 Open Access Article
Open Access ArticleSustainable biomass-derived activated carbons from Rosa brunonii seeds for high-efficiency textile dye removal from water
Muhammad
Bilal
*a,
Muhammad
Idrees
a,
Javed
Ali
a,
Amir Hossein
Behroozi
bf,
Noushad
Hussain
a,
Vahid
Vatanpour
cde and
Tizazu H.
Mekonnen
 *f
*f
aDepartment of Chemistry, Kohat University of Science and Technology, Kohat, 26000, KPK, Pakistan. E-mail: mbilal@kust.edu.pk
bDepartment of Chemical Engineering, Queen's University, Kingston, ON K7L 3N6, Canada
cDepartment of Applied Chemistry, Faculty of Chemistry, Kharazmi University, 15719-14911, Tehran, Iran
dNational Research Center on Membrane Technologies, Istanbul Technical University, Maslak, 34469, Istanbul, Türkiye
eResearch Center for Environmental Chemistry and Green Development, Khazar University, Mahsati Str. 41, AZ-1096, Baku, Azerbaijan
fDepartment of Chemical Engineering, Institute of Polymer Research, University of Waterloo, Waterloo, ON N2L 3G1, Canada. E-mail: tmekonnen@uwaterloo.ca
First published on 11th March 2026
Abstract
Sustainable activated carbons were produced from Rosa brunonii Lindl. seeds through physical activation (PAC) and chemical activation (CAC) routes and evaluated for the removal of representative textile dyes, Disperse Orange 1 (DO1) and Eriochrome Black T (EBT), from water. The activation process induced pronounced structural and chemical transformations, leading to porous carbon frameworks with enriched carbon content of approximately 93 wt% and excellent thermal stability. Adsorption experiments demonstrated that dye uptake is strongly influenced by initial dye concentration, solution pH, temperature, and adsorbent dosage, and particle size. The chemically activated carbon exhibited superior adsorption performance, particularly toward EBT, achieving a maximum capacity of 256.4 mg g−1, whereas the PAC reached 192.3 mg g−1. For DO1, adsorption capacities of 73.0 and 69.4 mg g−1 were obtained for PAC and CAC, respectively. Equilibrium adsorption behavior was best described by the Langmuir model, indicating predominantly monolayer adsorption, and the adsorption kinetics followed a pseudo-second-order model, suggesting strong dye–surface interactions. Adsorption was favoured at lower temperatures, consistent with an exothermic process. Solution pH had a pronounced effect on EBT adsorption, with maximum uptake under acidic conditions due to electrostatic attraction, whereas DO1 adsorption was largely pH independent. Overall, this study demonstrates the effective valorisation of agricultural waste into high-performance adsorbents and highlights Rosa brunonii seed-derived activated carbons as promising materials for sustainable textile wastewater remediation.
Environmental significanceThis work presents a sustainable approach to textile wastewater treatment by converting discarded Rosa brunonii seeds into efficient activated carbons for dye removal. The biomass-derived adsorbents exhibit strong performance toward both disperse and anionic dyes, offering a low-cost and environmentally benign alternative to commercial activated carbons. By integrating agricultural waste valorization with water remediation, the study supports circular economy principles and addresses a critical challenge in sustainable pollution control. |
1 Introduction
Industrialization has been a key driver of economic development, yet the rapid expansion of industrial activities has generated severe environmental challenges due to inadequate management of industrial wastes. Among these sectors, the textile industry is particularly problematic, as it is one of the most water-intensive manufacturing industries, especially during wet-processing operations such as dyeing and finishing. These processes typically consume 150–200 L of water per kilogram of textile produced1 and generate large volumes of complex wastewater containing residual dyes, salts, auxiliary chemicals, and trace heavy metals.2 Although more than 70 million tonnes of synthetic dyes are produced annually, incomplete fixation during textile processing results in the discharge of approximately 10–15% of dyes into effluents, corresponding to hundreds of thousands of tonnes released into aquatic environments each year.3 Such dye-laden wastewaters are highly persistent and recalcitrant, imparting intense coloration even at low concentrations, reducing light penetration, increasing chemical oxygen demand, and severely disrupting aquatic ecosystems.4 Collectively, these impacts indicate the urgent need for advanced wastewater treatment strategies to mitigate the environmental consequences of dye-contaminated effluents.Wastewater discharged from textile industries is typically characterized by intense coloration, elevated chemical and biological oxygen demand (COD and BOD), and an alkaline pH, reflecting its chemically complex nature. Such effluents often include a broad range of recalcitrant organic compounds, salts, and auxiliary chemicals utilized throughout dyeing and finishing processes, which further complicate treatment.5 This complexity poses remarkable challenges for treatment, as conventional wastewater treatment plants often fail to completely remove synthetic dyes due to their high stability and resistance to biodegradation.6 Textile dyes are broadly classified into reactive, disperse, direct, vat, sulfur, and acid dyes based on their solubility and application routes, with azo dyes accounting for nearly 70% of global dye production.7 These dyes contain aromatic structures linked by azo (–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–) groups, which impart vivid and durable coloration but also confer pronounced environmental persistence.8 Under anaerobic or reducing conditions, azo dyes may undergo reductive cleavage, which leads to the generation of aromatic amines that are often more toxic, mutagenic, and carcinogenic than the parent dye molecules.9 Once discharged into aquatic environments, dyes can form light-blocking layers that inhibit photosynthesis, reduce dissolved oxygen levels, and disrupt aquatic food webs, ultimately leading to ecological degradation. Beyond their environmental impacts, dye contaminants compromise water aesthetics and quality, limit recreational and domestic use, and pose potential human health risks through bioaccumulation and trophic transfer.10
N–) groups, which impart vivid and durable coloration but also confer pronounced environmental persistence.8 Under anaerobic or reducing conditions, azo dyes may undergo reductive cleavage, which leads to the generation of aromatic amines that are often more toxic, mutagenic, and carcinogenic than the parent dye molecules.9 Once discharged into aquatic environments, dyes can form light-blocking layers that inhibit photosynthesis, reduce dissolved oxygen levels, and disrupt aquatic food webs, ultimately leading to ecological degradation. Beyond their environmental impacts, dye contaminants compromise water aesthetics and quality, limit recreational and domestic use, and pose potential human health risks through bioaccumulation and trophic transfer.10
Among the numerous dyes released from textile effluents, Disperse Orange 1 (DO1) and Eriochrome Black T (EBT) are of environmental concern because of their widespread use and potential toxicity. DO1 is an azo-based disperse dye commonly applied in synthetic textile dyeing and has been reported as a sensitizing agent in humans, with evidence of cross-sensitization involving structurally related aromatic amines, such as 4-nitroaniline and p-aminodiphenylamine.11 The persistence of DO1 in wastewater and its potential to generate hazardous transformation products further heighten concerns regarding its environmental release. EBT, an azo dye widely employed both as a metallochromic indicator in EDTA titrations12 and as a textile colorant for fibers, such as wool, nylon, and silk,13 poses notable health and environmental risks. Exposure to EBT-contaminated water has been associated with skin, ocular, respiratory, and gastrointestinal irritation, whereas prolonged contact may induce dermatitis and potential mutagenic effects.14 Therefore, it is necessary to develop efficient and sustainable dye-removal strategies prior to wastewater discharge.
To mitigate the adverse impacts of such dyes, a variety of physical and chemical treatment methods have been investigated, including chemical precipitation, flocculation, oxidation, electrochemical processes, ion exchange, and adsorption.15 Although these approaches can achieve partial or complete dye removal, many suffer from inherent limitations, such as high operational costs, incomplete mineralization, secondary sludge generation, and complex process requirements. Advanced oxidation processes, for instance, are effective in degrading dye molecules but often rely on high energy inputs and expensive reagents, restricting their large-scale implementation, particularly in resource-limited settings.16 In contrast, adsorption has emerged as a highly attractive alternative due to its operational simplicity, cost-effectiveness, and environmental compatibility.17,18 Importantly, adsorption enables efficient dye removal without producing substantial secondary waste, making it a promising and sustainable strategy for treating dye-contaminated textile effluents.19
Among available adsorbents, activated carbon remains the benchmark material for the removal of organic and inorganic contaminants from water and air owing to its high surface area, well-developed porous structure, and favorable surface chemistry.20 However, the high production and regeneration costs associated with commercial activated carbon significantly limit its widespread application, particularly in large-scale wastewater treatment.21 In response, growing attention has been directed toward the valorization of agricultural residues as low-cost precursors for activated carbon production.22 Biomass-derived sources, such as fruit shells,23 seeds,24 husks,25 offer renewable, economically viable alternatives while simultaneously addressing agricultural waste disposal challenges. This integrated approach aligns environmental remediation with resource sustainability, positioning biomass-based activated carbons as promising candidates for cost-effective treatment of dye-contaminated textile effluents. Despite extensive research on common agricultural wastes, several regionally abundant and lignocellulose-rich biomasses remain unexplored as potential precursors for activated carbon synthesis.
