Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Performance of hydrophilic polyols in the formation of polyurethane based controlled release fertilizer coatings
Alex J.
Kosanovich
 ,
Omar
Jalife
,
Yasmin N.
Srivastava
,
Yi
Fan
and
Keith
Steelman
,
Omar
Jalife
,
Yasmin N.
Srivastava
,
Yi
Fan
and
Keith
Steelman
 *
*
Dow Chemical, Polyurethanes Business, 230 Abner Jackston Pkwy, Lake Jackson, TX, USA. E-mail: dksteelman@dow.com; yfan5@dow.com
First published on 6th February 2026
Abstract
A series of controlled-release coatings were developed for granular urea (46-0-0) using a layer-by-layer polyurethane system composed of polyether polyol blends, polymeric methylene diphenyldiisocyanate (pMDI), and a wax additive. A coating formulation utilizing a hydrophobic poly(propylene oxide) triol (P700) served as a baseline for release performance. Surprisingly, hydrophilic poly(ethylene oxide)-based diols and triols, when blended with approximately 20% glycerol, yielded coatings with improved crosslink density and moderated curing kinetics. Notably, glycerol inclusion did not enhance P700 performance, but its combination with poly(ethylene oxide)-based diol resulted in a synergistic improvement in release control for the coating formulations. These hydrophilic blended systems not only achieved excellent controlled-release behavior but also outperformed the hydrophobic P700-based coatings. This study demonstrates that hydrophilic polyols, when properly formulated, can rival or exceed the performance of traditional hydrophobic systems, offering new design strategies for environmentally responsive and tunable nutrient release coatings.
Environmental significanceThe United Nations sustainable development goals (SDG) define a set of targets which seek to better human life while enhancing environmental stewardship. This work which focuses on the usage of hydrophilic species for enhanced controlled release fertilizer coatings at low weights directly contributes to SDG #2 focused on ending hunger globally. The development of greater efficiency controlled release fertilizer coatings helps to enhance crop growth outputs while greatly reducing fertilizer waste and deleterious environmental effects that can come from uncontrolled release and loss of fertilizer into the environment. |
Introduction
The use of land for agriculture not only provides food to an ever-growing population but offers the opportunity for continuing innovation from which greater efficiencies are achieved. The United Nations maintains a set of Sustainable Development Goals (SDG), of which SDG #2 sets out aims to end global hunger.1 To meet product volume demands under cost-constrained conditions, farmers leverage the usage of natural and synthetic organic and inorganic fertilizers to match crop nutritional needs. Urea (46-0-0) is a primary nitrogen source used for commodity crops, is 46 wt% nitrogen, and its water solubility allows its simple uptake by crops through application to the soil. This solubility necessitates that urea (46-0-0) is metered out either multiple times or blended with other nutrients to avoid undesirable environmental impacts such as root burn or leaching.2 This need can increase labor costs and time for the farmers who use it.When a thin polymeric barrier material is applied to the surface of a fertilizer granule, the rate of solubilization, diffusion, and nutrient availability becomes limited.3 This coated, controlled release fertilizer (CRF) can achieve tuneable release across a desirable time frame through modification of polymer chemistry, coating conditions, material properties, reactivity, coating weight, and temperature.4 While laboratory conditions are closely monitored, real-life applications require that the coated fertilizer must contend with environmental variability including changing seasons, temperatures, and weather events.5 Such unpredictability necessitates that the polymer coating chemistry chosen is not only robust, but also resistant to changing conditions. Coatings of urea (46-0-0) must be of sufficient quantity and cure to provide uniform coverage of the prill, and their release performance in water is thought to be determined additionally by temperature, crosslink density, hydrophobicity and reactivity in the coating process.6 Multiple chemistries have been utilized for the controlled release of urea including molten sulfur,7 urea-formaldehyde,8 wax,9 polyolefins, and most successfully, polyurethanes.10 Polyurethanes are generated by the direct reaction of a hydroxyl-terminated species, or polyol, with an isocyanate-terminated component, or polyisocyanate. Where the functionality of the polyol and/or isocyanate is greater than 2, crosslinking of the polymeric network occurs, forming a thermoset coating. Various hydrophobic polyols such as bio-based castor oil11 and poly(propylene oxide) triols have been used for reaction with polyisocyanates, such as polymeric methylene diphenyldiisocyanate (pMDI).12 Additional isocyanates such as isophorone diisocyanate (IPDI) and 1,6-hexamethylene diisocyanate (HDI) have been used in polyurethane coating formulations for various applications.13 The reaction to form a urethane/carbamate can be performed directly but is often catalyzed either by organometallic (Zn),14 main group (Sn/Bi), or tertiary amine (triethylene diamine/1,8-diazabicyclo (5.4.0)undec-7-ene) catalysts.15 Additionally, an interstitial hydrophobic barrier material with a low melting point (<60 °C) can be utilized to enhance the conformal nature of the polymer coating. Often this material is a wax additive.16
This work utilizes layer-by-layer coating of polyurethane and a wax additive under standardized process conditions to generate controlled release fertilizers from urea (46-0-0). Counter to prior work which often leverages hydrophobic components17 this study uses hydrophilic blends constituted by poly(ethylene oxide) polyols of di- and trifunctionality in concert with glycerol and triethanolamine (TEOA) as a reactive amine catalyst, to react directly with polymeric methylene diphenyl diisocyanate (pMDI) and form the polyurethane barrier which provides enhanced controlled release performance. Structure–property relationships are established which show that the reactivity of poly(ethylene oxide) polyols paired with the greater crosslink density imbued by their mixture with glycerol provide synergistic effects which appear to overcome prior expectations that hydrophobicity should dominate controlled release efficiency.
