026, 18, 149, DOI:10.3390/w18020149.
- T. Vaz, M. J. Quina, R. C. Martins and J. Gomes, Sci. Total Environ., 2024, 931, 172676, DOI:10.1016/j.scitotenv.2024.172676.
- M. J. Fernandes, J. Gomes, P. Carvalho, R. C. Martins and E. Domingues, Chem. Eng. Sci., 2025, 305, 121145, DOI:10.1016/j.ces.2024.121145.
- I. E. Kapellakis, V. A. Tzanakakis and E. M. Kabourakis, Front. Sustain., 2025, 6, 1545806, DOI:10.3389/frsus.2025.1545806.
- A. Feyzioğlu, S. Ersoy, T. U. Omoruyi, D. Santoro and L. Piccinetti, Insights Reg Dev, 2024, 6, 92–102, DOI:10.9770/ird.2024.6.2(6).
- I. Michael, D. Panagiotopoulou, D. Fatta-Kassinos and D. D. Dionysiou, Water Res., 2014, 60, 28–40, DOI:10.1016/j.watres.2014.04.027.
- G. Z. Kyzas and K. A. Matis, Processes, 2018, 6, 116, DOI:10.3390/pr6080116.
- W. Hua, Y. Wen, B. Wang, M. Fan, H. Zhang and W. Huang, J. Indian Chem. Soc., 2024, 101, 101376, DOI:10.1016/j.jics.2024.101376.
- S. Wang and Y. Peng, Chem. Eng. J., 2010, 156, 11–24, DOI:10.1016/j.cej.2009.10.029.
- A. Dąbrowski, P. Podkościelny, Z. Hubicki and M. Barczak, Chemosphere, 2005, 58, 1049–1070, DOI:10.1016/j.chemosphere.2004.09.067.
- H. Polat, M. Molva and M. Polat, Int. J. Miner. Process., 2006, 79, 264–273, DOI:10.1016/j.minpro.2006.03.003.
- Z. Zhang, C. Wu and K. Yan, J. Mol. Liq., 2020, 319, 114175, DOI:10.1016/j.molliq.2020.114175.
- M. An, Y. Liao, R. Yin, E. Wang and Y. Yoon, Processes, 2021, 9, 404, DOI:10.3390/pr9030404.
- Y. Zhang, R. Ding, X. Cui, Y. Qin, G. Abaka-Wood and E. Li, ACS Omega, 2024, 9, 7108–7118, DOI:10.1021/acsomega.3c08918.
- M. Xu, A. Vanderbruggen, N. Kupka, H. Zhang and M. Rudolph, Miner. Eng., 2022, 185, 107714, DOI:10.1016/j.mineng.2022.107714.
- N. Russo, P. R. Avallone, N. Grizzuti and R. Pasquino, Colloids Surf., A, 2024, 701, 134776, DOI:10.1016/j.colsurfa.2024.134776.
- M. Coates, Interpretation of Infrared Spectra, A Practical Approach, in Encycl. Anal. Chem., 2006, DOI:10.1002/9780470027318.a5606.
- A. Mekki, A. Dhouib and S. Sayadi, Bioresour. Technol., 2007, 98, 1393–1397, DOI:10.1016/j.biortech.2006.05.030.
- E. Marguí, I. Queralt and E. de Almeida, Chemosphere, 2022, 303, 135006, DOI:10.1016/j.chemosphere.2022.135006.
- M. Arif, F. U. R. Awan, H. Zhang and M. Hosseini, Energy Fuels, 2024, 38, 15069–15084, DOI:10.1021/acs.energyfuels.4c03052.
- A. V. Delgado, F. González-Caballero, R. J. Hunter, L. K. Koopal and J. Lyklema, J. Colloid Interface Sci., 2007, 309, 194–224, DOI:10.1016/j.jcis.2006.12.075.
- G. S. Simate, N. Maledi, A. Ochieng, S. Ndlovu, J. Zhang and L. F. Walubita, J. Environ. Chem. Eng., 2016, 4, 2291–2312, DOI:10.1016/j.jece.2016.03.051.
- M. Polat, H. Polat and S. Chander, Int. J. Miner. Process., 2003, 72, 199–213, DOI:10.1016/s0301-7516(03)00099-1.
- C. Moreno-Castilla, Carbon, 2004, 42, 83–94, DOI:10.1016/j.carbon.2003.09.022.
- T. Lu, T. Wang, L. Yang, H. Wang and Y. Wang, Sep. Sci. Technol., 2023, 41(4), 1344–1355 CAS.
- X. Wang, Y. Zhao, H. Liu and Y. Xie, Minerals, 2024, 14, 952, DOI:10.3390/min14090952.
- J. G. Speight, The Chemistry and Technology of Coal, CRC Press, 3rd edn, 2013, ch. 2, pp. 45–70 Search PubMed.
- S. Dai, D. Ren, C.-L. Chou, R. B. Finkelman, V. V. Seredin and Y. Zhou, Int. J. Coal Geol., 2012, 94, 3–21, DOI:10.1016/j.coal.2011.02.003.
- D. Gui, S. Chen, S. Wang, X. Tao, L. Chen and R. Wang, Energy Sources, Part A, 2020, 46(1), 14699–14712 CrossRef.
- M. Ahmaruzzaman and D. K. Sharma, J. Colloid Interface Sci., 2005, 287, 14–24, DOI:10.1016/j.jcis.2005.01.075.
- G. Cheng, R. Guo, P. Gao, C. Xu, P. Duan, Z. Zhang, X. Deng and K. Yan, Chem. Eng. J., 2026, 529, 172241, DOI:10.1016/j.cej.2025.172241.
- G. Cheng, H. Han, P. Duan and E. V. Lau, Miner. Eng., 2026, 242, 110049, DOI:10.1016/j.mineng.2025.110049.
| This journal is © The Royal Society of Chemistry 2026 |