In this work, Rosa brunonii Lindl. seeds were investigated as a sustainable and underutilized biomass precursor for the preparation of physically and chemically activated carbons aimed at the removal of representative azo dyes, DO1 and EBT, from aqueous environments. Rosa brunonii is a widely distributed wild Rose, particularly abundant in mountainous and temperate regions, whose seeds are typically discarded as agricultural or forestry residue with no established value-added application. The seeds possess a high lignocellulosic and carbon content, low ash fraction, and dense organic structure, which are desirable characteristics for activated carbon production, yet their potential as an adsorbent precursor has not been previously reported. This study examines how different activation strategies influence the structural and surface properties of the resulting carbons and how these features govern their adsorption behavior. Batch adsorption experiments were used to evaluate the effects of solution pH, initial dye concentration, temperature, and adsorbent dosage, whereas kinetic and equilibrium modeling provided insight into the adsorption mechanisms. To the best of our knowledge, this work represents the first systematic investigation of activated carbons derived from Rosa brunonii seeds. By coupling biomass waste valorisation with dye remediation, this study contributes to the development of sustainable and cost-effective materials for textile wastewater treatment.
2 Materials and methods
2.1 Materials
All chemicals and reagents used in this study were of analytical grade and were used as received without further purification. Disperse Orange 1 (DO1) and Eriochrome Black T (EBT) dyes were purchased from Sigma-Aldrich (Karachi, Pakistan). Orthophosphoric acid (H3PO4, ≥85%), sodium hydroxide (NaOH, ≥98%), hydrochloric acid (HCl, 37%), nitric acid (HNO3, 65%), and sodium nitrate (NaNO3, ≥99%) were also obtained from Sigma-Aldrich (Karachi, Pakistan). High-purity nitrogen gas was used to provide an inert atmosphere during carbonization. Distilled water was employed throughout all experiments.2.2 Preparation of adsorbents
For the preparation of chemically activated carbon (CAC), 5 g of the biomass was first impregnated with 50 mL of 0.1 mol per L H3PO4, corresponding to a biomass-to-acid impregnation ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v), defined here as the mass of dry biomass (g) to the volume of phosphoric acid solution (mL), and agitated at 250 rpm for 60 min. The impregnated biomass was then repeatedly washed with distilled water (typically 4–6 successive washing cycles) until a near-neutral pH (≈7) was attained, which was used as the stopping criterion for washing, followed by carbonization under the same conditions used for PAC synthesis.27,28 Then, both PAC and CAC samples were sieved to obtain particle size fractions of 45–90 µm, 90–212 µm, and greater than 212 µm to unsure uniformity of the adsorbents for subsequent adsorption studies. The chemical activation with H3PO4 follows a more intricate mechanism dominated by acid-catalyzed dehydration, crosslinking modulation, and pore development. During the impregnation step, phosphoric acid penetrates the biomass matrix and interacts with hydroxyl groups present in cellulose, hemicellulose, and lignin, facilitating dehydration and cleavage of ether and ester linkages. These reactions inhibit tar formation during subsequent carbonization and promote the generation of thermally stable phosphate and polyphosphate intermediates. Upon heating, the phosphorus-containing species act as pore-directing agents, limiting structural shrinkage and fostering the formation of an interconnected porous network. The concurrent release of volatile byproducts, such as water vapor and low-molecular-weight oxygenated compounds, further contributes to pore expansion. Following carbonization, residual phosphorus species and soluble phosphate byproducts are removed during washing, leaving behind enlarged pores and surface oxygen functionalities that enhance the adsorption performance of the activated carbon.
10 (w/v), defined here as the mass of dry biomass (g) to the volume of phosphoric acid solution (mL), and agitated at 250 rpm for 60 min. The impregnated biomass was then repeatedly washed with distilled water (typically 4–6 successive washing cycles) until a near-neutral pH (≈7) was attained, which was used as the stopping criterion for washing, followed by carbonization under the same conditions used for PAC synthesis.27,28 Then, both PAC and CAC samples were sieved to obtain particle size fractions of 45–90 µm, 90–212 µm, and greater than 212 µm to unsure uniformity of the adsorbents for subsequent adsorption studies. The chemical activation with H3PO4 follows a more intricate mechanism dominated by acid-catalyzed dehydration, crosslinking modulation, and pore development. During the impregnation step, phosphoric acid penetrates the biomass matrix and interacts with hydroxyl groups present in cellulose, hemicellulose, and lignin, facilitating dehydration and cleavage of ether and ester linkages. These reactions inhibit tar formation during subsequent carbonization and promote the generation of thermally stable phosphate and polyphosphate intermediates. Upon heating, the phosphorus-containing species act as pore-directing agents, limiting structural shrinkage and fostering the formation of an interconnected porous network. The concurrent release of volatile byproducts, such as water vapor and low-molecular-weight oxygenated compounds, further contributes to pore expansion. Following carbonization, residual phosphorus species and soluble phosphate byproducts are removed during washing, leaving behind enlarged pores and surface oxygen functionalities that enhance the adsorption performance of the activated carbon.
2.3 Characterization of ACs
The raw biomass and the synthesized ACs were characterized using complementary analytical techniques. Surface functional groups were identified by Fourier transform infrared (FTIR) spectroscopy (Spectrum Two, PerkinElmer) over the range of 400–4000 cm−1. Thermogravimetric analysis (TGA) was performed using a Diamond Series analyzer (PerkinElmer, USA) under a nitrogen atmosphere to evaluate thermal stability and carbonization behaviour, with the temperature ramped from 20 to 900 °C at a heating rate of 10 °C min−1. Surface morphology and textural features were examined using scanning electron microscopy (SEM) (JSM-5910, JEOL, Japan) operated at an accelerating voltage of 10 kV and magnifications between 150× and 5000×. Elemental composition and surface elemental distribution were determined by energy-dispersive X-ray spectroscopy (EDX) using an INCA 200 system (Oxford Instruments, UK) coupled to the SEM. Crystallographic characteristics were analyzed using X-ray diffraction (XRD) (JDX-3532, JEOL, Japan) with Cu Kα radiation (λ = 1.5418 Å). UV-visible spectroscopy (Shimadzu UV-1800) was employed to quantify dye concentrations and evaluate adsorption performance, using a wavelength range of 190–800 nm and a slit width of 1.0 nm. Specific surface area measurements were performed using a NOVA 1200e gas sorption analyzer (Quantachrome Instruments, USA) based on nitrogen adsorption at 77 K, with BET analysis conducted over a relative pressure (P/P0) range of 0.05–0.30.The point of zero charge (pHPZC) of the ACs was determined using the pH drift method. Briefly, 40 mL aliquots of 0.1 M NaNO3 solution were adjusted to initial pH values between 2 and 12 using 0.1 M HNO3 or 0.1 M NaOH. Activated carbon (0.1 g) was then added to each solution, and the suspensions were agitated at 250 rpm for 24 h. The final pH was recorded after equilibration, and the pHPZC was identified as the pH at which the difference between initial and final pH (ΔpH) was zero, corresponding to a neutral surface charge.29
2.4 Adsorption experiments
The adsorption behaviour of Disperse Orange 1 (DO1) and Eriochrome Black T (EBT) on physically activated carbon (PAC) and chemically activated carbon (CAC) was systematically investigated through batch experiments by varying key operational parameters. The effects of initial dye concentration (5–1000 mg L−1), solution pH (2–9, adjusted using 0.1 mol per L HCl and 0.1 mol per L NaOH), adsorbent dosage (0.1–0.5 g per 50 mL, corresponding to 2–10 g L−1), contact time (up to 60 min), temperature (0–80 °C), and adsorbent particle size (90–212 µm) were evaluated. When one parameter was varied, all other parameters were maintained constant at pH 6.5–7.0, a contact time of 60 min (ensuring equilibrium), an adsorbent dosage of 0.1 g, an initial dye concentration of 50 mg L−1, and a temperature of 25 °C. All experiments were conducted using 50 mL dye solutions under constant agitation (250 rpm), unless otherwise specified. Equilibrium adsorption data obtained at near-neutral pH (6.5–7.0) and 25 °C were analyzed using Langmuir and Freundlich isotherm models to elucidate adsorption behavior and surface characteristics of the adsorbents. Following the adsorption process, the solutions were filtered through Whatman No. 42 filter paper and residual dye concentrations were determined by UV-visible spectrophotometry following filtration. The performance of ACs was evaluated in terms of adsorption capacity (qe, mg g−1) measured by the following equation:30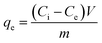 | (1) |
2.5 Kinetics and isotherm studies
Adsorption kinetics were evaluated using the pseudo-first order (PFO) and pseudo-second order (PSO) models to elucidate the rate-controlling mechanisms. The PFO model assumes that the adsorption rate is proportional to the number of unoccupied active sites and is described by the following equation:31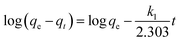 | (2) |
The PSO model, which presumes that adsorption follows a second-order mechanism with respect to the available sites, is expressed by the following equation:31
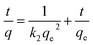 | (3) |
Equilibrium adsorption behavior was analyzed using the Langmuir and Freundlich isotherm models. The Langmuir model assumes monolayer adsorption on a homogeneous surface with finite identical sites and is given by the following equation:31
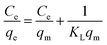 | (4) |
The favorability of adsorption was further assessed using the dimensionless separation factor RL, calculated according to the following equation:31
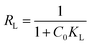 | (5) |
The Langmuir constant KL reflects the affinity between the adsorbate molecules and the adsorbent surface and is commonly associated with the adsorption energy. It is obtained from the slope and intercept of the linearized Langmuir plot and provides insight into the strength of adsorbate–adsorbent interactions.
The Freundlich model (eqn (6)) accounts for multilayer adsorption on energetically heterogeneous surfaces and is expressed as:
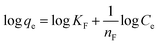 | (6) |
3 Results and discussion
3.1 Characterization of activated carbons
The surface morphology of the raw biomass and the derived activated carbons was investigated by SEM to assess the structural changes induced by carbonization and activation. As depicted in Fig. 2a, the untreated biomass exhibits a dense and relatively smooth surface with limited textural features, indicating the absence of a well-developed porous network prior to thermal treatment. After activation, substantial morphological transformations are observed. The PAC (Fig. 2b) shows a roughened surface with the formation of irregular cavities and fissures, which can be attributed to the thermal decomposition of lignocellulosic components and the release of volatile species during pyrolysis. In comparison, the CAC (Fig. 2c) prepared using H3PO4 displays a more pronounced and interconnected porous structure with widened pore openings and a sponge-like appearance, reflecting the role of acid in promoting dehydration, structural expansion, and pore development during carbonization. Overall, the enhanced surface accessibility and active site availability of PAC and CAC are favourable for adsorption applications in aqueous systems.FTIR spectroscopy was employed to identify the surface functional groups present in the raw biomass and to assess the chemical transformations induced by carbonization and activation, as displayed in Fig. 3a. The spectrum of the raw biomass displays several characteristic bands associated with oxygenated and aliphatic functionalities. The broad band centered at approximately 3293 cm−1 corresponds to O–H stretching vibrations, indicating the presence of hydroxyl groups from alcohols, phenols, and adsorbed moisture.32 The strong absorption at 2927 cm−1 is assigned to asymmetric C–H stretching of methylene groups, confirming the abundance of aliphatic hydrocarbons in the native biomass.33 The band observed at 1731 cm−1 is attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations of carbonyl groups, which may originate from aldehydes, ketones, or ester functionalities.34 Additional bands at 1644 cm−1 and 1518 cm−1 are associated with C
O stretching vibrations of carbonyl groups, which may originate from aldehydes, ketones, or ester functionalities.34 Additional bands at 1644 cm−1 and 1518 cm−1 are associated with C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching of conjugated structures35 and N–O stretching vibrations,36 respectively, whereas the peak at 1456 cm−1 corresponds to C–H bending vibrations of methyl and methylene groups.37 The bands at 1231 cm−1 and 1038 cm−1 are attributed to C–O stretching vibrations, consistent with the presence of ether, alcohol, and phenolic groups in the lignocellulosic framework.38 Following carbonization, the FTIR spectra of PAC and CAC show a pronounced reduction in band intensity across the 3500 to 1600 cm−1 region, reflecting the extensive decomposition of hydroxyl, carbonyl, and aliphatic functional groups due to thermal treatment at 600 °C. The disappearance of the O–H band and the strong attenuation of C–H stretching bands confirm effective devolatilization and aromatization of the carbon structure.39 Both activated carbons exhibit weak bands near 1630 to 1520 cm−1, which are attributed to C
C stretching of conjugated structures35 and N–O stretching vibrations,36 respectively, whereas the peak at 1456 cm−1 corresponds to C–H bending vibrations of methyl and methylene groups.37 The bands at 1231 cm−1 and 1038 cm−1 are attributed to C–O stretching vibrations, consistent with the presence of ether, alcohol, and phenolic groups in the lignocellulosic framework.38 Following carbonization, the FTIR spectra of PAC and CAC show a pronounced reduction in band intensity across the 3500 to 1600 cm−1 region, reflecting the extensive decomposition of hydroxyl, carbonyl, and aliphatic functional groups due to thermal treatment at 600 °C. The disappearance of the O–H band and the strong attenuation of C–H stretching bands confirm effective devolatilization and aromatization of the carbon structure.39 Both activated carbons exhibit weak bands near 1630 to 1520 cm−1, which are attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching within aromatic domains,40 indicating the formation of a graphitized carbon framework. A small but distinct band observed around 1130 cm−1 in both PAC and CAC can be assigned to C–O stretching vibrations of phenolic or ether groups, while its slightly enhanced intensity in CAC likely reflects the contribution of phosphorus-containing oxygen functionalities formed during H3PO4 activation. Additionally, the weak band near 866 cm−1 corresponds to out-of-plane C–H bending vibrations of aromatic rings.40 Overall, the substantial loss of oxygenated and aliphatic functionalities and the emergence of aromatic carbon structures confirm that carbonization and activation induced significant chemical bond cleavage and structural rearrangement, yielding activated carbons with chemically stable surfaces suitable for adsorption applications.
C stretching within aromatic domains,40 indicating the formation of a graphitized carbon framework. A small but distinct band observed around 1130 cm−1 in both PAC and CAC can be assigned to C–O stretching vibrations of phenolic or ether groups, while its slightly enhanced intensity in CAC likely reflects the contribution of phosphorus-containing oxygen functionalities formed during H3PO4 activation. Additionally, the weak band near 866 cm−1 corresponds to out-of-plane C–H bending vibrations of aromatic rings.40 Overall, the substantial loss of oxygenated and aliphatic functionalities and the emergence of aromatic carbon structures confirm that carbonization and activation induced significant chemical bond cleavage and structural rearrangement, yielding activated carbons with chemically stable surfaces suitable for adsorption applications.
 | ||
| Fig. 3 Structural characterization of raw biomass, PAC, and CAC: (a) FTIR spectra, (b) XRD patterns, (c) TGA curves, and (d) elemental analysis based on EDX. | ||
The XRD patterns of the activated carbons derived from Rosa brunonii Lindl. seeds are presented in Fig. 3b. Both PAC and CAC exhibit a broad diffraction feature centered near 2θ ≈ 21°, which is characteristic of turbostratic or amorphous carbon with limited long-range crystalline order. The broadening and low intensity of this peak indicate a predominantly disordered carbon structure, whereas its slight shift toward higher angles suggests partial development of aromatic stacking during thermal treatment. Such structural evolution is commonly associated with progressive aromatization rather than true graphitization and is beneficial for creating a porous framework with abundant defect sites that enhance surface reactivity and adsorption performance.41,42
TGA was conducted to evaluate the thermal behavior of the raw biomass and to confirm the suitability of the selected carbonization temperature for producing stable activated carbons, as depicted in Fig. 3c. The activated carbon samples exhibit minimal mass loss, approximately 5%, below 100 °C, which is attributed to the removal of physically adsorbed moisture. Beyond this temperature, both PAC and CAC demonstrate excellent thermal stability, with only a gradual weight reduction observed up to 900 °C and a total mass loss of ∼17%, indicating a highly carbonized and structurally reinforced framework. The high residual mass retained by PAC and CAC reflects their pre-carbonized nature and confirms that the selected carbonization temperature is sufficient to remove volatile components while preserving a stable carbonaceous backbone. In contrast, raw biomass undergoes extensive thermal degradation, with an overall mass loss exceeding 80%. The initial weight reduction below 120 °C corresponds to moisture evaporation, followed by a major decomposition stage between 200 °C and 600 °C arising from the thermal breakdown of hemicellulose, cellulose, lignin, and other volatile organic constituents. At 600 °C, the biomass retained ∼21 wt% of its initial mass under nitrogen, which is attributed to the formation of a carbonaceous char residue during inert pyrolysis. A final, slower degradation stage extending to 900 °C reflects the progressive decomposition of more thermally stable carbonaceous residues. The pronounced difference in thermal behavior between the raw precursor and the activated carbons confirms that carbonization and activation effectively transform the biomass into a thermally stable carbon material suitable for high-temperature and adsorption-based applications.43,44
The elemental composition of the materials was further examined by EDX analysis to confirm the formation and stability of the activated carbons, as shown in Fig. 3d. The EDX spectrum of the raw biomass indicates that it is primarily composed of carbon, oxygen, and nitrogen, with minor contributions from inorganic elements such as Mg, Al, K, and Ca, as well as trace amounts of P and S, which are commonly associated with naturally occurring mineral components. After carbonization and activation, a pronounced change in elemental composition is observed (Fig. 3d). The PAC and CAC consist predominantly of carbon and oxygen, with only trace levels of Mg, Al, Si, P, S, and Ca remaining, indicating effective removal of most noncarbonaceous constituents during thermal treatment. Quantitative analysis reveals a substantial increase in carbon content from 64.95% in the raw biomass to more than 93% in the ACs (both PAC and CAC), accompanied by a corresponding decrease in oxygen content from 21.27% to ∼5%. Notably, nitrogen detected in the raw precursor is no longer observed after carbonization, reflecting the decomposition of nitrogen-containing organic moieties at elevated temperatures. These results confirm that pyrolysis at 600 °C promotes extensive devolatilization and aromatization, leading to the formation of a carbon-rich structure dominated by condensed aromatic domains. The high carbon content and reduced heteroatom concentration are expected to enhance structural stability and contribute to the favorable textural properties of the ACs.45,46
The BET surface area parameters of PAC and CAC (Table 1) were obtained from multi-point BET analysis (Fig. S1a, SI). CAC exhibits a higher monolayer adsorption capacity (Wm) and a correspondingly larger specific surface area (95.6 m2 g−1) compared with PAC (79.7 m2 g−1), indicating more effective pore development upon chemical activation. The BET constants for both materials (CBET ≈ 3.9) are comparable, suggesting similar surface–adsorbate interaction energies for nitrogen adsorption at 77 K. The pore size distribution derived from nitrogen adsorption data is presented in Fig. S1b, indicating dominant pores at the micropore–mesopore boundary, with average pore radii of ∼1.72 nm for PAC and ∼1.89 nm for CAC and corresponding total pore volumes of 0.012 and 0.014 cm3 g−1, respectively. Overall, these results confirm that activation enhances the accessible surface area while preserving comparable adsorption energetics, which is consistent with the moderate surface heterogeneity expected for biomass-derived activated carbons.
| Sample | W m (g N2 per g) | C BET | S BET (m2 g−1) | V p (cm3 g−1) | r p (nm) |
|---|---|---|---|---|---|
| PAC | 0.0229 | 3.86 | 79.7 | 0.012 | 1.72 |
| CAC | 0.0275 | 3.94 | 95.6 | 0.014 | 1.89 |
The point of zero charge (pHPZC) is a key interfacial parameter that describes the surface charge behaviour of an adsorbent and provides insight into its surface chemistry under varying pH conditions. It represents the pH at which the net surface charge of the material is zero and therefore governs electrostatic interactions between the adsorbent and charged species in solution. The pHPZC values of the activated carbons were determined from the pH drift curves shown in Fig. 4. At very low pH values such as pH 2, the activated carbon surface becomes rapidly saturated with protons, limiting further proton uptake and resulting in a relatively small change in ΔpH. In contrast, at moderately acidic conditions around pH 4, surface functional groups are not fully protonated and actively participate in proton exchange, leading to a higher ΔpH. This behavior reflects the buffering capacity of oxygen-containing surface groups and their pKa-dependent acid–base equilibria. The negative ΔpH minimum observed near pH 10 is attributed to the deprotonation of acidic surface groups, particularly phenolic and carboxylic functionalities, which release protons into the solution and lower the final pH. For both PAC and CAC, the intersection of the curves with the zero ΔpH line occurs at approximately pH 7.9, indicating identical pHPZC values for the two materials. Although phosphoric acid activation is commonly associated with the introduction of acidic surface functionalities, extensive post-activation washing combined with subsequent thermal treatment can substantially diminish residual phosphate species on the carbon surface. As a result, the surface charge may shift toward near-neutral behavior, leading to pHPZC values close to neutrality, as observed in the present study. This behavior is consistent with the findings of Rai et al.,47 who reported a pHPZC of 6.8 for mango kernel-derived activated carbon prepared using 40% H3PO4 after thorough washing to a neutral filtrate pH, demonstrating that near-neutral pHPZC values are attainable for phosphoric-acid-activated biomass carbons following appropriate post-treatment. At solution pH values below 7.9, the surfaces of both activated carbons are predominantly positively charged, whereas at pH values above this value, the surface charge becomes increasingly negative. The near symmetry of the ΔpH extrema around the pHPZC reflects the amphoteric nature of the activated carbon surface, with protonation dominating under acidic conditions and deprotonation prevailing in alkaline media.48
3.2 Effects of operating conditions on dye adsorption
The adsorption behaviour of the PAC and CAC activated carbons derived from Rosa brunonii Lindl. seeds were systematically investigated through a series of batch adsorption experiments. Key operational parameters, including initial dye concentration for isotherm analysis, contact time for kinetic evaluation, temperature, solution pH, adsorbent dosage, and particle size, were independently varied to assess their influence on adsorption performance and to elucidate the governing adsorption mechanisms.3.3 Kinetics and isotherm modelling
Adsorption kinetics and equilibrium behavior are key aspects for understanding the interaction between dye molecules and adsorbent surfaces, as well as for evaluating the efficiency and feasibility of adsorption processes. Kinetic studies provide insight into the rate of adsorption and the time required to reach equilibrium, which are critical parameters for process design and optimization. Isotherm analyses, on the other hand, describe how adsorbate molecules distribute between the solid and liquid phases at equilibrium and offer information on adsorption capacity, surface characteristics, and adsorption mechanisms.The kinetic and isotherm data for the adsorption of DO1 and EBT onto PAC and CAC are presented in Fig. 6. The kinetic profiles in Fig. 6a show a rapid initial uptake of both dyes, followed by a gradual approach to equilibrium. For DO1, adsorption equilibrium was attained within approximately 30 min for both PAC and CAC, whereas EBT reached equilibrium much faster, within about 10 min, indicating a higher affinity of the activated carbon surfaces toward EBT molecules. Beyond these equilibrium times, further increases in contact duration resulted in negligible changes in adsorption capacity, and complete stabilization of the adsorption process was observed within ∼60 min for all systems. Accordingly, a contact time of 60 min was selected for all subsequent adsorption experiments to ensure that equilibrium was reached for every dye–adsorbent system and to allow meaningful and consistent comparison of adsorption performance across the different systems studied. The fast initial adsorption stage stems from the abundant availability of active surface sites on the activated carbons, which enables rapid interaction between dye molecules and adsorption sites. As adsorption proceeds, these sites become progressively occupied, leading to a reduced adsorption rate due to increased competition among dye molecules and diffusion limitations. This transition reflects a shift from surface-controlled adsorption at early stages to intraparticle and pore diffusion control at later stages, which is consistent with previously reported dye adsorption systems.59 The equilibrium adsorption behavior is illustrated in Fig. 6b, where the adsorption capacity of both DO1 and EBT increases with increasing dye concentration for PAC and CAC. This trend is associated with the enhanced concentration gradient at higher initial dye concentrations, which provides a stronger driving force for mass transfer and promotes more efficient interaction between dye molecules and the adsorbent surface.60
 | ||
| Fig. 6 Adsorption isotherm and kinetic data for DO1 and EBT on PAC and CAC: (a) kinetics; (b) isotherm. | ||
The adsorption kinetics of DO1 and EBT on PAC and CAC were analyzed using the PFO and PSO kinetic models to elucidate the rate behavior and controlling mechanisms of the adsorption process. The linearized kinetic fittings are presented in Fig. 7, where Fig. 7a illustrates the PFO model and Fig. 7b shows the PSO model for both dyes on the two ACs. The corresponding kinetic parameters are summarized in Table 2. The PFO model provides a reasonable description of DO1 adsorption, with correlation coefficients (R2) of 0.9186 for PAC and 0.9431 for CAC. However, the applicability of the PFO model to EBT adsorption is limited, particularly for EBT–PAC, where a markedly lower R2 value of 0.7726 is obtained. In addition, the equilibrium adsorption capacities calculated from the PFO model differ substantially from the experimentally measured values, indicating that this model does not adequately represent the adsorption kinetics of EBT on the prepared activated carbons.
| Model | Parameter | DO1 | EBT | ||
|---|---|---|---|---|---|
| PAC | CAC | PAC | CAC | ||
| Langmuir | K L (L mg−1) | 0.021 | 0.114 | 0.003 | 0.004 |
| q m (mg g−1) | 72.99 | 69.44 | 192.31 | 256.41 | |
| R L | 0.487 | 0.149 | 0.889 | 0.838 | |
| R 2 | 0.9664 | 0.9928 | 0.9724 | 0.9952 | |
| Freundlich | n F | 1.604 | 1.926 | 1.186 | 1.363 |
| K F | 1.518 | 2.114 | 0.881 | 1.249 | |
| R 2 | 0.9189 | 0.9122 | 0.9322 | 0.9326 | |
| PFO | k 1 | 0.021 | 0.024 | 0.018 | 0.032 |
| q e | 2.636 | 3.102 | 3.024 | 2.841 | |
| R 2 | 0.9186 | 0.9431 | 0.7726 | 0.8740 | |
| PSO | k 2 | 0.011 | 0.007 | 0.009 | 0.013 |
| q e | 17.54 | 21.27 | 32.05 | 36.90 | |
| R 2 | 0.9983 | 0.9967 | 0.9983 | 0.9997 | |
In contrast, the PSO model exhibits excellent agreement with the experimental data for both dyes and both adsorbents, as shown in Fig. 7b. High correlation coefficients close to unity are obtained, with R2 values of 0.9983 and 0.9967 for DO1 adsorption on PAC and CAC, respectively, and 0.9983 and 0.9997 for EBT adsorption on PAC and CAC. Moreover, the equilibrium adsorption capacities predicted by the PSO model closely match the experimental values, confirming the suitability of this model for describing the adsorption kinetics. The superior fitting of the PSO model suggests that the adsorption rate is governed by interactions between dye molecules and the available active sites on the activated carbon surfaces rather than by simple diffusion-controlled processes alone. Similar kinetic behavior has been widely reported for dye adsorption onto activated carbon materials,61 where the PSO model consistently provides the best representation of experimental data.
The equilibrium adsorption behavior of DO1 and EBT on PAC and CAC was evaluated using the Langmuir and Freundlich isotherm models, with the corresponding linear fittings shown in Fig. 8 and the derived parameters summarized in Table 2. As illustrated in Fig. 8a, the Langmuir model provides an excellent fit to the experimental data for both dyes and both adsorbents, as evidenced by high correlation coefficients ranging from 0.9664 to 0.9952. The maximum monolayer adsorption capacities obtained from the Langmuir model demonstrate a markedly higher affinity of the activated carbons toward EBT compared to DO1, with qm values of 192.31 and 256.41 mg g−1 for EBT on PAC and CAC, respectively, whereas DO1 exhibits lower capacities of 72.99 and 69.44 mg g−1. The dimensionless separation factor RL values lie between 0 and 1 for all systems, confirming that adsorption is favorable over the studied concentration range.62 The higher adsorption capacity observed for CAC, particularly toward EBT, arises from its more developed porous structure and greater availability of accessible adsorption sites.
 | ||
| Fig. 8 Isotherm model fitting for DO1 and EBT adsorption on PAC and CAC: (a) Langmuir model; (b) Freundlich model. | ||
The Freundlich isotherm model, which accounts for adsorption on heterogeneous surfaces with nonuniform site energies, was also applied to the equilibrium data, as illustrated in Fig. 8b. Although reasonable correlations were obtained, with R2 values between 0.9122 and 0.9326, these values are consistently lower than those obtained from the Langmuir model, indicating a comparatively weaker description of the adsorption behaviour. The Freundlich constants nF are greater than unity for all systems, indicating favourable adsorption, whereas the KF values suggest enhanced adsorption affinity for CAC relative to PAC.63 Overall, the isotherm analysis indicates that the equilibrium adsorption of both DO1 and EBT on the prepared ACs is best described by the Langmuir model, implying predominantly monolayer adsorption on relatively uniform surface sites under the investigated conditions.
3.4 Adsorption mechanisms
The plausible adsorption mechanisms governing the uptake of DO1 and EBT dyes onto PAC and CAC are schematically illustrated in Fig. 9 and arise from the synergistic contribution of multiple surface interactions. For DO1, adsorption is predominantly driven by hydrophobic interactions between the nonpolar aromatic backbone of the dye and the graphitized carbon domains of the activated carbons, which minimize interfacial free energy in aqueous media.64 In parallel, strong π–π stacking interactions occur between the planar aromatic rings of DO1 and the conjugated sp2-carbon structures present on PAC and CAC, facilitating favorable face-to-face alignment and stabilizing adsorption.65,66 In addition, pore filling plays an important role in DO1 uptake, as the molecular dimensions of the dye enable its diffusion into the micro- and mesoporous network of the activated carbons, leading to physical confinement and enhanced retention within the pore structure.19 Although the BET surface areas of PAC and CAC are moderate, the accessibility of the pore network allows effective utilization of the available internal surface, which supports efficient adsorption of aromatic molecules like DO1.67 Notably, the higher adsorption capacity of DO1 on PAC compared with CAC can be attributed to the more graphitized and less functionalized surface of PAC formed during physical activation, which enhances hydrophobic interactions and π–π stacking with nonpolar aromatic dyes.In contrast, the adsorption of EBT involves a more complex combination of electrostatic and noncovalent interactions due to its anionic and highly polar nature. The sulfonate groups of EBT impart a negative charge in aqueous solution, promoting electrostatic attraction with positively charged surface sites on PAC and CAC at solution pH values below the pHPZC.57 Hydrogen bonding interactions further contribute to EBT adsorption through interactions between oxygen-containing functional groups on the activated carbon surface and the hydroxyl and sulfonate moieties of the dye.68 These surface functional groups, which are enriched during chemical activation, increase surface polarity and provide favorable interaction sites, thereby enhancing dye affinity.69 Additionally, π–π stacking between the aromatic rings of EBT and the graphitic regions of the carbons enhances adsorption through noncovalent electronic interactions.60 Pore filling also contributes to EBT uptake, particularly in CAC, where chemical activation generates a more developed pore network and higher accessibility of internal adsorption sites. Moreover, the accessibility of the pore network facilitates diffusion of the relatively bulky EBT molecules, allowing effective interaction with internal adsorption sites even in materials with moderate BET surface areas. As a result, CAC exhibits a higher adsorption capacity for EBT than PAC (Table 3), reflecting the dominant role of electrostatic attraction and hydrogen bonding associated with the higher surface functionality introduced by phosphoric acid activation. The combined contributions of electrostatic attraction, hydrogen bonding, π–π interactions, hydrophobic effects, and pore filling collectively account for the higher adsorption capacity observed for EBT, especially on CAC, where surface functionality and porosity are more pronounced.
| Adsorbent | Dye | Optimal pH [pHPZC] | Adsorbent dosage (g L−1) | Kinetics model | Isotherm model | q max (mg g−1) | Ref. |
|---|---|---|---|---|---|---|---|
| AC from Euphorbia rigida | DO25 | Independent [—] | 1.2 | PSO | Langmuir | 118.9 | 52 |
| Cenospheres/AC composite | DO25 | 6 [—] | 0.3/0.1 | PSO | Langmuir | 90.9 | 70 |
| AC from rattan sawdust | DO30 | — [—] | 0.3 | — | Langmuir | 133.3 | 71 |
| AC from orange peel | DO7 | 2 [—] | 0.75 | PSO | Temkin | 357.1 | 72 |
| AC from Rosa brunonii Lindl. seeds | DO1 | Independent [7.9] | 2 | PSO | Langmuir | PAC: 73.0 | This work |
| CAC: 69.4 | |||||||
| AC from waste hemp | EBT | 3 [—] | 0.4 | PSO | Langmuir | 14.0 | 73 |
| AC from tea waste | EBT | 2 [—] | 11 | PSO | Langmuir | 151.3 | 55 |
| AC from Buxus wallichiana | EBT | 2 [5.9] | 1 | PSO | Freundlich | 383.0 | 74 |
| AC from Spartium junceum | EBT | — [7.2] | 0.6 | PSO | Langmuir | 261.4 | 75 |
| AC from Datura metel seeds | EBT | 2 [—] | 2 | PSO | Langmuir | 154.5 | 57 |
| AC from Rosa brunonii Lindl. seeds | EBT | 6–7 [7.9] | 2 | PSO | Langmuir | PAC: 192.3 | This work |
| CAC: 256.4 |
A comparative assessment of activated carbons reported in the literature for the adsorption of Disperse Orange and Eriochrome Black T dyes is summarized in Table 3. As shown, the activated carbons derived from Rosa brunonii Lindl. seeds in this work exhibit competitive and, in several cases, superior adsorption performance compared to previously reported biomass-based adsorbents. For DO dyes, the adsorption capacity achieved in this study is comparable to or higher than those reported for activated carbons prepared from rattan sawdust and cenosphere-based composites, while requiring a lower adsorbent dosage. More notably, for EBT adsorption, the prepared activated carbons demonstrate substantially higher maximum adsorption capacities, reaching 192.3 mg g−1 for PAC and 256.4 mg g−1 for CAC, outperforming many reported biomass-derived activated carbons under comparable or milder operating conditions. The superior performance of the present adsorbents can be attributed to their well-developed porous structure, high carbon content, and favorable surface chemistry, which collectively enhance dye–adsorbent interactions. These results highlight the effectiveness of Rosa brunonii Lindl. seed-derived activated carbons as promising and efficient materials for the removal of disperse and anionic dyes from aqueous systems.
4 Conclusions
This study demonstrates the successful valorization of Rosa brunonii Lindl. seeds into high-performance activated carbons prepared via physical and chemical activation routes and their effective application for the removal of hazardous dyes from aqueous media. Comprehensive structural and physicochemical characterization confirmed that thermal activation induces pronounced surface modification, pore development, and aromatization of the carbon framework. The prepared ACs reveal the formation of a predominantly amorphous to turbostratic carbon structure with high thermal stability up to 600 °C, accompanied by a substantial increase in carbon content to 93 at% and a marked reduction in oxygen content to about 5 at%. Chemical activation (CAC) produced a higher density of accessible surface functionalities, resulting in consistently enhanced adsorption performance relative to physically activated carbon (PAC), particularly for anionic dyes. Batch adsorption studies showed that dye uptake is governed by solution chemistry and material properties, including initial dye concentration, pH, and particle size. Equilibrium adsorption capacities reached 73.0 and 69.4 mg g−1 for DO1 on PAC and CAC, respectively, while significantly higher capacities were achieved for EBT, reaching 192.3 mg g−1 on PAC and 256.4 mg g−1 on CAC. The equilibrium data are best described by the Langmuir isotherm, indicating predominantly monolayer adsorption on relatively uniform surface sites, whereas the adsorption kinetics follow the pseudo-second-order model, reflecting strong dye–surface interactions. Adsorption was favoured at lower temperatures, consistent with an exothermic process. Solution pH played a critical role in EBT adsorption, with optimal uptake under acidic conditions due to electrostatic attraction, whereas DO1 adsorption remained largely pH independent owing to its weak ionization behavior. From a practical perspective, the use of an abundant, underutilized biomass precursor and straightforward activation procedures suggests favorable cost potential compared with conventional commercial activated carbons. Overall, the superior adsorption performance, favorable kinetics (reaching equilibrium during only 60 min), and low adsorbent dosage requirements position Rosa brunonii seed-derived activated carbons as sustainable, low-cost, and efficient materials for dye remediation.Author contributions
Muhammad Bilal: conceptualization, methodology, investigation, formal analysis, project administration, supervision, writing – original draft, writing – review & editing. Muhammad Idrees: methodology, investigation, formal analysis, writing – review & editing. Javed Ali: methodology, investigation, formal analysis, writing – review & editing. Amir Hossein Behroozi: conceptualization, methodology, investigation, formal analysis, project administration, supervision, writing – original draft, writing – review & editing. Noushad Hussain: methodology, investigation, formal analysis, writing – review & editing. Vahid Vatanpour: methodology, investigation, formal analysis, writing – review & editing. Tizazu H. Mekonnen: conceptualization, project administration, supervision, writing – review & editing.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The supporting data has been provided as part of the main manuscript and supplementary information (SI). Supplementary information: Fig. S1 is further experimental detail. See DOI: https://doi.org/10.1039/d5va00490j.Acknowledgements
The authors acknowledge and appreciate for financial support of Kohat University of Science and Technology, Kohat, KPK, Pakistan under ORIC Research and Development Fund Program (KUST/ORIC/22/2370). The authors also acknowledge the financial support of Natural Science and Engineering Research Council of Canada (NSERC). This research was conducted in part thanks to funding from the Canada Research Chairs Program.References
- Y. Liu, J. Chen, D. Duan, Z. Zhang, C. Liu, W. Cai and Z. Zhao, Environmental Impacts and Biological Technologies Toward Sustainable Treatment of Textile Dyeing Wastewater: A Review, Sustainability, 2024, 16, 10867 CrossRef CAS.
- A. Aghashiri, S. Hashemian and F. K. Fotooh, Adsorption of erochrom black T AzO dye by nanocomposite of activated carbon from sugar beet and LaFeO3, Desalin. Water Treat., 2021, 212, 173–184 CrossRef CAS.
- A. M. Jorge, K. Athira, M. B. Alves, R. L. Gardas and J. F. Pereira, Textile dyes effluents: A current scenario and the use of aqueous biphasic systems for the recovery of dyes, J. Water Process Eng., 2023, 55, 104125 CrossRef.
- S. Khan, T. Noor, N. Iqbal and L. Yaqoob, Photocatalytic dye degradation from textile wastewater: a review, ACS Omega, 2024, 9, 21751–21767 CrossRef CAS PubMed.
- S. I. Siddiqui, R. Ravi, G. Rathi, N. Tara, S. ul-Islam and S. A. Chaudhry, Decolorization of textile wastewater using composite materials, Nanomaterials in the Wet Processing of Textiles, 2018, pp. 187–218 Search PubMed.
- S. I. Siddiqui and S. A. Chaudhry, Nanohybrid composite Fe2O3-ZrO2/BC for inhibiting the growth of bacteria and adsorptive removal of arsenic and dyes from water, J. Cleaner Prod., 2019, 223, 849–868 CrossRef CAS.
- S. H. Hashemi and M. Kaykhaii, in Emerging Freshwater Pollutants, Elsevier, 2022, pp. 267–287, DOI:10.1016/B978-0-12-822850-0.00013-2.
- S. Benkhaya, S. M’rabet and A. El Harfi, Classifications, properties, recent synthesis and applications of azo dyes, Heliyon, 2020, 6, e03271 CrossRef CAS PubMed.
- K.-T. Chung, Azo dyes and human health: A review, J. Environ. Sci. Health, Part C, 2016, 34, 233–261 CrossRef CAS PubMed.
- R. A. E.-G. Mansour, M. G. Simeda and A. A. Zaatout, Removal of brilliant green dye from synthetic wastewater under batch mode using chemically activated date pit carbon, RSC Adv., 2021, 11, 7851–7861 RSC.
- L. Malinauskiene, E. Zimerson, M. Bruze, K. Ryberg and M. Isaksson, Sensitizing capacity of D isperse O range 1 and its potential metabolites from azo reduction and their cross-reactivity pattern, Contact Dermatitis, 2013, 69, 40–48 CrossRef CAS PubMed.
- B. Rietzler, A. P. Manian, D. Rhomberg, T. Bechtold and T. Pham, Investigation of the decomplexation of polyamide/CaCl2 complex toward a green, nondestructive recovery of polyamide from textile waste, J. Appl. Polym. Sci., 2021, 138, 51170 CrossRef CAS.
- J. K. Sahoo, R. K. Sukla, A. Pradhan, S. K. Sahoo and J. Rath, Removal of Eriochrome Black T dye using Plant Based Bio-Adsorbent: A Review, Lett. Appl. NanoBioScience, 2024, 13, 73 Search PubMed.
- S. Ali, Q. M. Hassan, C. Emshary and H. Sultan, Characterizing optical and morphological properties of Eriochrome Black T doped polyvinyl alcohol film, Phys. Scr., 2020, 95, 095814 CrossRef CAS.
- G. Bal and A. Thakur, Distinct approaches of removal of dyes from wastewater: A review, Mater. Today: Proc., 2022, 50, 1575–1579 CAS.
- M. Bilal, A. H. Behroozi, R. Jahan, J. Ali and V. Vatanpour, Sustainable TiO2/Diospyros texana seed-derived activated carbon composites for rapid photodegradation of oxytetracycline and amoxicillin under visible light, J. Chem. Technol. Biotechnol., 2025, 100, 2371–2382 CrossRef CAS.
- A. Pal, S. Das, S. R. Paul, A. Debnath, D. Datta, R. Bhoi and M. Yılmaz, Cost analysis and adsorption efficiency of chemically activated biochar from rubber wood sawdust for ciprofloxacin removal in environmental remediation, Biomass Convers. Biorefin., 2026, 16, 84 CrossRef CAS.
- K. Valizadeh, A. Bateni, N. Sojoodi, M. R. Ataabadi, A. H. Behroozi, A. Maleki and Z. You, Magnetized inulin by Fe3O4 as a bio-nano adsorbent for treating water contaminated with methyl orange and crystal violet dyes, Sci. Rep., 2022, 12, 22034 CrossRef CAS PubMed.
- F. M. Mohamed, M. R. El-Aassar, O. M. Ibrahim, A. Elsayed, M. F. Alrakshy, M. Abdel Rafea and K. A. Omran, Effective removal of carcinogenic azo dye from water using zea mays-derived mesoporous activated carbon, ACS Omega, 2024, 9, 13086–13099 CAS.
- A. Sharma, D. Mangla and S. A. Chaudhry, Recent advances in magnetic composites as adsorbents for wastewater remediation, J. Environ. Manage., 2022, 306, 114483 CrossRef CAS PubMed.
- D. H. da Silva Santos, Y. Xiao, N. Chaukura, J. M. Hill, R. Selvasembian, C. L. S. Zanta and L. Meili, Regeneration of dye-saturated activated carbon through advanced oxidative processes: A review, Heliyon, 2022, 8, e10205 CrossRef PubMed.
- J. Zhang, C. Duan, X. Huang, M. Meng, Y. Li, H. Huang, H. Wang, M. Yan and X. Tang, A review on research progress and prospects of agricultural waste-based activated carbon: preparation, application, and source of raw materials, J. Mater. Sci., 2024, 59, 5271–5292 CrossRef CAS.
- V. Mozhiarasi and T. S. Natarajan, Bael fruit shell–derived activated carbon adsorbent: effect of surface charge of activated carbon and type of pollutants for improved adsorption capacity, Biomass Convers. Biorefin., 2024, 14, 8761–8774 CrossRef CAS.
- N. Theamwong, W. Intarabumrung, S. Sangon, S. Aintharabunya, Y. Ngernyen, A. J. Hunt and N. Supanchaiyamat, Activated carbons from waste Cassia bakeriana seed pods as high-performance adsorbents for toxic anionic dye and ciprofloxacin antibiotic remediation, Bioresour. Technol., 2021, 341, 125832 CrossRef CAS PubMed.
- A. Kheddo, L. Rhyman, M. I. Elzagheid, P. Jeetah and P. Ramasami, Adsorption of synthetic dyed wastewater using activated carbon from rice husk, SN Appl. Sci., 2020, 2, 2170 CrossRef CAS.
- E. Altintig, B. Sarici, D. Bozdag, T. Over Ozcelik, M. Karakas and H. Altundag, Application of Optimization Response Surface for the Adsorption of Methylene Blue Dye onto Zinc-coated Activated Carbon, Environ. Monit. Assess., 2024, 196, 682 CrossRef CAS PubMed.
- A. Namane, A. Mekarzia, K. Benrachedi, N. Belhaneche-Bensemra and A. Hellal, Determination of the adsorption capacity of activated carbon made from coffee grounds by chemical activation with ZnCl2 and H3PO4, J. Hazard. Mater., 2005, 119, 189–194 CrossRef CAS PubMed.
- A. Lim, J. J. Chew, L. H. Ngu, S. Ismadji, D. S. Khaerudini and J. Sunarso, Synthesis, characterization, adsorption isotherm, and kinetic study of oil palm trunk-derived activated carbon for tannin removal from aqueous solution, ACS Omega, 2020, 5, 28673–28683 CrossRef CAS PubMed.
- S. Neusatz Guilhen, T. Watanabe, T. Tieko Silva, S. Rovani, J. Takehiro Marumo, J. Alberto Soares Tenório, O. Mašek and L. Goulart de Araujo, Role of point of zero charge in the adsorption of cationic textile dye on standard biochars from aqueous solutions: selection criteria and performance assessment, Recent Prog. Mater, 2022, 4, 1–30 CrossRef.
- L. R. Arenas, S. R. Gentile, S. Zimmermann and S. Stoll, Nanoplastics adsorption and removal efficiency by granular activated carbon used in drinking water treatment process, Sci. Total Environ., 2021, 791, 148175 CrossRef PubMed.
- S. Azizian and S. Eris, in Interface Science and Technology, Elsevier, 2021, vol. 33, pp. 445–509 Search PubMed.
- A. Bateni, K. Valizadeh, Y. Salahshour, A. H. Behroozi and A. Maleki, Fabrication and characterization of pectin-graphene oxide-magnesium ferrite-zinc oxide nanocomposite for photocatalytic degradation of diclofenac in an aqueous solution under visible light irradiation, J. Environ. Manage., 2022, 324, 116358 CrossRef CAS PubMed.
- H. Wang, R. Xie, J. Zhang and J. Zhao, Preparation and characterization of distillers' grain based activated carbon as low cost methylene blue adsorbent: Mass transfer and equilibrium modeling, Adv. Powder Technol., 2018, 29, 27–35 CrossRef CAS.
- P. Kaur, S. Kalpana, S. Kumar, A. Kumar and A. Kumar, Effect of various sodium hydroxide treatment parameters on the adsorption efficiency of rice husk for removal of methylene blue from water, Emergent Mater., 2023, 6, 1809–1824 CrossRef CAS.
- M. Mierzwa-Hersztek, K. Gondek, A. Nawrocka, H. Pinkowska, T. Bajda, J. Stanek-Tarkowska and M. Szostek, FT-IR analysis and the content of phenolic compounds in exogenous organic matter produced from plant biomass, J. Elementol., 2019, 24, 879–896 Search PubMed.
- J. K. Kabeya, N. K. Ngombe, P. K. Mutwale, J. B. Safari, G. G. Matlou, R. W. M. Krause and C. I. Nkanga, Antimicrobial capping agents on silver nanoparticles made via green method using natural products from banana plant waste, Artif. Cells Nanomed. Biotechnol., 2025, 53, 29–42 CrossRef CAS PubMed.
- N. Tolosa, K. Mendoza, D. L. Dumayas and J. M. D. De Silva, Preparation and characterization of activated carbon derived from Antidesma bunius L. in methylene blue removal from wastewater, J. Environ. Sci. Manage., 2020, 0119 Search PubMed.
- M. Tsarpali, J. N. Kuhn and G. P. Philippidis, Hydrothermal carbonization of residual algal biomass for production of hydrochar as a biobased metal adsorbent, Sustainability, 2022, 14, 455 CrossRef CAS.
- J. Zhuang, M. Li, Y. Pu, A. J. Ragauskas and C. G. Yoo, Observation of potential contaminants in processed biomass using fourier transform infrared spectroscopy, Appl. Sci., 2020, 10, 4345 CrossRef CAS.
- P. Feng, J. Li, H. Wang and Z. Xu, Biomass-based activated carbon and activators: preparation of activated carbon from corncob by chemical activation with biomass pyrolysis liquids, ACS Omega, 2020, 5, 24064–24072 CrossRef CAS PubMed.
- A. I. Bakti, P. L. Gareso and N. Rauf, Characterization of active carbon from coconut shell using X-ray diffraction (X-RD) and SEM-EDX techniques, JPFA, 2018, 8, 115–122 Search PubMed.
- H. Khalil, M. Jawaid, P. Firoozian, U. Rashid, A. Islam and H. M. Akil, Activated carbon from various agricultural wastes by chemical activation with KOH: preparation and characterization, J. Biobased Mater. Bioenergy, 2013, 7, 708–714 CrossRef.
- C. Călin, E.-E. Sîrbu, M. Tănase, R. Győrgy, D. R. Popovici and I. Banu, A thermogravimetric analysis of biomass conversion to biochar: experimental and kinetic modeling, Appl. Sci., 2024, 14, 9856 CrossRef.
- S. Szufa, M. Dzikuć, Ł. Adrian, P. Piersa, Z. Romanowska-Duda, W. Lewandowska, M. Marcza, A. Błaszczuk and A. Piwowar, Sustainable Drying and Torrefaction Processes of Miscanthus for Use as a Pelletized Solid Biofuel and Biocarbon-Carrier for Fertilizers, Molecules, 2020, 26, 1014 CrossRef PubMed.
- M. Amin, H. H. Shah, A. Iqbal, Z. U. R. Farooqi, M. Krawczuk and A. Zia, Conversion of waste biomass into activated carbon and evaluation of environmental consequences using life cycle assessment, Appl. Sci., 2022, 12, 5741 CrossRef CAS.
- T. S. Saleh, A. K. Badawi, R. S. Salama and M. M. M. Mostafa, Design and development of novel composites containing nickel ferrites supported on activated carbon derived from agricultural wastes and its application in water remediation, Materials, 2023, 16, 2170 CrossRef CAS PubMed.
- M. K. Rai, G. Shahi, V. Meena, R. Meena, S. Chakraborty, R. S. Singh and B. N. Rai, Removal of hexavalent chromium Cr (VI) using activated carbon prepared from mango kernel activated with H3PO4, Resour.-Effic. Technol., 2016, 2, S63–S70 Search PubMed.
- M. Abdu, S. Babaee, A. Worku, T. A. Msagati and J. F. Nure, The development of Giant reed biochar for adsorption of Basic Blue 41 and Eriochrome Black T. azo dyes from wastewater, Sci. Rep., 2024, 14, 18320 CrossRef PubMed.
- X. Zhang, Z. Wang, Y. Song, R. D. Tyagi and P. Drogui, in Decentralized Sanitation and Water Treatment, CRC Press, 2024, pp. 3–19 Search PubMed.
- D. Christian, A. Gaekwad, H. Dani, M. Shabiimam and A. Kandya, Recent techniques of textile industrial wastewater treatment: A review, Mater. Today: Proc., 2023, 77, 277–285 CAS.
- M. H. Abdel-Aziz, E. Z. El-Ashtoukhy, M. Bassyouni, A. F. Al-Hossainy, E. M. Fawzy, S. M. Abdel-Hamid and M. S. Zoromba, DFT and experimental study on adsorption of dyes on activated carbon prepared from apple leaves, Carbon Lett., 2021, 31, 863–878 CrossRef.
- Ö. Gerçel, H. F. Gerçel, A. S. Koparal and Ü. B. Öğütveren, Removal of disperse dye from aqueous solution by novel adsorbent prepared from biomass plant material, J. Hazard. Mater., 2008, 160, 668–674 CrossRef PubMed.
- Y. S. Al-Degs, M. I. El-Barghouthi, A. H. El-Sheikh and G. M. Walker, Effect of solution pH, ionic strength, and temperature on adsorption behavior of reactive dyes on activated carbon, Dyes Pigm., 2008, 77, 16–23 CrossRef CAS.
- M. Alkhabbas, A. M. Al-Ma'abreh, G. Edris, T. Saleh and H. Alhmood, Adsorption of anionic and cationic dyes on activated carbon prepared from oak cupules: Kinetics and thermodynamics studies, Int. J. Environ. Res. Public Health, 2023, 20, 3280 CrossRef CAS PubMed.
- M. Bansal, P. K. Patnala and T. Dugmore, Adsorption of Eriochrome Black-T (EBT) using tea waste as a low cost adsorbent by batch studies: A green approach for dye effluent treatments, Curr. Res. Green Sustainable Chem., 2020, 3, 100036 CrossRef.
- A. H. Behroozi, A. Mirahsani, P. Champagne and E. Koupaie, Protonated epichlorohydrin-crosslinked chitosan beads for ibuprofen removal from water: Insights into adsorption mechanisms, isotherms, kinetics, and reusability, J. Contam. Hydrol., 2025, 104786 Search PubMed.
- J. Ali, E. M. Bakhsh, N. Hussain, M. Bilal, K. Akhtar, T. M. Fagieh, E. Y. Danish, A. M. Asiri, X. Su and S. B. Khan, A new biosource for synthesis of activated carbon and its potential use for removal of methylene blue and eriochrome black T from aqueous solutions, Ind. Crops Prod., 2022, 179, 114676 CrossRef CAS.
- A. M. Aljeboree, A. N. Alshirifi and A. F. Alkaim, Kinetics and equilibrium study for the adsorption of textile dyes on coconut shell activated carbon, Arabian J. Chem., 2017, 10, S3381–S3393 CrossRef CAS.
- M. Açıkyıldız, A. Gürses, K. Güneş and E. Şahin, Adsorption of textile dyes from aqueous solutions onto clay: Kinetic modelling and equilibrium isotherm analysis, Front. Chem., 2023, 11, 1156457 CrossRef PubMed.
- B. M. Thamer, F. A. Al-Aizari and H. S. Abdo, Enhanced adsorption of textile dyes by a novel sulfonated activated carbon derived from pomegranate peel waste: isotherm, kinetic and thermodynamic study, Molecules, 2023, 28, 7712 CrossRef CAS PubMed.
- C. H. Pimentel, M. S. Freire, D. Gómez-Díaz and J. González-Álvarez, Preparation of activated carbon from pine (Pinus radiata) sawdust by chemical activation with zinc chloride for wood dye adsorption, Biomass Convers. Biorefin., 2023, 13, 16537–16555 CrossRef CAS.
- Y. El Maguana, N. Elhadiri, M. Benchanaa and R. Chikri, Activated carbon for dyes removal: modeling and understanding the adsorption process, J. Chem., 2020, 2020, 2096834 Search PubMed.
- J. Fito, M. Abewaa, A. Mengistu, K. Angassa, A. D. Ambaye, W. Moyo and T. Nkambule, Adsorption of methylene blue from textile industrial wastewater using activated carbon developed from Rumex abyssinicus plant, Sci. Rep., 2023, 13, 5427 CrossRef CAS PubMed.
- N. Mittal, M. Kaur and V. Singh, Adsorption studies on hydrophobic disperse dye using cellulose derived mesoporous activated carbon, Mater. Today: Proc., 2022, 62, 7595–7599 CAS.
- M. Kamau, S. Al-Najm and M. Nandeshwar, Recent advances in carbon-based nanocomposites for environmental remediation of industrial dyes, Compos. Interfaces, 2025, 1–34 Search PubMed.
- B. M. Choudhuri, K. Ramesh, A. Debnath and P. V. Chellam, Adsorptive removal of fluoroquinolones using bamboo culm biochar in a single and multi-component system, Biomass Convers. Biorefin., 2025, 1–12 Search PubMed.
- S. Rudra Paul, S. Das, A. Debnath and T. Kumar Misra, Boosted adsorptive removal of eosin yellow dye from aqueous solution with C–ZnO nanoparticles: optimization of process parameters, J. Dispersion Sci. Technol., 2026, 47, 128–144 CrossRef CAS.
- J. Rath, J. K. Sahoo, R. K. Sukla, A. Pradhan, S. K. Biswal, P. Dwivedy and S. K. Sahoo, Eriochrome Black T dye removal from an aqueous solution using graphene oxide-modified activated carbon materials, Indian Chem. Eng., 2024, 66, 573–592 CrossRef CAS.
- A. H. Behroozi, P. Champagne, T. H. Mekonnen and E. Koupaie, Selective removal of anionic contaminants via porous polyethylene glycol-templated chitosan aerogel beads, Sep. Purif. Technol., 2025, 136657 Search PubMed.
- M. Tiwari, S. Shukla and N. Dhiman, Characterization and adsorption of disperse dyes from wastewater onto cenospheres activated carbon composites, Environ. Earth Sci., 2017, 76, 702 CrossRef.
- A. Ahmad, B. Hameed and A. Ahmad, Removal of disperse dye from aqueous solution using waste-derived activated carbon: Optimization study, J. Hazard. Mater., 2009, 170, 612–619 CrossRef CAS PubMed.
- A. Khalil, C. Mangwandi, M. A. Salem, S. Ragab and A. El Nemr, Orange peel magnetic activated carbon for removal of acid orange 7 dye from water, Sci. Rep., 2024, 14, 119 CrossRef CAS PubMed.
- F. El Mansouri, G. Pelaz, A. Morán, J. C. E. Da Silva, F. Cacciola, H. El Farissi, H. Tayeq, M. H. Zerrouk and J. Brigui, Efficient removal of eriochrome black T dye using activated carbon of waste hemp (Cannabis sativa L.) grown in northern Morocco enhanced by new mathematical models, Separations, 2022, 9, 283 CrossRef CAS.
- M. Bilal, J. Ali, K. Bibi, S. B. Khan, M. Saqib, R. Saeed, R. Javeria, H. Khan, K. Akhtar and E. M. Bakhsh, Remediation of different dyes from textile effluent using activated carbon synthesized from Buxus Wallichiana, Ind. Crops Prod., 2022, 187, 115267 CrossRef CAS.
- A. Abiza, A. Reffas, H. Boubaker, R. Ben Arfi, D. Ghorbel, M. Rafique, G. L. Bachirou and A. Ghorbal, Comparative analysis of EBT dye removal using Spartium junceum and derived activated carbon: experimental and DFT insights, Int. J. Environ. Anal. Chem., 2024, 1–27 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |






