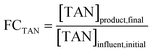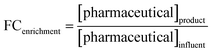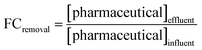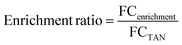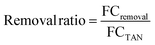Open Access Article
Open Access ArticleFate of organic contaminants in electrochemical nitrogen recovery from urine
Anna
Kogler
 a and
William A.
Tarpeh
a and
William A.
Tarpeh
 *ab
*ab
aDepartment of Civil and Environmental Engineering, Stanford University, 443 Via Ortega, Room 387, Stanford, CA 94305, USA
bDepartment of Chemical Engineering, Stanford University, 443 Via Ortega, Room 387, Stanford, CA 94305, USA. E-mail: wtarpeh@stanford.edu; Tel: +1 650-497-1324
First published on 21st April 2026
Abstract
This study compared the fate of pharmaceuticals and disinfection byproducts across three electrochemical nitrogen recovery processes treating urine: electrochemical stripping (ECS), electrodialysis (ED), and bipolar electrodialysis (BPED). ECS achieved greater TAN recovery efficiencies than ED and BPED and was the only studied process that recovered a urine-derived ammonium sulfate product rather than a mixed concentrate (containing TAN, sodium, and potassium). Enrichment and removal ratios based on target pharmaceutical quantification and suspect screening were used to evaluate the fate of organic compounds relative to nitrogen recovery. These metrics suggested that ECS prevented organic contamination of the product more effectively than the other two processes and that both ECS and BPED achieved greater organic removal than ED. No disinfection byproducts were detected in the product for any process, but formation in other chambers may require mitigation. Our findings inform pre- and post-treatment for electrochemical nitrogen recovery, as well as modifications to reactor configuration and operating conditions. With future work using suspect screening and identification of additional compounds to build greater mechanistic understanding of compound fate and to characterize the toxicity of contaminants detected in treated urine and recovered products, our work will advance electrochemical technologies that recover high-purity, safely applied products and enable a circular nitrogen economy.
Environmental significanceNitrogen recovery from wastewater can reduce the environmental impacts and cost of wastewater treatment and ammonia production. Electrochemical processes are an emerging technology that can enable nitrogen recovery in a modular, scalable, and automatable way using high-nitrogen waste streams like urine. However, pharmaceutical prevalence in urine poses a risk to recovered nitrogen product purity and could contribute to the already common occurrence of pharmaceuticals in the environment. Non-target analysis is needed to broadly understand transformation and fate of organic contaminants and to ensure that electrochemically recovered nitrogen products and treated waste streams are safe. Addressing concerns about organic micropollutants would eliminate a barrier to using recovered nitrogen products among farmers and consumers to facilitate environmentally responsible implementation. |
1 Introduction
Nitrogen in wastewater has negative environmental, human health, and economic impacts, including impairing drinking water quality and causing widespread eutrophication that leads to tens to hundreds of billions of dollars in annual economic losses in the U.S. alone.1 To reduce nitrogen discharges to the environment, wastewater treatment currently uses substantial chemical and energy inputs.2,3 Meanwhile, nitrogen is a critical component of fertilizers and other industrial chemicals,4 and ammonia synthesis via the Haber–Bosch process accounts for about 1% of global fossil fuel energy consumption5 and 1.4% of global carbon dioxide emissions.6 Recovering nitrogen from wastewater for beneficial reuse in a circular economy can reduce environmental impacts and costs of wastewater treatment and chemical production. Because 80% of nitrogen in wastewater comes from urine that accounts for only 1% of wastewater volume,7 nitrogen can be removed more efficiently from urine than combined wastewater.8 Multiple studies have demonstrated acceptance of urine-derived fertilizers among farmers and food consumers, indicating a potential market and supporting the economic viability of urine-derived fertilizers.9–15 Urine's high conductivity (16–35 mS cm−1 for urine16–19 compared to 1 mS cm−1 for domestic wastewater20–22) also facilitates electrochemical treatment with minimal energy consumption because of low solution resistance. Electrochemical urine treatment processes have achieved nitrogen removal, pharmaceutical degradation or separation, energy recovery (e.g., via hydrogen generation), and selective nitrogen recovery.23 Electrochemical nitrogen recovery technologies may be particularly promising for distributed urine treatment (e.g., toilet-scale, building scale) due to their modularity, process stability, reduced chemical inputs, automation, and ease of maintenance and operation.23–25However, on average 65% of pharmaceuticals are excreted via urine,26 and concerns about environmental contamination with urine-derived pharmaceuticals and the purity of urine-derived products pose a major barrier to implementing nitrogen recovery from urine. Many pharmaceuticals are poorly removed in conventional wastewater treatment trains.27,28 Furthermore, residual contaminants and their transformation products are becoming prevalent in the environment (e.g., pharmaceuticals detected in 80% of U.S. streams29), threatening humans and aquatic species.29–31 Farmers and the general public mention micropollutants, especially pharmaceuticals, among the most important risks associated with urine-derived fertilizers.9–11,13–15 To be widely implemented, nitrogen recovery processes must prevent further environmental contamination and effectively separate pharmaceuticals from recovered products. Thus, it is critical to understand the fate of pharmaceuticals in electrochemical nutrient recovery systems to enable development of mitigation strategies.
To date, research on pharmaceutical fate in electrochemical and other urine treatment processes has largely focused on specific pharmaceuticals via target analysis. Targeted studies have documented the extent of removal of particular pharmaceuticals in processes designed for micropollutant removal from urine, such as granular activated carbon adsorption,32 nanofiltration,33 electrochemical oxidation,34 and UV-based advanced oxidation processes.35 Researchers have also evaluated the fate of pharmaceuticals in urine treatment processes focusing on volume reduction (e.g., evaporation36) and nutrient recovery (e.g., ion exchange,37 electrochemical stripping,19 struvite precipitation38). Similar evaluations have been conducted for processes that simultaneously recover nutrients and remove micropollutants from urine.36,39 Electrochemical urine treatment is still nascent and at lower technology readiness levels than most other urine treatment technologies;23 thus studies of pharmaceutical fate during electrochemical treatment of other wastewaters (e.g., anaerobic digestate, reverse osmosis concentrate, secondary effluent) could supplement understanding of urine treatment. These studies also typically use target analysis to evaluate pharmaceutical removal in direct and indirect electrochemical oxidation,40–46 electrodialysis,47,48 and electrochemical membrane biofilm reactors49 and to evaluate pharmaceutical fate in electrodialysis for nutrient recovery.50–52 Target analysis facilitates evaluation of fate and treatment performance for a limited number of species but leaves many compounds, including transformation products that may be more toxic and persistent than parent compounds, unidentified and unquantified.25,39,53
Analyzing larger data sets via nontarget analysis of trace organic compounds enables more comprehensive understanding of the electrochemical fate of pharmaceuticals and transformation products. Whereas true nontarget analysis makes no assumptions about the likelihood of detecting any compound, suspect screening (one type of nontarget analysis) uses a list of compounds (i.e., suspects) that are likely present based on the sample matrix (e.g., urine).54 Both approaches facilitate detection of pharmaceuticals, metabolites formed in humans, and transformation products using mass spectral data for tentative compound identification without reference standards and without a priori selection criteria for compounds of interest. These techniques can semi-quantitatively relate effluent and influent samples and enable retrospective analysis of compounds identified or synthesized in the future.53,55–62 By expanding the set of identifiable compounds beyond target analysis, non-target analysis and suspect screening can more broadly elucidate the fate of pharmaceuticals and other organic contaminants in electrochemical urine treatment, but such studies remain limited. Suspect screening and non-target analysis have been preliminarily conducted for electrochemical oxidation processes43,63,64 that do not specifically target nutrient removal or recovery from urine to identify transformation products and elucidate degradation pathways for a select few pharmaceuticals (e.g., carbamazepine43,63). Suspect screening and non-target analysis are needed to identify risks associated with a broad array of pharmaceutical and pharmaceutical-derived contaminants. This knowledge can inform strategies for mitigation in electrochemical nitrogen recovery processes treating urine and support the establishment of urine treatment standards65 that can facilitate widespread implementation of urine diversion, treatment, and resource recovery for sustainable nitrogen and wastewater management.
In addition to pharmaceuticals, disinfection byproducts (DBPs) pose an obstacle to electrochemical water and urine treatment23,25,66,67 because they are reported as a barrier or knowledge gap for 40% of electrochemical nutrient removal or recovery technologies.68 Many DBPs are cytotoxic, neurotoxic, mutagenic, or genotoxic69–71 and are associated with increased cancer risk and other detrimental health impacts (e.g., congenital disorders).71–74 Some DBPs, including trihalomethanes (THMs), haloacetic acids (HAAs), and dichloroacetonitrile (DCAN), are regulated drinking water contaminants.75 DBPs are increasingly prevalent throughout the environment76,77 and have negative impacts on aquatic organisms,78,79 such as endocrine disruption in shrimp80 and toxicity in various species.81–83 Electrochemical processes often expose chloride-rich urine (1600–7700 mg/L chloride23,84) to an oxidizing environment, which can lead to DBP formation. For example, during electrochemical oxidation of latrine wastewater, two regulated DBPs (chloroform and haloacetic acids) were observed at concentrations 10 to 1000 times the EPA drinking water maximum contaminant levels.85 Characterizing DBP formation and fate in electrochemical nutrient recovery systems is essential to developing mitigation strategies that reduce effluent toxicity and enhance product purity, enabling these technologies to up-concentrate nutrients without concomitant accumulation of harmful byproducts.
This study evaluates potential risks of electrochemical technologies for nitrogen recovery from urine by characterizing the fate of pharmaceuticals and DBPs in electrochemical stripping (ECS), electrodialysis (ED), and bipolar electrodialysis (BPED). These three technologies were selected because they rely on similar mechanisms for nitrogen removal/recovery (i.e., electromigration across ion exchange membranes), which enables evaluation of the impacts of reactor architecture and different types of membranes on nitrogen removal/recovery and byproduct fate. Moreover, ECS is representative of an emerging nitrogen recovery technique combining electrodialysis with membrane stripping.xkogler19,86 ED is a more established electrochemical treatment process that can be useful for benchmarking newer processes like ECS;23,87 and BPED utilizes bipolar membranes, which are increasingly studied due to their lower energy consumption.88,89 For urine treatment using these three technologies, the specific objectives of this study were to (1) compare pharmaceutical concentrations in treated effluent to existing regulatory standards, (2) evaluate recovered product purity and safety based on the fate of pharmaceuticals and their transformation products within the reactors, and (3) elucidate mechanisms that govern organic compound fate during electrochemical nitrogen treatment. Our results inform pre- and post-treatment strategies and motivate concrete objectives for optimizing reactor configuration and operating conditions, which are among the most common knowledge gaps facing nutrient technologies at large.68 Based on elucidating and addressing these barriers, outcomes of this study will advance electrochemical nutrient recovery technologies toward implementation.