Experimental
Materials
All chemical components were used as received. Glycerol (99.5%) was purchased from Sigma Aldrich. Poly(ethylene oxide) polyols (PEG-400, TRI-625) and poly(propylene oxide) triol (P700) were obtained from the Dow Chemical Company and are available as CARBOWAX™ products and VORANOL™ 2070 polyols, respectively. Polymeric methylenediphenyldiisocyanate (pMDI) was obtained from the Dow Chemical Company and is available as PAPI™ 27 polymeric MDI (PAPI™ 27 pMDI). Triethanolamine (TEOA, 99%) was obtained from the Dow Chemical Company. C30+HA wax granules were purchased from the Chevron Phillips Chemical Company. Urea (46-0-0) granules (SGN 250) were purchased from American Plant Food Corp.Coating procedure
Urea prills (1.0 kg) were stored at 50 °C for 2–24 h prior to running the coating experiments. Using gloves for hot material handling, 1 kg of the prills were added to the coater (see SI for coater geometry) and rotation was initiated at 40 rpm. An externally placed heat gun was used to heat and maintain the temperature of the rotating prills at 80 °C, and an IR temperature probe was utilized to ensure temperature stability prior to addition of coating materials. Once the temperature was stabilized, the coating was applied as three layers in layer-by-layer fashion where each of the first two polyurethane layers were followed by addition of a wax layer, with a third, final polyurethane application as the outermost layer. Following application of the coating, the granules were allowed to cool to below 50 °C, whereupon the wax additive resolidified and free flowing granule behavior was observed. The coated prills were then removed from the coater and allowed to sit for 7 days under ambient conditions prior to assessment of release performance.Analysis of urea release performance
After a minimum of 7 days post-coating, the urea release profiles were measured by taking 10 g of coated material and placing it in a jar with 100 mL of DI H2O and kept at room temperature. The refractive index of a small aliquot was collected after 14, 28, 56, and 84 days, or until >95% urea release was measured (see SI for full details).Measuring reactivity of coating formulations
Into a 250 mL Flakteck cup was weighed polyether polyol blend containing 10 wt% triethanolamine (TEOA) and PAPI™ 27 pMDI to achieve an isocyanate index of 140 with a mass of 50 g total. This was mixed at 2300 rpm for 30 seconds. The resultant mixture was then poured directly into a 250 mL plastic tripour and the viscosity recorded using a BYK BYKO-VISC DS viscometer. Once viscosity had reached 4000 cPs, the time was recorded, the mixture was removed, and the spindle was then cleaned with ethyl acetate and dried before the next measurements. Shortly after reaching 4000 cPs, the mixture would often begin to cure quickly, making removal and cleanup difficult.Results & discussion
The ultimate release efficiency of coated urea substrates is strongly influenced by coating process parameters such as coating weight, timing of additions, temperature, rotation frequency, and the coater's angle of repose. These factors directly impact uniformity, thickness at the substrate surface, and batch cycle times for production. With process conditions being held constant, the chemical composition of the polyurethane coating can be refined by carefully selecting the isocyanate and polyol components. This allows for precision tuning of the cure rate, hydrophobicity, and crosslink density which are critical to achieving controlled nutrient release. Typically, the release from the coated, water-soluble granule occurs in stages: water first saturates the coating shell and subsequent solvation of the urea (46-0-0) core is initiated. Nutrient release then proceeds via solute diffusion through the crosslinked polymeric structure.18 In a well-applied, defect-free polyurethane coating, this process occurs gradually and fosters release of the urea at a controlled, consistent rate.Chemical composition of coatings
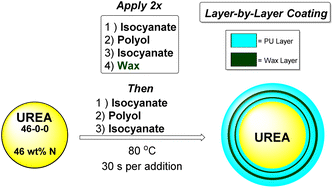
Layer-by-layer application of the coating formulation was undertaken at 80 °C in a rotating steel drum equipped with bevels (see SI). To ensure curing of the thermoset polyurethane coating and maintain usable molar ratios, each polyol was mixed with 10 wt% of triethanolamine (TEOA) as a reactive catalyst. The polyisocyanate was additionally added to the coater in two portions (40% and 60% of total for selected formulation) with the full amount of isocyanate-reactive polyol/TEOA blend added in between to form the polyurethane layer (Scheme 1).While other polyisocyanates such as toluene diisocyanate (TDI), 1,6-hexamethylene diisocyanate (HDI), and even 4,4′-methylene diphenyldiisocyanate (MDI) could be utilized to form a polyurethane coating, for this study PAPI™ 27 polymeric methylenediphenyldiisocyanate (pMDI) of 2.7 average isocyanate functionality, was utilized for its low viscosity, low volatility, and high functionality providing better processibility and safety considerations in handling. Prior studies were performed to optimize the coating conditions for layer-by-layer application of a controlled release coating of urea, and were therefore maintained in this study, including an isocyanate index19 of 140 to provide an optimal coating. Three polyurethane layers were applied, with two wax layers placed between each, for five total layers. The purpose of the wax additive is to bolster minimum hydrophobic performance and promote coating uniformity across the surface of the prill. For this purpose, a granular C30 wax with a melting range of ca. 50–60 °C was used. For the majority of the coatings, a total coating weight of 3.2 wt% (2.7 wt% PU/0.5 wt% wax) was applied to each granule. The usage of a minimal coating weight provides protection to the prill from solvation while preserving nitrogen content in the granule. The general chemical structures of the chemically reactive components used are shown in Scheme 2.
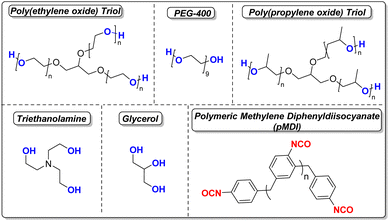 | ||
| Scheme 2 Chemical structures of isocyanate-reactive components and polymeric MDI used to generate polyurethane fertilizer coatings for this study. | ||
For this study, polyurethane coatings were generated from poly(ethylene oxide)-derived diol and triol both with and without inclusion of glycerol as a small molecular weight, trifunctional polyol expected to increase crosslink density of the isocyanate-reactive blend. A poly(propylene oxide) triol was also utilized to provide a hydrophobic component as a comparative example for controlled release performance. It is generally expected and was shown by prior work that once complete prill coverage is achieved, hydrophobicity in the isocyanate-reactive component proves a dominating variable for slowed release performance. Once a requisite amount of hydrophobicity is achieved, significant performance enhancements can be achieved through the increase of crosslink density in the polyurethane coating.20 While poly(propylene oxide) triols provide suitable hydrophobicity and coverage under chosen coating conditions, their reactivity is significantly less than that of poly(ethylene oxide)-derived polyols. The origin of this reactivity difference is well known in polyurethane chemistries and comes from the majority of poly(propylene oxide) endgroups being secondary hydroxyls, and therefore sterically encumbered.21 In the case of homopolymers of ethylene oxide, there are only primary hydroxyl endgroups, and therefore significantly increased reactivity is expected.
Coating formulations and calculated crosslink density
Each coating formulation chosen was applied to 1 kg of the urea (46-0-0) prills in layer-by-layer fashion using a rotating steel coater and performed at 80 °C. The components and their requisite amounts are shown below in Table 1. Generally, for a 1 kg sample 2.7 wt% (27 g total) of the PU coating was applied, with an additional 0.5 wt% (5 g) of the wax additive, for a total 3.2 wt% (32 g) coating. A slight variation was performed in Coating D2 where 3.0 wt% (30 g total) of polyurethane was added with the standard 0.5 wt% (5 g) of wax additive, to provide a thicker coating at 3.5 wt% (35 g total) total using the same isocyanate index and general ratios as Coating D1. This was done to determine the effect of a heavier coating weight on controlled release performance of the hydrophilic poly(ethylene oxide) triol, TRI-625. In each case, the triethanolamine (TEOA) was pre-blended with all other isocyanate-reactive components prior to layer-by-layer addition in the coating apparatus.| Component | Coating formulation code | |||||||
|---|---|---|---|---|---|---|---|---|
| A | B | C | D1 | D2 | E | F | G | |
| P700 | 11.66 g | 6.41 g | ||||||
| Glycerol | 3.65 g | 1.60 g | 1.66 g | 1.82 g | ||||
| PEG-400 | 10.93 g | 5.90 g | ||||||
| TRI-625 | 11.18 g | 12.42 g | 5.47 g | |||||
| Triethanolamine | 1.30 g | 0.41 g | 1.22 g | 1.24 g | 1.38 g | 0.89 | 0.84 g | 0.81 g |
| PAPI™ 27 pMDI | 14.04 g | 22.94 g | 14.85 g | 14.58 g | 16.20 g | 18.10 g | 18.60 g | 18.90 g |
| C30+HA wax | 5.0 g | 5.0 g | 5.0 g | 5.0 g | 5.0 g | 5.0 g | 5.0 g | 5.0 g |
| PU wt% | 2.7% | 2.7% | 2.7% | 2.7% | 3.0% | 2.7% | 2.7% | 2.7% |
| Wax wt% | 0.5% | 0.5% | 0.5% | 0.5% | 0.5% | 0.5% | 0.5% | 0.5% |
| Total coating wt% | 3.2% | 3.2% | 3.2% | 3.2% | 3.5% | 3.2% | 3.2% | 3.2% |
To further establish and support observable structure–property relationships in the controlled release coatings, the theoretical crosslink density of each formulation was calculated using a known, modified formula which relates equivalent weight and functionality (Table 2) with the total mass of all components added (see SI). The coatings and their calculated, theoretical crosslink densities are shown in Table 3.
| Component | Equivalent weight | Functionality |
|---|---|---|
| P700 | 233 g mol−1 | 3 |
| Glycerol | 30.7 g mol−1 | 3 |
| TRI-625 | 209 g mol−1 | 3 |
| PEG-400 | 200 g mol−1 | 2 |
| Triethanolamine | 50 g mol−1 | 3 |
| PAPI™ 27 pMDI | 134 g mol−1 | 2.7 |
| Coating | Calculated crosslink density |
|---|---|
| A | 2.75 mmol g−1 |
| B | 4.46 mmol g−1 |
| C | 1.88 mmol g−1 |
| D | 2.86 mmol g−1 |
| E | 3.55 mmol g−1 |
| F | 3.11 mmol g−1 |
| G | 3.71 mmol g−1 |
The usage of trifunctional isocyanate-reactive components such as P700, glycerol, TRI-625 and triethanolamine brings inherently high crosslink density in reaction with the nominally 2.7 functional PAPI™ 27 pMDI for Coating A (2.75 mmol g−1), Coating B (4.46 mmol g−1), and Coating D (2.86 mmol g−1). The usage of the poly(ethylene oxide) diol, PEG-400, in Coating C results in a very low relative crosslink density (1.88 mmol g−1) due to the difunctional nature of the polyether polyol providing no crosslinking contributions. However, the inclusion of glycerol in small amounts (ca. 20 wt%) in the polyol blend results in a large increase in theoretical crosslink density for the polyurethane, which may be expected to provide enhanced controlled release performance. In Coating E a crosslink density of 3.55 mmol g−1 from the combination of P700 with glycerol results in an increase of 29% versus Coating A (2.75 mmol g−1), however it should be noted that the blend itself shows visual phase separation due to the incompatibility of hydrophobic P700 with hydrophilic glycerol. Coating F using a homogeneous glycerol blend of PEG-400 shows a 65% increase in calculated crosslink density with a value of 3.11 mmol g−1versus Coating C (1.88 mmol g−1). Coating G with a similarly homogeneous blend of TRI-625 and glycerol provides a calculated crosslink density of 3.71 mmol g−1 which is 30% higher than Coating D with a calculated crosslink density of 2.86 mmol g−1 using only TRI-625. The amount of glycerol chosen for inclusion was maintained such that an increase in theoretical crosslink density could be achieved, while a physically suitable amount of isocyanate-reactive blend could be applied to provide prill coverage for uniform coating under standardized process conditions for this study.
Comparative reactivity of the polyurethane formulations
Triethanolamine (TEOA) is added to the system in 10 wt% inclusion to achieve multiple goals in its role as a reactive, non-leaching catalyst which promotes the reaction of polyol with polyisocyanate to form the urethane layer on kinetically relevant timescales while autocatalytically crosslinking itself into the polymeric network. The speed of polymer formation and subsequent curing of the discrete layers at the surface of the urea (46-0-0) prill is of the utmost importance for reducing batch cycle times and improving volume efficiencies in production. Additionally, once the formulation has completely covered the prill in liquid form it is paramount that it achieves a quick, tack-free cure, which is promoted by the inclusion of TEOA in the polyol system. If the balance of curing kinetics is too slow, this can result in vulnerable coatings which may remain tacky or delaminate from the surface under handling conditions. On the other hand, if the cure is too fast, then there is pooling which can occur in any surface morphological features, leading to non-uniform coatings which fail to provide any protection to the highly water-soluble urea (46-0-0) substrate. Either of these imbalances can lead to increased scrap rates equating to loss of product and value.Reactivity tests for the formulations were performed by measuring the viscosity build of the freshly made bulk mixture in a plastic tripour with PAPI™ 27 pMDI at an isocyanate index of 140. Once the viscosity of the mixture reached 4000 cPs, the time was recorded and the cup removed to avoid destruction of the spindle. This reactivity data is shown in Table 4 below.
| Coating | Polyol | Polyol HEW | TEOA wt% | TEOA mol% | Mol% N | Time @ 4000 cPs |
|---|---|---|---|---|---|---|
| A | P700 | 233 g mol−1 | 10% | 34.2 mol% | 11.4 mol% | 225 s |
| B | Glycerol | 30 g mol−1 | 10% | 6.4 mol% | 2.1 mol% | No cure |
| C | PEG-400 | 200 g mol−1 | 10% | 30.8 mol% | 10.3 mol% | n.d. |
| D | TRI-625 | 208 g mol−1 | 10% | 31.8 mol% | 10.6 mol% | 25 s |
| F | PEG-400/glycerol | 90.4 g mol−1 | 10% | 16.7 mol% | 5.6 mol% | 110 s |
| G | TRI-625/glycerol | 85 g mol−1 | 10% | 15.9 mol% | 5.3 mol% | 170 s |
The reactivity of the hydrophilic formulations based on poly(ethylene oxide) were compared to that of the hydrophobic formulation using poly(propylene oxide) triol, P700. At 10 wt% inclusion of TEOA (HEW = 50 g mol−1), the mol% of TEOA was calculated using the hydroxyl equivalent weight (HEW) of the polyether polyol as determined by OH#. The reactivity of the PEG-400 used on its own (Coating C) was unable to be determined by viscometry in the bulk state, as foaming side reactivity consistently occurred due to moisture content. However, this observed reactivity was not deleterious in the formation of the thin coating when applied in layer-by-layer fashion. Additionally, the reactivity of glycerol on its own (Coating B) with 10 wt% TEOA was unable to be determined as no cure, and little viscosity buildup was observed even after 600 s. This is due to the low HEW of glycerol as a small molecule, and therefore 10 wt% of TEOA provides only 6.4 mol% of TEOA and ca. 2.1 mol% catalytic nitrogen content which is unsuitable for curing kinetics. The reactivity of the inhomogeneous P700/Glycerol (Coating E) blend was omitted due to lack of difference in controlled release performance. However, the TRI-625 formulation (Coating D) with 10 wt% TEOA had 31.8 mol% TEOA based on the equivalent weight of the pure TEOA triol, of which 1/3rd is catalytically active nitrogen (10.3 mol%) and provided a quick rise in viscosity to 4000 cPs in 25 s. Due to the presence of only primary hydroxyls, this fast reactivity is to be expected. The P700 formulation (Coating A) with a greater molar amount of TEOA (34.2 mol%) and catalytically active nitrogen content (11.4 mol%) provided a much longer time to reach 4000 cPs of 225 s. Despite the larger amount of TEOA reactive catalyst and similar functionality, the presence of mostly secondary hydroxyl endgroups leads to a slower observed reactivity. However, while observing the P700 performance in controlled release coatings, Coating A shows greater benefit than the faster reactivity TRI-625 based Coating D. When a homogeneous blend of glycerol and the poly(ethylene oxide) polyols is utilized as the isocyanate-reactive component, an interesting shift in reactivity rate commensurate with heightened controlled release performance occurs. The inclusion of glycerol appears to significantly slow the kinetics of the poly(ethylene oxide) triol TRI-625 with the TRI-625/glycerol blend (Coating G) having 15.9 mol% TEOA and 5.3 mol% nitrogen, yet still reaching a viscosity of 4000 cPs at 170 s. Despite having nearly half the molar amount of TEOA as the P700 example (Coating A), the all-primary hydroxyl nature of the majority poly(ethylene oxide) triol component appears to allow for faster reactivity. Additionally, the PEG-400/glycerol blend (Coating F) showed good curing activity at similar TEOA content (16.7 mol%) and nitrogen content (5.6 mol%), reaching 4000 cPs after 110 s. The observed moderated reactivity matched with the increased theoretical crosslink density from homogeneous glycerol inclusion in the poly(ethylene oxide)-based polyols appear to allow for the right balance of coating uniformity and cure kinetics to achieve the enhanced controlled release performance as observed.
Controlled release performance of polyurethane coatings
Following layer-by-layer application of the polyurethane coating to urea (46-0-0), the prills were removed from the rotating coater and set aside at room temperature for a period of 7 days to allow for complete cure. Subsequently, 10 g of material was placed in a glass jar with 100 g of DI H2O and covered with a screw cap lid. Aliquots were taken at predetermined time points (14 days, 28 days, 56 days) and tested via refractive index (R. I.) to determine the amount of urea substrate which had released (see SI for full details). Table 5 shows the general composition and release performance of Coatings A–G which are depicted graphically in Fig. 1.| Coating | Polyol 1 | Polyol 2 | PU | Wax | Urea release | ||
|---|---|---|---|---|---|---|---|
| 14 days | 28 days | 56 days | |||||
| A | P700 | — | 2.7 wt% | 0.5 wt% | 24% | 49% | 76% |
| B | Glycerol | — | 2.7 wt% | 0.5 wt% | >95% | — | — |
| C | PEG-400 | — | 2.7 wt% | 0.5 wt% | 70% | >95% | — |
| D1 | TRI-625 | — | 2.7 wt% | 0.5 wt% | 30% | 64% | >95% |
| D2 | TRI-625 | — | 3.0 wt% | 0.5 wt% | 20% | 38% | 90% |
| E | P700 | Glycerol | 2.7 wt% | 0.5 wt% | 30% | 54% | 71% |
| F | PEG-400 | Glycerol | 2.7 wt% | 0.5 wt% | 15% | 31% | 73% |
| G | TRI-625 | Glycerol | 2.7 wt% | 0.5 wt% | 10% | 34% | 75% |
Coating A utilized P700, a hydrophobic 700 g per mol poly(propylene oxide)triol with similar hydroxyl equivalent weight (HEW) to the other polyols used in this study to generate a benchmark polyurethane formulation that has been previously reported.4 Coating A provided suitable performance with a release of 24% after 14 days, 49% after 28 days, and 76% after 56 days. Glycerol, such as is reported in Coating B provides a formulation which fails on its own and was previously shown to do so.22 Additionally, Coating C, which utilizes a hydrophilic poly(ethylene glycol) diol, PEG-400 as its isocyanate-reactive component provides unsuitable release performance with 70% release after 14 days and >95% release at 28 days. Coating D1 utilizes a 625 g per mol poly(ethylene oxide)triol, TRI-625, as the isocyanate-reactive component and with its increased functionality provides a moderate release performance of 30% at 14 days, 64% at 28 days, and >95% after 56 days. Even increasing the total weight of the TRI-625 based coating from 2.7 wt% to 3.0 wt% in D2 only moderately improves release at earlier times with 20% at 14 days, 54% after 28 days, and 90% after 56 days. Despite the increase in thickness of the coating, the ultimate performance of the hydrophilic TRI-625 based coating still falls short of Coating A using hydrophobic P700 at only 2.7 wt%. The initial performance of Coating D1 remains surprising providing controlled release performance with an entirely hydrophilic polyol blend. While hydrophobicity is a dominating factor in the ability of polyurethane coatings to impart controlled release, crosslink density is also considered an important factor. Glycerol on its own did not provide any performance benefits, but it remained of interest to determine if glycerol addition to existing polyols might increase crosslink density and therefore boost performance. Coating E used a 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 blend of the hydrophobic poly(propylene oxide) triol P700 and glycerol, however, the blend itself showed physical phase separation even after speedmixing. Despite this increased theoretical crosslink density, the addition of glycerol to P700 did not prove beneficial as Coating E gave 30% release at 14 days, 54% release at 28 days, and 71% release after 56 days. Thus, at earlier times, Coating E containing glycerol and P700, showed degraded performance versus Coating A, where P700 was used on its own. It is suspected the inhomogeneity of the P700/glycerol blend contributed to a lack of benefit. Furthermore, when glycerol was blended with either PEG-400 or TRI-625 in similar portions for Coating F and Coating G, a clear homogeneous blend was achieved, and in fact effects of the theoretically increased crosslink density were borne out synergistically in the release performance of Coating F using PEG-400/glycerol giving 15% release at 14 days, 31% release at 28 days, and 73% release at 56 days. Additionally, Coating G which employed a TRI-625/glycerol blend also provided a step-change in release performance with 10% after 14 days, 34% after 28 days, and 73% after 56 days.
20 blend of the hydrophobic poly(propylene oxide) triol P700 and glycerol, however, the blend itself showed physical phase separation even after speedmixing. Despite this increased theoretical crosslink density, the addition of glycerol to P700 did not prove beneficial as Coating E gave 30% release at 14 days, 54% release at 28 days, and 71% release after 56 days. Thus, at earlier times, Coating E containing glycerol and P700, showed degraded performance versus Coating A, where P700 was used on its own. It is suspected the inhomogeneity of the P700/glycerol blend contributed to a lack of benefit. Furthermore, when glycerol was blended with either PEG-400 or TRI-625 in similar portions for Coating F and Coating G, a clear homogeneous blend was achieved, and in fact effects of the theoretically increased crosslink density were borne out synergistically in the release performance of Coating F using PEG-400/glycerol giving 15% release at 14 days, 31% release at 28 days, and 73% release at 56 days. Additionally, Coating G which employed a TRI-625/glycerol blend also provided a step-change in release performance with 10% after 14 days, 34% after 28 days, and 73% after 56 days.
Conclusions
Granular urea (46-0-0) was coated in layer-by-layer fashion with selected polyether polyol blends, polymeric methylene diphenyldiisocyanate, and a wax additive to provide a tack-free, controlled release polyurethane coating. A hydrophobic poly(propylene oxide) triol, P700, was utilized as an isocyanate-reactive component to provide a baseline of controlled release performance. Counter to the expected factors of hydrophobicity which generally dominate controlled release coatings, hydrophilic blends based on poly(ethylene oxide) diol and triol were successfully shown to provide access to moderate controlled release performance. When these hydrophilic blends were made with ca. 20% glycerol inclusion to improve crosslink density and moderate curing kinetics, a better performing controlled release coating was furnished which outperformed the hydrophobic coating based on P700, all while using entirely hydrophilic isocyanate-reactive components. While glycerol inclusion failed to improve the performance of the P700 polyol, it was found that the homogeneous blend formed with either PEG-400 or TRI-625 led to an adventitious and synergistic step-change in controlled release performance, showing that coatings made from hydrophilic polyols could be successful with properly modified crosslink density and reactivity profiles.Conflicts of interest
There are no conflicts of interest to declare.Abbreviations
| PU | Polyurethanes |
| pMDI | Polymeric methylene diphenyldiisocyanate |
Data availability
All available data is provided in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5va00459d.Acknowledgements
All studies were funded by the Dow Chemical Company, Polyurethanes Business. The authors would like to acknowledge the Dow Polyurethanes Business, Yuri Alencar and Yin Tang for support of this work.References
- https://www.ungeneva.org/en/about/topics/sustainable-development-goals .
- S. Savci, Investigation of Effect of Chemical Fertilizers on Environment, APCBEE Procedia, 2012, 1, 287–292 CrossRef.
- F. Versino, M. Urriza and M. Garcia, Eco-compatible Cassava Starch Films for Fertilizer Controlled-Release, Int. J. Biol. Macromol., 2019, 134, 302–307 CrossRef.
- A. J. Kosanovich, P. Agarwal, J. C. Medina, Y. N. Srivastava, M. J. Cotanda Santapau and Y. Fan. Coated Fertilizer Granule Compositions, US20250019321A1, 2025.
- A. J. Messiga, K. Dyck, K. Ronda, K. van Baar, D. Haak, S. Yu and M. Dorais, Nutrients Leaching in Response to Long-Term Fertigation and Broadcast Nitrogen in Blueberry Production, Plants, 2020, 9, 1530 CrossRef PubMed.
- Y. N. Srivastava, A. J. Kosanovich, Y. Fan, D. G. Abebe, P. Boopalachandran, K. Pal and J. C. Medina, Coated Fertilizer Granule Compositions, US20250197311A1, 2025.
- F. Eghbali Babadi, R. Yunus, S. Abdul Rashid, M. A. Mohd Salleh and S. Ali, New Coating Formulation for the Slow Release of Urea Using a Mixture of Gypsum and Dolomitic Limestone, Particulogy, 2015, 23, 62–67 CrossRef.
- V. Glasser, P. Stajer, J. Vidensky, P. Svandova and V. Knor, Urea-formaldehyde resins as packaging materials for industrial fertilisers with protracted action. Part 4, Int. Polym. Sci. Technol., 1987, 14, 127 Search PubMed.
- Y.-C. Yang, M. Zhang, Y. Li, X.-H. Fan and Y.-Q. Geng, Improving the quality of polymer-coated urea with recycled plastic, proper additives, and large tablets, J. Agric. Food Chem., 2012, 60, 11229–11237 CrossRef.
- R. Gil-Ortiz, M. Á. Naranjo, A. Ruiz-Navarro, M. Caballero-Molada, S. Atares, C. García and O. Vicente, New Eco-Friendly Polymeric-Coated Urea Fertilizers Enhanced Crop Yield in Wheat, Agronomy, 2020, 10, 438 Search PubMed.
- G. Dong Feng, Y. Ma, M. Zhang, P. You Jia, L. Hong Hu, C. Guo Liu and Y. Hong Zhou, Polyurethane-coated urea using fully vegetable oil-based polyols: Design, nutrient release and degradation, Prog. Org. Coat., 2019, 133, 267–275 Search PubMed.
- I. Kassem, E.-H. Ablouh, F.-M. El Bouchtaoui, M. Jaouahar and M. El Achaby, Polymer Coated Slow/Controlled Release Granular Fertilizers: Fundamentals and Research Trends, Prog. Mater Sci., 2024, 144, 101269 CrossRef.
- G. Wang, K. Li, A. Hu, C. Hu, Y. Zhu, C. Chen, G. Guo, A. Yang, R. Drumright and J. Argyropoulos, Synthesis of HDI/IPDI Hybrid Isocyanurate and its Application in Polyurethane Coating, Prog. Org. Coat., 2015, 78, 225–233 CrossRef.
- L. Ruiduan, L. Ling, L. Yanjie, W. Ben, Y. Jia Jun and Z. Jibo, Research Progress of Amine Catalysts for Polyurethane, Topics in Chemical & Material Engineering, 2018, vol. 1, pp. 54–57 Search PubMed.
- N. Malwitz, S.-W. Wong, K. C. Frisch and P. A. Manis, Amine Catalysis of Polyurethane Foams, J. Cell. Plast., 1987, 23(5), 461–502 Search PubMed.
- B. Su, M. Yang, B. Gao, Z. Li, X. Zhao, S. Zhang, D. Cheng, T. Shen, Y. Yao and Y. Tang, Ceresing Wax Enhances Hydrophobicity and Density of Bio-based Polyurethane of Controlled-Release Fertilizers, J. Cleaner Prod., 2024, 452, 142145 CrossRef.
- C. Dai, L. Yang, J. Xie and T.-J. Wang, Nutrient diffusion control of fertilizer granules coated with a gradient hydrophobic film, Colloids Surf., A, 2020, 588, 124361 CrossRef.
- D. Lawrencia, S. K. Wong, D. Y. S. Low, B. H. Goh, J. K. Goh, U. R. Ruktanonchai, A. Soottitantawat, L. H. Lee and S. Y Yang, Controlled Release Fertilizers: A Review on Coating Materials and Mechanism of Release, Plants, 2021, 10, 238 CrossRef PubMed.
- Isocyanate index is calculated as the molar ratio of isocyanate to hydroxyl groups in the formulation and expressed as such: Index = (MolNCO/MolOH) × 100.
- A. J. Kosanovich, O. Jalife, Y. Fan, A. Shete and Y. Srivastava, Effects of polyether polyol hydroxyl equivalent weight on controlled release polyurethane coatings of urea (46-0-0), RSC Appl. Polym., 2026, 4, 284–290 RSC.
- T. Miyajima, K. Nishiyama, M. Satake, T. Tsuji and T. Synthesis, Process Development of Polyether Polyol with High Primary Hydroxyl Content Using a New Propoxylation Catalyst, Polym. J., 2015, 47, 771–778 CrossRef.
- A. J. Kosanovich, Y. N. Srivastava and O. E. Jalife Ruz, Controlled Release Fertilizer Coatings Using Hydrophilic Blends, EP4540205A1, 2025.
| This journal is © The Royal Society of Chemistry 2026 |