2 Methods
2.1 Urine collection, storage, and treatment
All processes evaluated in this study treated hydrolyzed human urine collected in the Shriram Center for Chemical Engineering and Bioengineering at Stanford University. Urine was collected from July 3 to October 8, 2021 with approximately equal contributions from male and female donors over age 18 (Internal Review Board Protocol 60601). Urine was stored in a closed container at room temperature (25 °C) and 0.6 mg/L urease (41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 050 units per g solid, Sigma-Aldrich, St. Louis, MO) was added to accelerate urea hydrolysis. Due to low natural pharmaceutical levels, collected urine was spiked with 20 µg/L of acetaminophen, emtricitabine, iopromide, atenolol (ATE), sulfamethoxazole, N-acetyl-sulfamethoxazole, trimethoprim (TMP), metoprolol (MET), naproxen, bezafibrate (BZF), propranolol (PRO), carbamazepine (CBZ), and diclofenac (DIC); this concentration is approximately representative of realistic urine.26 This set of compounds enabled quantification of target pharmaceuticals for validation of the non-target analysis workflow. Urine was characterized comprehensively (including total ammonia nitrogen (TAN), ions, and pH (Section S1.1), as well as pharmaceuticals (Section 2.3)) prior to each experiment to verify that composition remained consistent over time (Table S1-1).
050 units per g solid, Sigma-Aldrich, St. Louis, MO) was added to accelerate urea hydrolysis. Due to low natural pharmaceutical levels, collected urine was spiked with 20 µg/L of acetaminophen, emtricitabine, iopromide, atenolol (ATE), sulfamethoxazole, N-acetyl-sulfamethoxazole, trimethoprim (TMP), metoprolol (MET), naproxen, bezafibrate (BZF), propranolol (PRO), carbamazepine (CBZ), and diclofenac (DIC); this concentration is approximately representative of realistic urine.26 This set of compounds enabled quantification of target pharmaceuticals for validation of the non-target analysis workflow. Urine was characterized comprehensively (including total ammonia nitrogen (TAN), ions, and pH (Section S1.1), as well as pharmaceuticals (Section 2.3)) prior to each experiment to verify that composition remained consistent over time (Table S1-1).
Electrochemical stripping (ECS) and bipolar electrodialysis (BPED) (Fig. 1) were evaluated using 12-hour batch experiments in which solutions within each chamber were recirculated internally. Electrodialysis (ED) was evaluated using 4-hour batch experiments due to potentiostat limits (Fig. S1-1). We chose batch over continuous operation to facilitate comparison of our results for electrochemical processes to prior non-target analyses and suspect screens conducted for other urine treatment processes.90 Batch experiments also facilitated collection of sufficient sample volume at each time point to characterize both pharmaceuticals and disinfection byproducts. For each reactor, a constant current density of 100 A/m2 was applied using a VMP-300 potentiostat (Biologic, Seyssinet-Pariset, France) with a leakless eDAQ Ag/AgCl reference electrode placed in the chamber containing the anode (eDAQ, Colorado Springs, CO). Solutions (prepared as described in Section S1.2) were recirculated within each reactor chamber at a flowrate of 75 mL/min for 4 (ED) or 12 (ECS and BPED) hours. Additional details about reactor setup and operation and sample collection (Table S1-2) are provided in Section S1.3.
2.2 Evaluation of nitrogen recovery performance
Total ammonia nitrogen (TAN) concentrations were measured via segmented flow analysis (SEAL Analytical, Mequon, WI) using a phenol-based colorimetric method (Section S1.1). The fold change in TAN concentration from influent to product, FCTAN, was calculated as follows:We expected FCTAN to be approximately one because experiments were designed to achieve complete TAN recovery.
2.3 Pharmaceutical analysis
Samples for pharmaceutical analysis were prepared by solid phase extraction (SPE) as described in Section S1.4.1. Extracts were stored at −20 °C until analysis via liquid chromatography-mass spectrometry (LC-MS) for target analysis and liquid chromatography-high resolution mass spectrometry (LC-HRMS) for suspect screening.where [pharmaceutical] is the concentration or peak intensity for a single compound at a particular time in one of the reactor chambers. The specific samples for each reactor denoted by the subscripts in the formulas above are shown in Table S1-13 and Fig. S1-3.
Pharmaceutical fate relative to nutrient recovery was quantified according to enrichment and removal ratios:
Optimal performance would achieve a low enrichment ratio (i.e., high TAN recovery and low pharmaceutical contamination in product). The removal ratio captures the extent of pharmaceutical removal achieved (relative to ammonia recovery) via chemical transformation and physical migration across ion exchange membranes. With a focus on maximizing nutrient recovery, a low removal ratio would be optimal. However, if pharmaceuticals are transformed to less toxic and less persistent compounds, higher removal ratios would be desirable. Fold changes and ratios were calculated for target compounds and features found via suspect screening to facilitate comparison of the two ratios and evaluate how representative target compounds are of the broader array of pharmaceuticals and organic compounds found in urine.94
2.4 Disinfection byproduct analysis
DBPs in seven classes (trihalomethanes or THMs, haloacetic acids or HAAs, haloacetonitriles or HANs, haloacetaldehydes or HALs, haloketones or HKs, halonitromethanes or HNMs, and haloacetamides or HAMs, Table S1-14) were extracted using modified EPA methods 551.1 and 552.3 and analyzed via gas chromatography-mass spectrometry (GC-MS, Agilent, Santa Clara, CA). Details on analytes and methods are included in Section S1.5 (detection limits in Table S1-14). Both regulated and unregulated DBPs were evaluated because previous work suggested that regulated DBP concentrations may not adequately indicate overall DBP exposure96 and unregulated DBPs, such as HAMs and HALs, exhibit higher cytotoxicity than regulated DBP classes.97 Iodinated trihalomethanes (I-THMs) were excluded from analysis because initial screening experiments showed no I-THM formation. We chose to report only presence/absence of DBPs, rather than measured concentrations. Concentration variability between experiments was high, but presence/absence of compounds was consistent. Presence was defined as detection in at least 5 of 6 replicate samples.3 Results and discussion
3.1 Nitrogen removal and recovery performance
The three electrochemical processes – ECS, ED, and BPED – rely on electrochemically generated pH changes, electromigration, and diffusion to remove and recover nitrogen from urine. For ECS, current applied between Chambers 1 and 2 lowers the pH of influent urine in Chamber 1 and converts NH3 to NH4+. NH4+ ions migrate across the CEM from Chamber 1 to 2. In Chamber 2, applied current raises the pH, converting NH4+ ions into NH3. NH3 volatilizes and diffuses across the GPM from Chamber 2 to 3. Sulfuric acid in Chamber 3 converts NH3 to NH4+, allowing recovery of ammonium sulfate solution. For ED, urine is introduced into Chamber 2 (diluate chamber). Electric current applied between Chambers 1 and 3 drives NH4+ across the CEM from Chamber 2 to 3 (concentrate) while driving anions across the anion exchange membrane (AEM) from Chamber 2 to 1 (anode). The pH in Chamber 3 rises, while the pH in Chamber 1 decreases and the pH in Chamber 2 remains approximately constant at the wastewater's initial pH. For BPED, urine is also introduced into Chamber 2. Electric current applied between Chambers 1 and 4 drives water splitting in the BPM separating Chambers 1 and 2. The BPM releases protons into Chamber 2, lowering the pH of the urine and converting NH3 to NH4+. NH4+ ions electromigrate across the CEM into Chamber 3 along with other cations. An AEM separating Chambers 3 and 4 allows hydroxide ions generated in Chamber 4 to pass to Chamber 3, raising the pH in Chambers 3 and 4. Electric current and associated pH changes (Fig. S2-1) determine the fate of TAN and organic compounds in the system.Before investigating the fate of organic compounds, we characterized TAN removal and recovery performance (Tables S2-1, S2-2 and Fig. S2-2). ECS and BPED achieved nearly complete TAN removal within 12 hours (96.9 ± 0.5% and 88.9 ± 1.1%, respectively), while ED achieved 56.6 ± 4.0% removal. ECS achieved more complete recovery (97.2 ± 11% or fold change of 0.972 ± 0.11) than ED (64.3 ± 5.9% or fold change of 0.643 ± 0.059) and BPED (79.0 ± 6.2% or fold change of 0.790 ± 0.062). The lower removal and recovery efficiencies of ED were caused by shorter operation and higher pH in the diluate chamber (Fig. S2-1). At higher pH, more TAN in the diluate was present as NH3, which limited removal and recovery with only ion exchange membranes present in ED. Neutral NH3, unlike cationic NH4+, cannot be removed by electro-migration, only by diffusion from the diluate to the concentrate. However, TAN accumulation in the concentrate and the fact that NH3 was the dominant TAN species at the concentrate's high pH (11.4 ± 0.04 to 12.7 ± 0.03) reduced the driving force for diffusion. BPED's lower recovery efficiency was likely caused by its less closed TAN mass balance compared to ECS and ED (Fig. S2-3). In BPED, TAN loss was more likely than in ED and ECS: the concentrate compartment (Chamber 3) pH was alkaline while Chamber 3 in ECS was acidic and BPED was operated for 12 hours compared to 4 hours for ED, allowing NH3 volatilization. Given these performance metrics and the fact that ED and BPED recovered a mixed concentrate (containing all cations removed from urine, including Na+, K+, and NH4+) while ECS recovered ammonium sulfate, ECS was a more effective nitrogen recovery process than ED and BPED.
3.2 Fate of organic contaminants
The fate of organic contaminants was evaluated using both a target analysis of seven pharmaceuticals and a suspect screen relying on the Pubchemlite database and validated using the target analysis. Identification of detected features by the suspect screening workflow was validated by verifying detection of target compounds (Table S2-3). For ECS and BPED, 7 of 7 target formulas and compounds were annotated, while 6 of 7 targets were annotated for ED. These statistics help validate the suspect screening workflow although having a larger set of target compounds for validation would further improve confidence.No maximum allowed concentrations have been defined for our target compounds in recycled water policies in the U.S. but such levels have been set in Australia's recycled drinking water guideline (Table S1-12). For all target compounds except diclofenac, initial pharmaceutical concentrations in urine were below Australian guidelines (Table S1-1). Diclofenac concentrations were reduced to below the guideline in ECS and BPED, but not in ED (Table S2-1). Therefore, pharmaceutical degradation under acidic conditions may be important for enabling reuse of urine after electrochemical nutrient recovery.
Mass balances on target pharmaceuticals (i.e., totaling to less than 100% of initial mass present) indicate that the selected pharmaceuticals were transformed to varying degrees in the investigated electrochemical processes (Fig. S2-4). For example, trimethoprim, propranolol, and diclofenac were largely transformed during ECS, and metoprolol and carbamazepine to a lesser extent. Previous studies suggested that reaction with surface-bound reactive chlorine species significantly contributes to transformation of several of these compounds.43 ED less completely transformed compounds with only propranolol and diclofenac being removed to some extent. In BPED, propranolol and diclofenac were largely transformed, while trimethoprim, metoprolol, bezafibrate, and carbamazepine were partially transformed. For the compounds that were only partially transformed, most compounds remaining at the end of treatment were found in urine (i.e., influent), but some compounds migrated to other chambers. For example, atenolol crossed the CEM into the cathode or concentrate chambers in all three processes. Trimethoprim, propranolol, and diclofenac crossed the AEM toward the anode chamber in ED.
Compound properties explain observed target compound fates (Fig. S2-4 and Table S2-4). Diclofenac and bezafibrate are the only compounds with pKa below 5, but bezafibrate was mostly conserved (especially in ECS and ED) while diclofenac was largely removed from solution (especially in ECS and BPED). Both compounds are neutral at pH values observed in urine during ECS and the later part of BPED, preventing electromigration. Both are negatively charged at pH values observed in urine during ED, facilitating some electromigration across the AEM to the anode chamber. Diclofenac has been reported to adsorb to ion exchange membranes due to its high molecular volume and hydrophobicity when in neutral form, facilitating greater removal from solution during ECS and BPED than ED.98 Trimethoprim has a near-neutral pKa. Therefore, it is neutral at pH values observed in urine during ED, causing it to be mostly conserved. By contrast, trimethoprim becomes positively charged at low pH occurring in urine in ECS and BPED, enabling electromigration to Chamber 2 in ECS and Chamber 3 in BPED, where high pH converts the molecule to its neutral, adsorption-prone form and could lead to base-catalyzed hydrolysis. Trimethoprim's low Henry's constant makes it unlikely that volatilization would contribute to removal. More complete removal during ECS than BPED could result from urine pH decreasing more quickly during ECS than BPED, allowing more time for migration.
Atenolol, metoprolol, and propranolol have pKa values between 9.1 and 9.6. These compounds exist as a mixture of cationic and neutral forms in urine in ED but are mostly cationic at low pH occurring in urine in ECS and BPED. Greater electromigration in ECS and BPED than ED could enhance adsorption to CEMs and enable degradation under alkaline conditions occurring in Chamber 2 of ECS and Chamber 3 of BPED. Notably, the extent of removal of these three compounds matched the order of hydrophobicity with atenolol being least hydrophobic and least removed and propranolol being most hydrophobic and most removed. Greater hydrophobicity likely increases adsorption onto ion exchange membranes,99 and propranolol has been shown to adsorb to mixed metal anodes due to hydrophobicity.43 Carbamazepine is the target compound with the highest pKa (13.9). It was conserved in ED and BPED, and its fate in ECS was somewhat uncertain, suggesting some removal. Carbamazepine remains neutral across the pH range observed in all three processes, limiting electromigration. Removal during ECS could be due to degradation via indirect electrochemical oxidation in the presence of active chlorine species formed during ECS, with more effective degradation at lower pH.100 Adsorption to mixed metal anodes due to organic compound hydrophobicity43 can also contribute to removal in ECS, which is the only process studied that places urine in direct contact with the electrode. Carbamazepine is the most volatile of the target compounds with a Henry's constant 1 to 7 orders of magnitude greater than other compounds. The oxygen evolution reaction occurring at the anode in ECS could enhance removal via volatilization due to bubble formation, which does not occur in urine in the other processes. These patterns illustrate that pKa, hydrophobicity, and volatility are important properties affecting the fate of organic contaminants in electrochemical nitrogen recovery processes.
Based on suspect screening, volcano plots indicate a wide range of enrichment and removal ratios for all processes (Fig. 2A, S2-5, S2-6 and Table S2-5). ERs were shifted slightly leftward for ECS compared to those for ED and BPED, suggesting that having two barriers between influent urine and the product chamber (i.e., CEM and GPM) more effectively prevented organic contaminants from reaching the product than a single barrier (i.e., CEM). Differences were statistically significant except for the ER calculated from suspect data for ECS and BPED (Table S2-5). Average ER was less than 1 when considering only target compounds, indicating that TAN recovery was preferred over organic transport to the product (Fig. 2B, C and S2-7). The higher initial TAN concentration compared to that of pharmaceuticals leads to a higher concentration gradient, increasing the driving force for diffusion. TAN transport by diffusion in addition to electromigration would contribute to greater TAN recovery compared to pharmaceutical accumulation in the product. Average RRs were greater than ERs, and RRs for ED were shifted rightward compared to those for ECS and BPED. This difference was likely caused by lower TAN recovery achieved by ED than ECS and BPED. Differences in urine pH for ED compared to ECS and BPED could also have affected transformation and migration of contaminants, leading to different RRs. RR differences were statistically significant except for the RR calculated from target intensities for ECS and BPED (Table S2-5). Average RRs were also often less than 1, except for ED, indicating that the processes more effectively performed their intended function (i.e., TAN recovery) than a potential additional function (i.e., organic contaminant removal) and that organic compounds generally tended to be removed rather than accumulated in the treated urine. ERs and RRs for the same features detected during suspect screening did not appear to be strongly correlated (Fig. S2-8).
Average enrichment and removal ratios calculated for all suspect compounds differed from those calculated for suspects identified as target compounds with some statistically significant but perhaps not practically significant differences between calculation methods and datasets (Fig. 2B, C, Tables S2-6 and S2-7). Average ERs for target features were statistically significantly lower than those determined for all features found during suspect screening. Average RRs were also statistically significantly lower for target features than for all features, but the values overlapped more than ERs did (Table S2-6). Average ERs calculated based on target analysis were generally similar to ER calculated for features identified as target compounds based on suspect screening (Fig. S2-7A and Table S2-7). For ECS and ED, average ER for all features from suspect screening overlapped with ERs for target compounds calculated in at least one way. However, for BPED, the ER based on all features from suspect screening was greater than the values for target compounds regardless of calculation method. Average RRs showed more overlap for target compounds and the complete dataset compared to ERs (Fig. S2-7B and Table S2-7). Generally, target data showed higher variability than suspect data, which is expected because of the large difference in the number of target vs. suspect screened compounds. These differences between target and suspect data indicate that selecting additional standards could allow the fate of target compounds to more completely reflect the range of behavior of all suspect compounds.
3.3 Product purity
Product purity is a crucial consideration for implementation of electrochemical nutrient recovery processes.9–11,13–15 We evaluated product purity based on detection of three groups of species: target pharmaceuticals, target disinfection byproducts, and suspect features (Fig. 3). No target pharmaceuticals were detected in the ammonium sulfate product recovered by ECS, while a few target pharmaceuticals were detected in ED and BPED products. These results suggest that two barriers with different separation mechanisms between urine and the product – a cation exchange membrane and a hydrophobic gas permeable membrane in ECS – facilitated more effective separation of contaminants from nutrients than a single barrier – a cation exchange membrane for ED and BPED. However, the number of suspect features detected in the product did not illustrate such an advantage for ECS.Notably, no DBPs were detected in the product chambers for all processes despite formation of multiple DBPs in the anode chambers (Fig. S2-9). DBPs were detected in treated urine and the anode chambers for all processes, suggesting that DBPs might be a concern for effluent quality and overall safety (Fig. S2-10 to S2-14, Section S2.3). On the time scales of operation evaluated in this study, DBPs posed a minimal risk to product purity and safety for all processes, even at extended treatment times due to batch operation. Nevertheless, we recommend studies with longer treatment and larger volumes of urine to confirm these findings.
3.4 Identification of priority compounds
Identifying undesired compounds in recovered products is important from a product safety perspective. Identifying priority compounds based on their degree of removal or formation in treated urine could show co-removal of organics with nitrogen recovery processes and help evaluate the safety of treated effluent. We selected priority features that exhibit both (1) significant differences in enrichment or removal ratios between the process influent and effluent samples (p < 0.05 with two-sided t-test) and (2) extreme enrichment or removal of the compounds compared to nutrient recovery (ER or RR > 2, ER or RR < 0.5). Table 1 and Fig. S2-15 through S2-17 summarize the distribution of features among priority and other categories, as well as compound identification rates. For all processes, more features were detected only in the influent than only in the effluent or product, indicating that more compounds were removed than formed. For ECS and BPED, more features had ER < 0.5 than ER > 2, while ED showed the opposite relationship. Therefore, ECS and BPED may be more effective at separating nitrogen from organic contaminants. ECS facilitated more removal of organic contaminants than ED and BPED simultaneously with nitrogen recovery because more feature groups were removed to a larger extent (i.e., RR < 0.5) and fewer feature groups were up-concentrated (i.e., RR > 2). This difference is likely due to urine being in direct contact with the anode in ECS, but not in ED and BPED. Across processes, priority features accounted for small fractions of total detected features. Focusing on this relatively small set of features that show significant and large changes between influent and effluent or product yields a manageable dataset and centers analysis on the most important contaminants and potential indicator compounds. However, loosening the definition of priority compounds to include more features could broaden analysis and associated insights.| Process | ECS | ED | BPED |
|---|---|---|---|
| Feature breakdown | |||
| Features retained based on ER | 17.4 [2268] | 8.8 [1604] | 12.9 [2167] |
| Priority features based on ER | 0.42 [55] | 0.24 [44] | 1.3 [213] |
| ER < 0.5 | 0.29 [38] | 0.09 [17] | 0.83 [139] |
| ER > 2 | 0.13 [17] | 0.15 [27] | 0.44 [74] |
| Features retained based on RR | 16.9 [2199] | 6.6 [1204] | 10.9 [1838] |
| Priority features based on RR | 1.2 [150] | 0.55 [101] | 0.78 [131] |
| RR < 0.5 | 0.86 [112] | 0.04 [8] | 0.41 [69] |
| RR > 2 | 0.29 [38] | 0.51 [93] | 0.37 [62] |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| Identification rates | |||
| All features retained based on ER | 73.9 [1677] | 74.9 [1202] | 71.2 [1543] |
| Priority features based on ER | 65.9 [29] | 57.1 [16] | 82.8 [140] |
| Priority features with ER < 0.5 | 61.3 [19] | 70.6 [12] | 91.0 [122] |
| Priority features with ER > 2 | 76.9 [10] | 36.4 [4] | 51.4 [18] |
| All features retained based on RR | 74.8 [1645] | 75.2 [906] | 76.3 [1402] |
| Priority features based on RR | 74.6 [85] | 69.2 [0] | 77.9 [53] |
| Priority features with RR < 0.5 | 70.8 [68] | 62.5 [5] | 73.8 [31] |
| Priority features with RR > 2 | 94.4 [17] | 80.0 [4] | 84.6 [22] |
Identification rates were generally similar across processes and across different groups of compounds. This similarity is expected because all processes treated the same influent urine (i.e., started with the same contaminants present); in addition, similar reactor materials, separation mechanisms, and reactions are involved in the three electrochemical processes. In some cases, identification rates differed more between processes as the groups of compounds considered became smaller (e.g., ER > 2 vs. all priority features based on ER). Given that data quality should be similar across all groups of features, lower or higher identification rates for smaller groups likely resulted from less averaging of probabilities of matching to a compound when working with a smaller sample size. Some priority compounds were matched to two features (14 out of 91 features that were annotated with compounds). Typically, the features had similar ER or RR values, except for 6 features. In most cases, the features that were annotated as the same compound had the same masses and similar retention times, suggesting that the features should have been grouped throughout analysis. Additional discussion about some priority compounds based on ER is provided next, while such discussion about priority compounds based on RR is included in Section S2.4.
Of identified priority compounds (Table S2-8), no compounds had ER > 2 or ER < 0.5 in all three processes. The absence of such a group of compounds likely resulted from the ER being a complex metric dependent on multiple variables, such as the type(s) of membrane(s) separating influent urine from product solutions and pH conditions over time in the influent, intermediate, and product chambers. Nevertheless, we observed similar behavior of some compounds in two of the three processes (Tables S2-9 and S2-10). For example, features identified as linoleic acid, ricinoleic acid, and methyl ricinoleate all had ER > 2 in ED and BPED but were not identified as priority compounds in ECS. Linoleic acid and ricinoleic acid have pKa values of 4.7–4.8. At acidic pH values occurring in urine during BPED, the species would be mostly neutral and able to diffuse across the CEM to the product. The higher pH in urine during ED would lead to deprotonation of the compounds, but the neutral fraction could still diffuse across the CEM. The compounds would largely be deprotonated in the concentrate chambers for ED and BPED and in Chamber 2 of ECS, effectively trapping the compounds in these chambers. The compounds are non-volatile with Henry's constant values on the order of 10−7 atm/m3-mol, making it difficult for them to cross the GPM into the product in ECS. Features identified as carbamazepine and bakuchiol had ER < 0.5 in ED and BPED. Aromatic rings in these compounds and their largely neutral nature across a large pH range could lead to adsorption onto ion exchange membranes and reduced transport to product chambers.98,99
For ECS and BPED, Ne-Boc-L-lysine was the only identified priority compound with ER > 2. This compound has a pKa of 2.53 for the carboxyl group and likely between 9 and 10 for the amino group, putting its isoelectric point around pH 6.101,102 Therefore, at low pH occurring in urine in ECS and BPED, this compound can electromigrate across the CEM but would not do so at the higher pH observed during ED. Although the alkaline conditions in Chamber 2 of ECS would lead to an overall negative charge on the molecule, the low-molecular weight, neutral fraction in equilibrium could diffuse across the GPM. Several priority compounds with ER < 0.5 in both ECS and BPED were identified: (2E,6Z)-N-ethylnona-2,6-dienamide; benzylideneacetone; and beta-damascenone. Benzylideneacetone and beta-damascenone are both small, neutral molecules that could diffuse across CEMs and possibly GPMs. They both contain aromatic rings that could lead to adsorption onto CEMs, reducing accumulation in the product chambers. (2E,6Z)-N-Ethylnona-2,6-dienamide contains an amino group that could be protonated in acidified urine during ECS and BPED, facilitating migration across CEMs. In alkaline conditions like those in Chamber 2 of ECS, the molecule would be neutral, potentially enabling diffusion across the GPM. One priority compound, pinonic acid (pKa = 4.82), had ER > 2 for ECS and ER < 0.5 for BPED. Pinonic acid would be protonated and neutral at low pH values occurring in urine during ECS and BPED, enabling diffusion across CEMs. However, urine pH remained elevated in BPED for part of the treatment time, causing deprotonation and a negative charge that enhances retention in the urine (as also occurs in ED). For ECS and ED, 1,4-dimethyl-2,3,4,5,6,7-hexahydro-1h-1,6-methano-4-benzazonin-10-ol was the only priority compound identified with ER > 2 and bicyclo(2.2.2)octane was the only priority compound identified with ER < 0.5. Both of these molecules could diffuse across CEMs and GPMs; bicyclo(2.2.2)octane is more hydrophobic, which could increase adsorption to the organic components of CEMs and therefore reduce its ER. These priority compounds included cosmetic ingredients, flavoring agents, pharmaceuticals for specific conditions or purposes (e.g., seizures, contraception), and compounds used in pharmaceutical manufacturing (Table S2-11). With molecular properties (e.g., pKa, molecular weight, hydrophobicity) affecting compound fate, different temporal trends in pH across processes explain differences in compound fate and may guide future process design setpoints for pH during operation.
Useful indicator compounds would have very high or very low ERs or RRs (Fig. S2-18). For example, for ECS, beta-damascenone had an ER of ∼0.008 while (2S)-2-amino-6-(((tert-butoxy)carbonyl)amino)hexanoic acid had an ER of ∼5.6. Galanthamine had an RR of ∼0.02, while 1-ethylpiperidine had an RR of ∼6. These two compounds with the most extreme RRs essentially cover the entire range of RRs observed for ECS (Fig. S2-6), but identification of additional compounds could provide better indicator compounds for the variation of ERs observed (Fig. 2).
3.5 Mechanistic understanding of compound fate
Our dataset did not illustrate strong correlations between compound properties and fate, making it difficult to mechanistically understand compound fate (Fig. 4, S2-19 through S2-24 and Tables S2-12 through S2-17). Only a few correlations between ER or RR and 27 properties obtained from PubChem were statistically significant. For example, for ECS, ER was weakly negatively correlated with hydrogen bond donor count and weakly positively correlated with anion count. The presence of more hydrogen bond donors increases polarity, which could hinder molecules from crossing the hydrophobic membrane into the product. Anionic functional groups should hinder compounds from migrating across the CEM and GPM, reducing enrichment, so the observed relationship between anion count and ER indicates insufficient narrowing of priority compounds, incorrect identification of compounds, or paired transport with cations. For ECS, RR and charge were weakly negatively correlated, suggesting that more negatively charged species were removed to a greater extent. While electromigration would not contribute significantly to removal of negatively charged molecules because they generally cannot cross the CEM between Chambers 1 and 2, electrochemical transformations within Chamber 1 could explain the removal observed. Negatively charged species would remain in Chamber 1 and be attracted to the anode, increasing exposure to reactive species (e.g., HOCl). Extent of removal would likely be affected by residence time in continuous operation of the process, potentially changing how compound properties impact fate.Looking at priority compounds for ED and BPED, a few statistically significant correlations between fate and property were identified. For ED, ER positively correlated with log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P, bond stereo count, and defined bond stereo count. More lipophilic molecules (i.e., higher log
P, bond stereo count, and defined bond stereo count. More lipophilic molecules (i.e., higher log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P) could cross the CEM from the urine to concentrate by interacting with the CEM's polymer matrix. The association between ER and the presence of stereocenters is difficult to explain but the correlation is weak. No significant correlations between RR and compound properties were identified for ED. For BPED, we observed negative correlations between ER and several properties: exact mass, complexity, heavy atom count, volume, Z steric quadrupole, feature count, and feature ring count. Larger molecules (e.g., higher mass, higher heavy atom count (total number of non-hydrogen atoms), greater complexity, higher volume, more features and feature rings, higher Z steric quadrupole (analogous to molecule height)) less readily pass through the CEM separating urine and concentrate, reducing their accumulation in the product. We observed a positive correlation between RR and atom stereo count and feature count for BPED. The presence of more stereocenters could reduce conformational flexibility, making it more difficult for molecules to pass through the CEM. A higher feature count (i.e., higher total number of hydrogen bond acceptors, hydrogen bond donors, anions, cations, hydrophobes, and rings) could indicate larger molecules that pass less easily through the CEM. Differences in correlations between compound fate and properties for the three processes indicate that differences in reactor architecture and solution conditions affect compound fate.
P) could cross the CEM from the urine to concentrate by interacting with the CEM's polymer matrix. The association between ER and the presence of stereocenters is difficult to explain but the correlation is weak. No significant correlations between RR and compound properties were identified for ED. For BPED, we observed negative correlations between ER and several properties: exact mass, complexity, heavy atom count, volume, Z steric quadrupole, feature count, and feature ring count. Larger molecules (e.g., higher mass, higher heavy atom count (total number of non-hydrogen atoms), greater complexity, higher volume, more features and feature rings, higher Z steric quadrupole (analogous to molecule height)) less readily pass through the CEM separating urine and concentrate, reducing their accumulation in the product. We observed a positive correlation between RR and atom stereo count and feature count for BPED. The presence of more stereocenters could reduce conformational flexibility, making it more difficult for molecules to pass through the CEM. A higher feature count (i.e., higher total number of hydrogen bond acceptors, hydrogen bond donors, anions, cations, hydrophobes, and rings) could indicate larger molecules that pass less easily through the CEM. Differences in correlations between compound fate and properties for the three processes indicate that differences in reactor architecture and solution conditions affect compound fate.
Extending the compounds considered in the correlation analysis often increased the number of statistically significant correlations observed but correlations remained weak (Fig. S2-21 through S2-24 and Tables S2-14 through S2-17). For example, when considering all identified compounds for ECS, no significant correlations between ER and compound properties were observed, but RR was weakly positively correlated with topological polar surface area (TPSA), hydrogen bond donor count, hydrogen bond acceptor count, and feature count. There was no overlap between significant correlations for priority compounds and all compounds. Considering all compounds that showed significant change in intensity between influent and effluent or product (p < 0.05), results were somewhat similar to those for all compounds though with fewer significant correlations. No significant correlations were observed for ER, but RR was weakly positively correlated with TPSA and hydrogen bond acceptor count. As for ECS, expanding the dataset beyond priority compounds for ED and BPED did not reveal any correlations that were significant in all three cases. Increasing confidence in compound identification could improve mechanistic understanding of compound fate.
4 Conclusion
This study compared the fate of pharmaceuticals and disinfection byproducts across three electrochemical nitrogen recovery processes: electrochemical stripping, electrodialysis, and bipolar electrodialysis. ECS achieved greater TAN recovery efficiencies than ED and BPED and was the only process that recovered an ammonium sulfate product rather than a mixed concentrate, containing TAN, sodium, and potassium extracted from urine. Products from ED and BPED would require additional separation (e.g., sodium removal) to improve product efficacy (e.g., as a fertilizer) and prevent negative side effects of application (e.g., increasing soil salinity). Target pharmaceuticals and organic contaminants detected during suspect screening were removed and/or transported to product chambers to varying extents. Enrichment ratios were generally lower for ECS than ED and BPED, suggesting that having two barriers between influent urine and the product chamber (i.e., CEM and GPM) more effectively prevented organic contaminants from reaching the product than a single barrier (i.e., CEM). Removal ratios were generally higher for ED than ECS and BPED due to lower TAN recovery in ED and the impact of pH on compound transformation and migration. The products recovered by all processes had little to no contamination with DBPs, suggesting that the membranes in the reactors were effective barriers to the transfer of these contaminants. However, DBP formation in other reactor chambers was a significant concern, especially in the anode chambers for ECS and ED.Our study highlighted the importance of selecting broader sets of target compounds to be more representative of the range of behaviors observed for organic contaminants. Through suspect screening, we tentatively identified other compounds with very high or low ERs or RRs, which could be useful indicator compounds. Identification of additional compounds could provide better indicator compounds. Our data did not show strong correlations between compound fate and properties. Therefore, future work utilizing suspect screening is needed to facilitate mechanistic understanding of compound fate and characterization of the potential toxicity of treated urine, recovered products, and other process solutions. Such investigations will enable mitigation of potential negative side effects (e.g., integrating BPMs to reduce overall DBP formation) and build confidence in the safety of products recovered from urine. These advances will support deployment of electrochemical nitrogen recovery from urine, which could replace about one-quarter of current nitrogen fertilizers worldwide,103 enabling a circular nitrogen economy.
Author contributions
Anna Kogler: conceptualization, data curation, formal analysis, investigation, methodology, resources, software, validation, visualization, writing – original draft, writing – review & editing. William A. Tarpeh: conceptualization, funding acquisition, methodology, validation, resources, writing – review & editing.Conflicts of interest
The authors have no conflicts of interest to declare.Data availability
Data for this study are available in an online data repository (https://doi.org/10.25740/ps371sj4433). Ref. 104–143 are cited in the supplementary information (SI) file. Supplementary information: details on all experimental and analytical procedures, as well as tables and figures presenting additional data on enrichment and removal ratios, disinfection byproduct species detected, priority compound identities and properties, and results of statistical analyses. See DOI: https://doi.org/10.1039/d5va00496a.Acknowledgements
We thank the Tarpeh lab for productive discussions in developing this work and all the urine donors (affectionately and colloquially known as “pee-ple”) for enabling this research. We thank Bill Mitch, Jessica McDonald, and Stephanie Lau for training related to target analysis methods and for instrument use. We thank Theresa McLaughlin for conducting LC-HRMS for suspect screening. This work utilized the Thermo Exploris 240 LC/MS system (RRID: SCR_022216) that was purchased with funding from Stanford C-ShaRP (RRID: SCR_022986) and was supported by Stanford University Mass Spectrometry (RRID: SCR_017801). The project was funded by the Stanford University King Center on Global Development, Stanford University Center for Innovation in Global Health, and the Sustainability Accelerator from the Stanford Doerr School of Sustainability. Authors also acknowledge the following additional support: AK – Stanford University Vice Provost for Graduate Education (Lieberman Fellowship) and WAT – Camille and Henry Dreyfus Foundation (Dreyfus Teacher-Scholar Award).References
- D. J. Sobota, J. E. Compton, M. L. McCrackin and S. Singh, Cost of reactive nitrogen release from human activities to the environment in the United States, Environ. Res. Lett., 2015, 10, 025006 CrossRef.
- United States Environmental Protection Agency, Biological Nutrient Removal Processes and Costs, EPA-823-R-07-002, 2007 Search PubMed.
- P. L. McCarty, J. Bae and J. Kim, Domestic Wastewater Treatment as a Net Energy Producer–Can This be Achieved?, Environ. Sci. Technol., 2011, 45, 7100–7106 CrossRef CAS PubMed.
- I. Chorkendorff and J. W. Niemantsverdriet, Concepts of Modern Catalysis and Kinetics, Wiley, 1st edn, 2003 Search PubMed.
- J. W. Erisman, M. A. Sutton, J. Galloway, Z. Klimont and W. Winiwarter, How a century of ammonia synthesis changed the world, Nat. Geosci., 2008, 1, 636–639 CrossRef CAS.
- V. Kyriakou, I. Garagounis, A. Vourros, E. Vasileiou and M. Stoukides, An Electrochemical Haber-Bosch Process, Joule, 2020, 4, 142–158 CrossRef CAS.
- H. Jönsson, A. Baky, U. Jeppsson, D. Hellström and E. Kärrman, Composition of Urine, Faeces, Greywater and Biowaste, 2005, vol. 49 Search PubMed.
- T. A. Larsen, A. C. Alder, R. I. L. Eggen, M. Maurer and J. Lienert, Source Separation: Will We See a Paradigm Shift in Wastewater Handling?, Environ. Sci. Technol., 2009, 43, 6121–6125 CrossRef CAS PubMed.
- J. Lienert, M. Haller, A. Berner, M. Stauffacher and T. A. Larsen, How farmers in Switzerland perceive fertilizers from recycled anthropogenic nutrients (urine), Water Sci. Technol., 2003, 48, 47–56 CrossRef CAS PubMed.
- C. Pahl-Wostl, A. Schönborn, N. Willi, J. Muncke and T. A. Larsen, Investigating consumer attitudes towards the new technology of urine separation, Water Sci. Technol., 2003, 48, 57–65 CrossRef CAS PubMed.
- T. A. Larsen, H. Gruendl and C. Binz, The potential contribution of urine source separation to the SDG agenda – a review of the progress so far and future development options, Environ. Sci.: Water Res. Technol., 2021, 7, 1161–1176 RSC.
- A. Segrè Cohen, N. G. Love, K. K. Nace and J. Árvai, Consumers' Acceptance of Agricultural Fertilizers Derived from Diverted and Recycled Human Urine, Environ. Sci. Technol., 2020, 54, 5297–5305 CrossRef PubMed.
- T. Schreiber, S. Opperman, R. Hardin, J. Cavicchi, A. Pallmeyer, K. Nace and N. Love, Nested risks and responsibilities: Perspectives on fertilizer from human urine in two U.S. regions, J. Agric. Food Syst. Community Dev., 2021, 10, 1–22 Search PubMed.
- J. Lienert and T. A. Larsen, High Acceptance of Urine Source Separation in Seven European Countries: A Review, Environ. Sci. Technol., 2010, 44, 556–566 CrossRef CAS PubMed.
- P. Simha, M. A. Barton, L. F. Perez-Mercado, J. R. McConville, C. Lalander, M. E. Magri, S. Dutta, H. Kabir, A. Selvakumar, X. Zhou, T. Martin, T. Kizos, R. Kataki, Y. Gerchman, R. Herscu-Kluska, D. Alrousan, E. G. Goh, D. Elenciuc, A. Głowacka, L. Korculanin, R. V. Tzeng, S. S. Ray, C. Niwagaba, C. Prouty, J. R. Mihelcic and B. Vinnerås, Willingness among food consumers to recycle human urine as crop fertiliser: Evidence from a multinational survey, Sci. Total Environ., 2021, 765, 144438 CrossRef CAS PubMed.
- P. Kuntke, M. Rodríguez Arredondo, L. Widyakristi, A. ter Heijne, T. H. J. A. Sleutels, H. V. M. Hamelers and C. J. N. Buisman, Hydrogen Gas Recycling for Energy Efficient Ammonia Recovery in Electrochemical Systems, Environ. Sci. Technol., 2017, 51, 3110–3116 CrossRef CAS PubMed.
- P. Zamora, T. Georgieva, A. Ter Heijne, T. H. J. A. Sleutels, A. W. Jeremiasse, M. Saakes, C. J. N. Buisman and P. Kuntke, Ammonia recovery from urine in a scaled-up Microbial Electrolysis Cell, J. Power Sources, 2017, 356, 491–499 CrossRef CAS.
- P. Kuntke, K. M. Śmiech, H. Bruning, G. Zeeman, M. Saakes, T. H. J. A. Sleutels, H. V. M. Hamelers and C. J. N. Buisman, Ammonium recovery and energy production from urine by a microbial fuel cell, Water Res., 2012, 46, 2627–2636 CrossRef CAS PubMed.
- W. A. Tarpeh, J. M. Barazesh, T. Y. Cath and K. L. Nelson, Electrochemical Stripping to Recover Nitrogen from Source-Separated Urine, Environ. Sci. Technol., 2018, 52, 1453–1460 CrossRef CAS PubMed.
- D. Appling, M. Habteselassie, D. Radcliffe and J. Bradshaw, Preliminary Study on the Effect of Wastewater Storage in Septic Tank on E. coli Concentration in Summer, Water, 2013, 5, 1141–1151 CrossRef CAS.
- H. Dong, L. Wei and W. A. Tarpeh, Electro-assisted regeneration of pH-sensitive ion exchangers for sustainable phosphate removal and recovery, Water Res., 2020, 184, 116167 CrossRef CAS PubMed.
- Codiga Resource Recovery Center, CR2C Treatment Monitoring Dashboard, https://cr2c-monitoring.appspot.com/, accessed 8 March 2021.
- T. A. Larsen, M. E. Riechmann and K. M. Udert, State of the art of urine treatment technologies: A critical review, Water Res.: X, 2021, 13, 100114 CAS.
- B. P. Chaplin, The Prospect of Electrochemical Technologies Advancing Worldwide Water Treatment, Acc. Chem. Res., 2019, 52, 596–604 CrossRef CAS PubMed.
- J. Radjenovic and D. L. Sedlak, Challenges and Opportunities for Electrochemical Processes as Next-Generation Technologies for the Treatment of Contaminated Water, Environ. Sci. Technol., 2015, 49, 11292–11302 CrossRef CAS PubMed.
- J. Lienert, T. Bürki and B. I. Escher, Reducing micropollutants with source control: substance flow analysis of 212 pharmaceuticals in faeces and urine, Water Sci. Technol., 2007, 56, 87–96 CrossRef CAS PubMed.
- T. Heberer, Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: a review of recent research data, Toxicol. Lett., 2002, 131, 5–17 CrossRef CAS PubMed.
- O. González, B. Bayarri, J. Aceña, S. Pérez and D. Barceló, Treatment Technologies for Wastewater Reuse: Fate of Contaminants of Emerging Concern, Handb. Environ. Chem., 2016, 45, 5–37 Search PubMed.
- D. W. Kolpin, E. T. Furlong, M. T. Meyer, E. M. Thurman, S. D. Zaugg, L. B. Barber and H. T. Buxton, Pharmaceuticals, Hormones, and Other Organic Wastewater Contaminants in U.S. Streams, 1999–2000: A National Reconnaissance, Environ. Sci. Technol., 2002, 36, 1202–1211 CrossRef CAS PubMed.
- T. R. Romeyn, W. Harijanto, S. Sandoval, S. Delagah and M. Sharbatmaleki, Contaminants of emerging concern in reverse osmosis brine concentrate from indirect/direct water reuse applications, Water Sci. Technol., 2016, 73, 236–250 CrossRef CAS PubMed.
- S. H. Joo and B. Tansel, Novel technologies for reverse osmosis concentrate treatment: A review, J. Environ. Manage., 2015, 150, 322–335 CrossRef CAS PubMed.
- I. Köpping, C. S. McArdell, E. Borowska, M. A. Böhler and K. M. Udert, Removal of pharmaceuticals from nitrified urine by adsorption on granular activated carbon, Water Res.: X, 2020, 9, 100057 Search PubMed.
- W. Pronk, H. Palmquist, M. Biebow and M. Boller, Nanofiltration for the separation of pharmaceuticals from nutrients in source-separated urine, Water Res., 2006, 40, 1405–1412 CrossRef CAS PubMed.
- J. A. Clark, Y. Yang, N. C. Ramos and H. W. Hillhouse, Selective oxidation of pharmaceuticals and suppression of perchlorate formation during electrolysis of fresh human urine, Water Res., 2021, 198, 117106 CrossRef CAS PubMed.
- N. Demissie, P. Simha, F. Y. Lai, L. Ahrens, D. Mussabek, A. Desta and B. Vinnerås, Degradation of 75 organic micropollutants in fresh human urine and water by UV advanced oxidation process, Water Res., 2023, 242, 120221 CrossRef CAS PubMed.
- A. Recherche, F. Tettenborn, J. Behrendt and R. Otterpohl, Resource Recovery and Removal of Pharmaceutical Residues Treatment of Separate Collected Urine, Institute of Wastewater Management and Water Protection Hamburg University of Technology, 2007 Search PubMed.
- W. A. Tarpeh, I. Wald, M. Wiprächtiger and K. L. Nelson, Effects of operating and design parameters on ion exchange columns for nutrient recovery from urine, Environ. Sci.: Water Res. Technol., 2018, 4, 828–838 RSC.
- B. Schürmann, W. Everding, D. Montag and J. Pinnekamp, Fate of pharmaceuticals and bacteria in stored urine during precipitation and drying of struvite, Water Sci. Technol., 2012, 65, 1774–1780 CrossRef PubMed.
- B. I. Escher, W. Pronk, M. J.-F. Suter and M. Maurer, Monitoring the Removal Efficiency of Pharmaceuticals and Hormones in Different Treatment Processes of Source-Separated Urine with Bioassays, Environ. Sci. Technol., 2006, 40, 5095–5101 CrossRef CAS PubMed.
- Z. Pan, C. Song, L. Li, H. Wang, Y. Pan, C. Wang, J. Li, T. Wang and X. Feng, Membrane technology coupled with electrochemical advanced oxidation processes for organic wastewater treatment: Recent advances and future prospects, Chem. Eng. J., 2019, 376, 120909 CrossRef CAS.
- E. Mordačíková, M. Vojs, K. Grabicová, M. Marton, P. Michniak, V. Řeháček, A. Bořík, R. Grabic, J. Bruncko, T. Mackuľak and A. Vojs Staňová, Influence of boron doped diamond electrodes properties on the elimination of selected pharmaceuticals from wastewater, J. Electroanal. Chem., 2020, 862, 114007 CrossRef.
- C. Heim, M. Rajab, G. Greco, S. Grosse, J. E. Drewes, T. Letzel and B. Helmreich, Fate of Diclofenac and Its Transformation and Inorganic By-Products in Different Water Matrices during Electrochemical Advanced Oxidation Process Using a Boron-Doped Diamond Electrode, Water, 2020, 12, 1686 CrossRef CAS.
- J. T. Jasper, O. S. Shafaat and M. R. Hoffmann, Electrochemical Transformation of Trace Organic Contaminants in Latrine Wastewater, Environ. Sci. Technol., 2016, 50, 10198–10208 CrossRef CAS PubMed.
- S. Garcia-Segura, J. Keller, E. Brillas and J. Radjenovic, Removal of organic contaminants from secondary effluent by anodic oxidation with a boron-doped diamond anode as tertiary treatment, J. Hazard. Mater., 2015, 283, 551–557 CrossRef CAS PubMed.
- A. Farhat, J. Keller, S. Tait and J. Radjenovic, Removal of Persistent Organic Contaminants by Electrochemically Activated Sulfate, Environ. Sci. Technol., 2015, 49, 14326–14333 CrossRef CAS PubMed.
- Y. Chen, S. Li and J. Hu, Photoelectrocatalytic degradation of organics and formation of disinfection byproducts in reverse osmosis concentrate, Water Res., 2020, 168, 115105 CrossRef CAS PubMed.
- H. Lu, W. Zou, P. Chai, J. Wang and L. Bazinet, Feasibility of antibiotic and sulfate ions separation from wastewater using electrodialysis with ultrafiltration membrane, J. Clean. Prod., 2016, 112, 3097–3105 CrossRef CAS.
- A. R. Ferreira, N. Couto, P. Guedes, J. Pinto, E. P. Mateus and A. B. Ribeiro, Electrodialytic 2-compartment cells for emerging organic contaminants removal from effluent, J. Hazard. Mater., 2018, 358, 467–474 CrossRef CAS PubMed.
- Z. Li, R. Dai, B. Yang, M. Chen, X. Wang and Z. Wang, An electrochemical membrane biofilm reactor for removing sulfonamides from wastewater and suppressing antibiotic resistance development: Performance and mechanisms, J. Hazard. Mater., 2021, 404, 124198 CrossRef CAS PubMed.
- P. Guedes, E. P. Mateus, J. Almeida, A. R. Ferreira, N. Couto and A. B. Ribeiro, Electrodialytic treatment of sewage sludge: Current intensity influence on phosphorus recovery and organic contaminants removal, Chem. Eng. J., 2016, 306, 1058–1066 CrossRef CAS.
- L. J. Banasiak and A. I. Schäfer, Sorption of steroidal hormones by electrodialysis membranes, J. Membr. Sci., 2010, 365, 198–205 CrossRef CAS.
- K. Arola, A. Ward, M. Mänttäri, M. Kallioinen and D. Batstone, Transport of pharmaceuticals during electrodialysis treatment of wastewater, Water Res., 2019, 161, 496–504 CrossRef CAS PubMed.
- K. Noguera-Oviedo and D. S. Aga, Lessons learned from more than two decades of research on emerging contaminants in the environment, J. Hazard. Mater., 2016, 316, 242–251 CrossRef CAS PubMed.
- K. E. Manz, A. Feerick, J. M. Braun, Y.-L. Feng, A. Hall, J. Koelmel, C. Manzano, S. R. Newton, K. D. Pennell, B. J. Place, K. J. Godri Pollitt, C. Prasse and J. A. Young, Non-targeted analysis (NTA) and suspect screening analysis (SSA): a review of examining the chemical exposome, J. Expo. Sci. Environ. Epidemiol., 2023, 33, 524–536 CrossRef CAS PubMed.
- D. E. Helbling, J. Hollender, H.-P. E. Kohler, H. Singer and K. Fenner, High-Throughput Identification of Microbial Transformation Products of Organic Micropollutants, Environ. Sci. Technol., 2010, 44, 6621–6627 CrossRef CAS PubMed.
- M. Krauss, H. Singer and J. Hollender, LC–high resolution MS in environmental analysis: from target screening to the identification of unknowns, Anal. Bioanal. Chem., 2010, 397, 943–951 CrossRef CAS PubMed.
- A. Agüera, P. Plaza-Bolaños and F. G. A. Fernández, in Current Developments in Biotechnology and Bioengineering, ed. S. Varjani, A. Pandey, R. D. Tyagi, H. H. Ngo and C. Larroche, Elsevier, 2020, pp. 503–525 Search PubMed.
- T. Bader, W. Schulz, T. Lucke, W. Seitz and R. Winzenbacher, in ACS Symposium Series, ed. J. E. Drewes and T. Letzel, American Chemical Society, Washington, DC, 2016, vol. 1242, pp. 49–70 Search PubMed.
- A. Lai, R. R. Singh, L. Kovalova, O. Jaeggi, T. Kondic and E. L. Schymanski, Retrospective Non-target Analysis to Support Regulatory Water Monitoring: From Masses of Interest to Recommendations via in silico workflows, Environ. Sci. Eur., 2021, 33, 43 CrossRef CAS.
- W. Brack, J. Hollender, M. L. de Alda, C. Müller, T. Schulze, E. Schymanski, J. Slobodnik and M. Krauss, High-resolution mass spectrometry to complement monitoring and track emerging chemicals and pollution trends in European water resources, Environ. Sci. Eur., 2019, 31, 62 CrossRef.
- A. López, P. Dualde, V. Yusà and C. Coscollà, Retrospective analysis of pesticide metabolites in urine using liquid chromatography coupled to high-resolution mass spectrometry, Talanta, 2016, 160, 547–555 CrossRef PubMed.
- B. González-Gaya, N. Lopez-Herguedas, D. Bilbao, L. Mijangos, A. M. Iker, N. Etxebarria, M. Irazola, A. Prieto, M. Olivares and O. Zuloaga, Suspect and non-target screening: the last frontier in environmental analysis, Anal. Methods, 2021, 13, 1876–1904 RSC.
- S. Murgolo, S. Franz, H. Arab, M. Bestetti, E. Falletta and G. Mascolo, Degradation of emerging organic pollutants in wastewater effluents by electrochemical photocatalysis on nanostructured TiO2 meshes, Water Res., 2019, 164, 114920 CrossRef CAS PubMed.
- M. Rajab, G. Greco, C. Heim, B. Helmreich and T. Letzel, Serial coupling of RP and zwitterionic hydrophilic interaction LC–MS: Suspects screening of diclofenac transformation products by oxidation with a boron-doped diamond electrode, J. Sep. Sci., 2013, 36, 3011–3018 CrossRef CAS PubMed.
- Rich Earth Institute, Rich Earth Institute: Regulation, https://richearthinstitute.org/our-work/regulation/, accessed 11 May 2022.
- B. P. Chaplin, Critical review of electrochemical advanced oxidation processes for water treatment applications, Environ. Sci.: Processes Impacts, 2014, 16, 1182–1203 RSC.
- S. Garcia-Segura, A. B. Nienhauser, A. S. Fajardo, R. Bansal, C. L. Coonrod, J. D. Fortner, M. Marcos-Hernández, T. Rogers, D. Villagran, M. S. Wong and P. Westerhoff, Disparities between experimental and environmental conditions: Research steps toward making electrochemical water treatment a reality, Curr. Opin. Electrochem., 2020, 22, 9–16 CrossRef CAS.
- A. Kogler, M. Farmer, J. A. Simon, S. Tilmans, G. F. Wells and W. A. Tarpeh, Systematic Evaluation of Emerging Wastewater Nutrient Removal and Recovery Technologies to Inform Practice and Advance Resource Efficiency, ACS ES&T Eng., 2021, 1, 662–684 Search PubMed.
- S. D. Richardson, M. J. Plewa, E. D. Wagner, R. Schoeny and D. M. DeMarini, Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: A review and roadmap for research, Mutat. Res., Rev. Mutat. Res., 2007, 636, 178–242 CrossRef CAS PubMed.
- E. D. Wagner, K.-M. Hsu, A. Lagunas, W. A. Mitch and M. J. Plewa, Comparative genotoxicity of nitrosamine drinking water disinfection byproducts in Salmonella and mammalian cells, Mutat. Res., 2012, 741, 109–115 CAS.
- E. D. Wagner and M. J. Plewa, CHO cell cytotoxicity and genotoxicity analyses of disinfection by-products: An updated review, J. Environ. Sci., 2017, 58, 64–76 CrossRef CAS PubMed.
- M. Diana, M. Felipe-Sotelo and T. Bond, Disinfection byproducts potentially responsible for the association between chlorinated drinking water and bladder cancer: A review, Water Res., 2019, 162, 492–504 CrossRef CAS PubMed.
- C. M. Villanueva, S. Cordier, L. Font-Ribera, L. A. Salas and P. Levallois, Overview of Disinfection By-products and Associated Health Effects, Curr. Environ. Health Rep., 2015, 2, 107–115 CrossRef CAS PubMed.
- S. D. Richardson and M. J. Plewa, To regulate or not to regulate? What to do with more toxic disinfection by-products?, J. Environ. Chem. Eng., 2020, 8, 103939 CrossRef CAS.
- X.-F. Li and W. A. Mitch, Drinking Water Disinfection Byproducts (DBPs) and Human Health Effects: Multidisciplinary Challenges and Opportunities, Environ. Sci. Technol., 2018, 52, 1681–1689 CrossRef CAS PubMed.
- Y. Chen, W. Chen, H. Huang, H. Zeng, L. Tan, Y. Pang, J. Ghani and S. Qi, Occurrence of N-nitrosamines and their precursors in the middle and lower reaches of Yangtze River water, Environ. Res., 2021, 195, 110673 CrossRef CAS PubMed.
- Z. Li, G. Song, Y. Bi, W. Gao, A. He, Y. Lu, Y. Wang and G. Jiang, Occurrence and Distribution of Disinfection Byproducts in Domestic Wastewater Effluent, Tap Water, and Surface Water during the SARS-CoV-2 Pandemic in China, Environ. Sci. Technol., 2021, 55, 4103–4114 CrossRef CAS PubMed.
- X. Luan, X. Liu, C. Fang, W. Chu and Z. Xu, Ecotoxicological effects of disinfected wastewater effluents: a short review of in vivo toxicity bioassays on aquatic organisms, Environ. Sci.: Water Res. Technol., 2020, 6, 2275–2286 RSC.
- R. Xiao, T. Ou, S. Ding, C. Fang, Z. Xu and W. Chu, Disinfection by-products as environmental contaminants of emerging concern: a review on their occurrence, fate and removal in the urban water cycle, Crit. Rev. Environ. Sci. Technol., 2022, 1–28 Search PubMed.
- K. Watson, G. Shaw, F. D. L. Leusch and N. L. Knight, Chlorine disinfection by-products in wastewater effluent: Bioassay-based assessment of toxicological impact, Water Res., 2012, 46, 6069–6083 CrossRef CAS PubMed.
- H. Cui, B. Chen, Y. Jiang, Y. Tao, X. Zhu and Z. Cai, Toxicity of 17 Disinfection By-products to Different Trophic Levels of Aquatic Organisms: Ecological Risks and Mechanisms, Environ. Sci. Technol., 2021, 55, 10534–10541 CrossRef CAS PubMed.
- J. Liu and X. Zhang, Comparative toxicity of new halophenolic DBPs in chlorinated saline wastewater effluents against a marine alga: Halophenolic DBPs are generally more toxic than haloaliphatic ones, Water Res., 2014, 65, 64–72 CrossRef CAS PubMed.
- M. Yang and X. Zhang, Comparative Developmental Toxicity of New Aromatic Halogenated DBPs in a Chlorinated Saline Sewage Effluent to the Marine Polychaete Platynereis dumerilii, Environ. Sci. Technol., 2013, 47, 10868–10876 CrossRef CAS PubMed.
- A. Fumasoli, B. Etter, B. Sterkele, E. Morgenroth and K. M. Udert, Operating a pilot-scale nitrification/distillation plant for complete nutrient recovery from urine, Water Sci. Technol., 2016, 73, 215–222 CrossRef CAS PubMed.
- J. M. Barazesh, C. Prasse and D. L. Sedlak, Electrochemical Transformation of Trace Organic Contaminants in the Presence of Halide and Carbonate Ions, Environ. Sci. Technol., 2016, 50, 10143–10152 CrossRef CAS PubMed.
- M. Rodríguez Arredondo, P. Kuntke, A. ter Heijne, H. V. M. Hamelers and C. J. N. Buisman, Load ratio determines the ammonia recovery and energy input of an electrochemical system, Water Res., 2017, 111, 330–337 CrossRef PubMed.
- E. Lacasa, S. Cotillas, C. Saez, J. Lobato, P. Cañizares and M. A. Rodrigo, Environmental applications of electrochemical technology. What is needed to enable full-scale applications?, Curr. Opin. Electrochem., 2019, 16, 149–156 CrossRef CAS.
- M. Rodrigues, T. T. de Mattos, T. Sleutels, A. ter Heijne, H. V. M. Hamelers, C. J. N. Buisman and P. Kuntke, Minimal Bipolar Membrane Cell Configuration for Scaling Up Ammonium Recovery, ACS Sustainable Chem. Eng., 2020, 8, 17359–17367 CrossRef CAS PubMed.
- M. A. Blommaert, D. Aili, R. A. Tufa, Q. Li, W. A. Smith and D. A. Vermaas, Insights and Challenges for Applying Bipolar Membranes in Advanced Electrochemical Energy Systems, ACS Energy Lett., 2021, 6, 2539–2548 CrossRef CAS PubMed.
- W. A. Tarpeh, Y. Du, C. M. G. Carpenter, E. E. Rodriguez, D. E. Helbling, D. S. Aga, N. G. Love and K. R. Wigginton, A Unit Process Approach to Nontarget Screening of Organic Contaminants during Urine Treatment, ACS ES&T Eng., 2023, 3, 590–601 Search PubMed.
- R. Helmus, T. L. ter Laak, A. P. van Wezel, P. de Voogt and E. L. Schymanski, patRoon: open source software platform for environmental mass spectrometry based non-target screening, J. Cheminf., 2021, 13, 1 CAS.
- R. Helmus, B. van de Velde, A. M. Brunner, T. L. ter Laak, A. P. van Wezel and E. L. Schymanski, patRoon 2.0: Improved non-target analysis workflows including automated transformation product screening, J. Open Source Softw., 2022, 7, 4029 CrossRef.
- A. L. Pochodylo and D. E. Helbling, Emerging investigators series: prioritization of suspect hits in a sensitive suspect screening workflow for comprehensive micropollutant characterization in environmental samples, Environ. Sci.: Water Res. Technol., 2017, 3, 54–65 RSC.
- E. Parry and T. M. Young, Comparing targeted and non-targeted high-resolution mass spectrometric approaches for assessing advanced oxidation reactor performance, Water Res., 2016, 104, 72–81 CrossRef CAS PubMed.
- J. E. Schollée, E. L. Schymanski and J. Hollender, in ACS Symposium Series, ed. J. E. Drewes and T. Letzel, American Chemical Society, Washington, DC, 2016, vol. 1241, pp. 45–65 Search PubMed.
- K. E. Furst, R. M. Coyte, M. Wood, A. Vengosh and W. A. Mitch, Disinfection Byproducts in Rajasthan, India: Are Trihalomethanes a Sufficient Indicator of Disinfection Byproduct Exposure in Low-Income Countries?, Environ. Sci. Technol., 2019, 53, 12007–12017 CrossRef CAS PubMed.
- Y.-H. Chuang, D. L. McCurry, H. Tung and W. A. Mitch, Formation Pathways and Trade-Offs between Haloacetamides and Haloacetaldehydes during Combined Chlorination and Chloramination of Lignin Phenols and Natural Waters, Environ. Sci. Technol., 2015, 49, 14432–14440 CrossRef CAS PubMed.
- M. Roman, L. H. Van Dijk, L. Gutierrez, M. Vanoppen, J. W. Post, B. A. Wols, E. R. Cornelissen and A. R. D. Verliefde, Key physicochemical characteristics governing organic micropollutant adsorption and transport in ion-exchange membranes during reverse electrodialysis, Desalination, 2019, 468, 114084 CrossRef CAS.
- M. Roman, L. Gutierrez, L. H. Van Dijk, M. Vanoppen, J. W. Post, B. A. Wols, E. R. Cornelissen and A. R. D. Verliefde, Effect of pH on the transport and adsorption of organic micropollutants in ion-exchange membranes in electrodialysis-based desalination, Sep. Purif. Technol., 2020, 252, 117487 CrossRef CAS.
- J. D. García-Espinoza, P. Mijaylova-Nacheva and M. Avilés-Flores, Electrochemical carbamazepine degradation: Effect of the generated active chlorine, transformation pathways and toxicity, Chemosphere, 2018, 192, 142–151 CrossRef PubMed.
- Ne-Boc-L-Lysine, https://www.chembk.com/en/chem/Ne-Boc-L-Lysine, accessed 27 November 2025.
- Amino Acids, https://www.vanderbilt.edu/AnS/Chemistry/Rizzo/stuff/AA/AminoAcids.html, accessed 27 November 2025.
- C. Wald, The urine revolution: how recycling pee could help to save the world, Nature, 2022, 602, 202–206 CrossRef CAS PubMed.
- N. Moore, S. Ebrahimi, Y. Zhu, C. Wang, R. Hofmann and S. Andrews, A comparison of sodium sulfite, ammonium chloride, and ascorbic acid for quenching chlorine prior to disinfection byproduct analysis, Water Supply, 2021, ws2021059 Search PubMed.
- M. J. Liu, B. S. Neo and W. A. Tarpeh, Building an operational framework for selective nitrogen recovery via electrochemical stripping, Water Res., 2020, 169, 115226 CrossRef CAS PubMed.
- W. Pronk, M. Biebow and M. Boller, Electrodialysis for Recovering Salts from a Urine Solution Containing Micropollutants, Environ. Sci. Technol., 2006, 40, 2414–2420 CrossRef CAS PubMed.
- W. Pronk, S. Zuleeg, J. Lienert, B. Escher, M. Koller, A. Berner, G. Koch and M. Boller, Pilot experiments with electrodialysis and ozonation for the production of a fertiliser from urine, Water Sci. Technol., 2007, 56, 219–227 CrossRef CAS PubMed.
- L. Gurreri, A. Tamburini, A. Cipollina and G. Micale, Electrodialysis Applications in Wastewater Treatment for Environmental Protection and Resources Recovery: A Systematic Review on Progress and Perspectives, Membranes, 2020, 10, 146 CrossRef CAS PubMed.
- J. De Paepe, R. E. F. Lindeboom, M. Vanoppen, K. De Paepe, D. Demey, W. Coessens, B. Lamaze, A. R. D. Verliefde, P. Clauwaert and S. E. Vlaeminck, Refinery and concentration of nutrients from urine with electrodialysis enabled by upstream precipitation and nitrification, Water Res., 2018, 144, 76–86 CrossRef CAS PubMed.
- Y.-K. Wang, Y.-K. Geng, X.-R. Pan and G.-P. Sheng, In situ utilization of generated electricity for nutrient recovery in urine treatment using a selective electrodialysis membrane bioreactor, Chem. Eng. Sci., 2017, 171, 451–458 CrossRef CAS.
- F. Gao, L. Wang, J. Wang, H. Zhang and S. Lin, Nutrient recovery from treated wastewater by a hybrid electrochemical sequence integrating bipolar membrane electrodialysis and membrane capacitive deionization, Environ. Sci.: Water Res. Technol., 2020, 6, 383–391 RSC.
- N. van Linden, G. L. Bandinu, D. A. Vermaas, H. Spanjers and J. B. van Lier, Bipolar membrane electrodialysis for energetically competitive ammonium removal and dissolved ammonia production, J. Clean. Prod., 2020, 259, 120788 CrossRef CAS.
- X. Wang, Y. Wang, X. Zhang, H. Feng, C. Li and T. Xu, Phosphate Recovery from Excess Sludge by Conventional Electrodialysis (CED) and Electrodialysis with Bipolar Membranes (EDBM), Ind. Eng. Chem. Res., 2013, 52, 15896–15904 CrossRef CAS.
- W. Pronk, M. Biebow and M. Boller, Treatment of source-separated urine by a combination of bipolar electrodialysis and a gas transfer membrane, Water Sci. Technol., 2006, 53, 139–146 CrossRef CAS PubMed.
- European Molecular Biology Laboratory, Compound: Bezafibrate (CHEMBL264374), https://www.ebi.ac.uk/explore/compound/CHEMBL264374, accessed 14 September 2025.
- R. M. A. Viegas, M. L. Melo, L. C. Brandão Lima, R. R. P. Garcia, E. C. S. Filho, J. A. Osajima and O. Chiavone-Filho, Carbamazepine adsorption with a series of organoclays: removal and toxicity analyses, Appl. Water Sci., 2024, 14, 133 CrossRef CAS.
- National Center for Biotechnology Information, PubChem Compound Summary for CID 3033, Diclofenac, https://pubchem.ncbi.nlm.nih.gov/compound/3033, accessed 14 September 2025.
- K. Chetnik, L. Petrick and G. Pandey, MetaClean: a machine learning-based classifier for reduced false positive peak detection in untargeted LC–MS metabolomics data, Metabolomics, 2020, 16, 117 CrossRef CAS PubMed.
- State Water Resources Control Board California Environmental Protection Agency, Water Quality Control Policy for Recycled Water, 2019 Search PubMed.
- N. R. M. M. C. (Australia) Environment Protection and Heritage Council (Australia), Australian Guidelines for Water Recycling: Managing Health and Environmental Risks (Phase 2): Augmentation of Drinking Water Supplies, Natural Resource Management Ministerial Council: Environment Protection and Heritage Council, Canberra, 2008 Search PubMed.
- Disinfection By-Products|The Safe Water System|CDC, https://www.cdc.gov/safewater/chlorination-byproducts.html, accessed 12 February 2021.
- Z. Zhang, Y.-H. Chuang, A. Szczuka, K. P. Ishida, S. Roback, M. H. Plumlee and W. A. Mitch, Pilot-scale evaluation of oxidant speciation, 1,4-dioxane degradation and disinfection byproduct formation during UV/hydrogen peroxide, UV/free chlorine and UV/chloramines advanced oxidation process treatment for potable reuse, Water Res., 2019, 164, 114939 CrossRef CAS PubMed.
- S. S. Lau, X. Wei, K. Bokenkamp, E. D. Wagner, M. J. Plewa and W. A. Mitch, Assessing Additivity of Cytotoxicity Associated with Disinfection Byproducts in Potable Reuse and Conventional Drinking Waters, Environ. Sci. Technol., 2020, 54, 5729–5736 CrossRef CAS PubMed.
- L. Font-Ribera, E. Marco, J. O. Grimalt, S. Pastor, R. Marcos, L. Abramsson-Zetterberg, M. Pedersen, T. Grummt, R. Junek, E. Barreiro, D. Heederik, J. Spithoven, R. Critelli, A. Naccarati, C. Schmalz, C. Zwiener, J. Liu, X. Zhang, W. Mitch, E. Gracia-Lavedan, L. Arjona, J. de Bont, L. Tarès, P. Vineis, M. Kogevinas and C. M. Villanueva, Exposure to disinfection by-products in swimming pools and biomarkers of genotoxicity and respiratory damage – The PISCINA2 Study, Environ. Int., 2019, 131, 104988 CrossRef CAS PubMed.
- T. Zeng and W. A. Mitch, Impact of Nitrification on the Formation of N -Nitrosamines and Halogenated Disinfection Byproducts within Distribution System Storage Facilities, Environ. Sci. Technol., 2016, 50, 2964–2973 CrossRef CAS PubMed.
- T. Zeng, M. J. Plewa and W. A. Mitch, N-Nitrosamines and halogenated disinfection byproducts in U.S. Full Advanced Treatment trains for potable reuse, Water Res., 2016, 101, 176–186 CrossRef CAS PubMed.
- L. Font-Ribera, M. Kogevinas, C. Schmalz, C. Zwiener, E. Marco, J. O. Grimalt, J. Liu, X. Zhang, W. Mitch, R. Critelli, A. Naccarati, D. Heederik, J. Spithoven, L. Arjona, J. de Bont, E. Gracia-Lavedan and C. M. Villanueva, Environmental and personal determinants of the uptake of disinfection by-products during swimming, Environ. Res., 2016, 149, 206–215 CrossRef CAS PubMed.
- A. Küster, A. C. Alder, B. I. Escher, K. Duis, K. Fenner, J. Garric, T. H. Hutchinson, D. R. Lapen, A. Péry, J. Römbke, J. Snape, T. Ternes, E. Topp, A. Wehrhan and T. Knacker, Environmental risk assessment of human pharmaceuticals in the European Union: A case study with the β-blocker atenolol, Integrated Environ. Assess. Manag., 2010, 6, 514–523 CrossRef PubMed.
- United States Environmental Protection Agency, CompTox Chemicals Dashboard v2.8.0, October 2025 Search PubMed.
- Drug Discovery and Chemical Biology Biocenter Finland, DrugMapper, https://drugmapper.helsinki.fi/CompoundInfo?compound_id=2127933, accessed 14 March 2026.
- C. Tixier, H. P. Singer, S. Oellers and S. R. Müller, Occurrence and Fate of Carbamazepine, Clofibric Acid, Diclofenac, Ibuprofen, Ketoprofen, and Naproxen in Surface Waters, Environ. Sci. Technol., 2003, 37, 1061–1068 CrossRef CAS PubMed.
- R. Pärnamäe, S. Mareev, V. Nikonenko, S. Melnikov, N. Sheldeshov, V. Zabolotskii, H. V. M. Hamelers and M. Tedesco, Bipolar membranes: A review on principles, latest developments, and applications, J. Membr. Sci., 2021, 617, 118538 CrossRef.
- R. Wódzki and J. Nowaczyk, Membrane transport of organics. III. Permeation of some carboxylic acids through bipolar polymer membrane, J. Appl. Polym. Sci., 2001, 80, 2705–2717 CrossRef.
- T. Bond, E. H. Goslan, S. A. Parsons and B. Jefferson, Treatment of disinfection by-product precursors, Environ. Technol., 2011, 32, 1–25 CrossRef CAS PubMed.
- K. M. Udert, T. A. Larsen and W. Gujer, Fate of major compounds in source-separated urine, Water Sci. Technol., 2006, 54, 413–420 CrossRef CAS PubMed.
- A. D. Shah and W. A. Mitch, Halonitroalkanes, Halonitriles, Haloamides, and N-Nitrosamines: A Critical Review of Nitrogenous Disinfection Byproduct Formation Pathways, Environ. Sci. Technol., 2012, 46, 119–131 CrossRef CAS PubMed.
- A. C. Diehl, G. E. Speitel Jr, J. M. Symons, S. W. Krasner, C. J. Hwang and S. E. Barrett, DBP formation during chloramination, J. AWWA, 2000, 92, 76–90 CrossRef CAS.
- G. Hua and D. A. Reckhow, Comparison of disinfection byproduct formation from chlorine and alternative disinfectants, Water Res., 2007, 41, 1667–1678 CrossRef CAS PubMed.
- Y.-H. Chuang, A. Szczuka and W. A. Mitch, Comparison of Toxicity-Weighted Disinfection Byproduct Concentrations in Potable Reuse Waters and Conventional Drinking Waters as a New Approach to Assessing the Quality of Advanced Treatment Train Waters, Environ. Sci. Technol., 2019, 53, 3729–3738 CrossRef CAS PubMed.
- Y. Chen, J. A. Wrubel, W. E. Klein, S. Kabir, W. A. Smith, K. C. Neyerlin and T. G. Deutsch, High-Performance Bipolar Membrane Development for Improved Water Dissociation, ACS Appl. Polym. Mater., 2020, 2, 4559–4569 CrossRef CAS PubMed.
- Fuel Cell Store, Technical data sheet for fumasep FBM, accessed 17 April 2024 Search PubMed.
- G. Mohandass, W. Chen, S. Krishnan and T. Kim, Asymmetric and Symmetric Redox Flow Batteries for Energy-Efficient, High-Recovery Water Desalination, Environ. Sci. Technol., 2022, 56, 4477–4488 CrossRef CAS PubMed.
- A. Xie and S. C. Popat, Electrochemical ammonia stripping from non-nitrified animal rendering wastewater, Chem. Eng. J. Adv., 2020, 3, 100020 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |