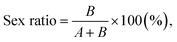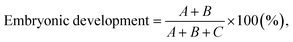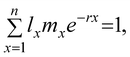Open Access Article
Open Access ArticleSublethal impacts of fragmented polyethylene nanoplastics on Daphnia magna following chronic exposure
Jinyoung
Song
a,
In Young
Kim
b,
Seonae
Hwangbo
 c,
Tae Geol
Lee
*bd and
June-Woo
Park
c,
Tae Geol
Lee
*bd and
June-Woo
Park
 *ae
*ae
aCenter for Ecotoxicology and Environmental Future Research, Korea Institute of Toxicology (KIT), Jinju, 52834, Republic of Korea
bNanobio Measurement Group, Korea Research Institute of Standards and Science (KRISS), Daejeon, 34113, Republic of Korea
cFocused Ultra-Sonic Tech. (FUST) Lab, Daejeon, 34015, Republic of Korea
dAdvanced Bio-Convergence Program, Korea University of Science and Technology (UST), Daejeon, 34113, Republic of Korea
eHuman and Environmental Toxicology Program, Korea University of Science and Technology (UST), Daejeon, 34113, Republic of Korea
First published on 2nd February 2026
Abstract
Polyethylene (PE) and fragment-shaped particles are among the most prevalent polymer types and morphologies in aquatic environments. However, chronic effects of fragmented PE nanoplastics (NPs) on aquatic organisms remain largely unexplored. We evaluated the chronic toxicity (27 days) of PE NP fragments in Daphnia magna at 0.2 mg C L−1 food level. Although D. magna ingested NP fragments, no significant changes in mortality were observed. However, high concentrations (10 and 100 µg mL−1) of NP fragments statistically decreased the number of offspring and reduced their body length. In addition, the proportions of males and stillborn offspring were statistically increased at 10 and 100 µg mL−1 of NP fragments without changing the body length of those offspring. The population growth rate of D. magna exposed to NP fragments at 1, 10, and 100 µg mL−1 was statistically lower than the control. It should be noted that the concentrations tested, particularly 10 and 100 µg mL−1, substantially exceed currently reported environmental NP concentrations. These findings provide chronic toxicity data for PE NP fragments that may contribute to NP risk assessment frameworks.
Environmental significanceNanoplastics represent an emerging environmental concern, as plastic debris continuously fragments into particles smaller than 1000 nm that can penetrate biological membranes and potentially cause adverse effects in aquatic organisms. While numerous studies have examined nanoplastic toxicity, few have assessed the chronic effects of polyethylene nanoplastic fragments, despite polyethylene and fragmented shapes being among the most prevalent polymer types and morphologies in aquatic environments. This study evaluated the chronic toxicity of polyethylene nanoplastic fragments to Daphnia magna and demonstrated significant sublethal effects, including reduced reproduction, altered offspring fitness, and decreased population growth rate at high concentrations, despite no lethal toxicity. These findings provide toxicity data for polyethylene nanoplastic fragments that can contribute to nanoplastic risk assessment, although the concentrations tested substantially exceed currently reported environmental levels. |
1. Introduction
Plastic pollution remains a pressing environmental issue, with microplastics (MPs, <5000 µm) and nanoplastics (NPs, <1 µm) increasingly recognized as emerging contaminants of global concern.1 While the ecological impacts of MPs have been extensively investigated across various organisms and exposure scenarios, knowledge regarding the toxicity of NPs remains limited.2 Despite this limited understanding, the smaller size, higher surface reactivity, and potential for tissue penetration of NPs introduce additional layers of ecological complexity that are only beginning to be understood.3 Addressing these knowledge gaps is essential for a more complete assessment of plastic pollution impacts at the nanoscale.Evidence from MP studies demonstrates that fragment-shaped particles induce more severe adverse effects, including immobilization, oxidative stress, impaired reproduction, and increased gut retention time, compared with their spherical counterparts.4–6 These findings suggest that particle morphology plays a role in determining toxicity outcomes. Similarly, shape-dependent differences in toxicity may also occur at the nanoscale.2,7 However, direct evidence for shape-dependent toxicity of NPs remains limited, and further investigation is warranted. NP toxicity research has predominantly focused on spherical particles, particularly polystyrene (PS) spheres,8,9 due to their commercial availability and ease of synthesis. Polyethylene (PE), the most abundant polymer in aquatic environments,10–12 predominantly exists in irregular shapes.12,13 Ecological risks of NPs manifest at the organism level through mortality, reduced growth, and reproductive alteration in aquatic organisms.14–16
To address the limited data on PE NP fragment toxicity, the present study evaluated the chronic ecotoxicity of PE NP fragments in the freshwater crustacean Daphnia magna. We previously manufactured these PE NP fragments using a focused ultrasonic system that incorporated both physical (cavitation and collapse) and chemical (UV weathering) processes to simulate natural degradation.17D. magna was selected as the test organism due to its sensitivity to toxicants, wide distribution in the Northern Hemisphere freshwater, and short generation time. To identify chronic effects of PE NP fragments, D. magna was exposed to the NPs for 21 days across a concentration gradient (0.1–10 µg mL−1). Lethal (survival rate) and sublethal endpoints (number of offspring, time to first brood, developmental ratio, sex ratio, and body length) were evaluated. The results provide chronic toxicity data for PE NPs and contribute to the NP ecotoxicology literature, facilitating environmental risk assessments.
2. Materials and methods
2.1. PE NP fragments
PE NP fragments used in this study were manufactured following the methods described by Hwangbo et al.17 Fluorescent green high-density PE beads (850–1000 µm; Cospheric LLC, CA, USA) were fragmented using a cylindrically focused ultrasonic system (400 kHz frequency and 150 W energy output) to nanosized plastics (means with a standard deviation of 85.14 ± 5.37 nm; range of 25–350 nm). The ultrasonic irradiation for 3 hours induced cavitation and sonoluminescence, including UV wavelengths, which resulted in surface oxidation and mechanical fragmentation of the PE beads. Unlike spherical NPs commonly used in toxicity studies, these fragments exhibited irregular and rock-like rounded shapes as confirmed by transmission electron microscopy (TEM) and scanning electron microscopy (SEM). Fourier-transform infrared spectroscopy (FT-IR) analysis revealed that the fragmentation process introduced oxygen-containing functional groups, including OH and C–O bonds, on the particle surfaces. This indicated surface oxidation typical of environmentally weathered plastics. These physicochemical characteristics have been detailed in our previous study.172.2. Chronic toxicity tests of PE NP fragments
D. magna used were sourced from the National Institute of Environmental Research (NIER), Republic of Korea. The organisms were cultured in two 1 L glass beakers containing M4 medium (means with a standard deviation of pH 7.8 ± 0.1; hardness 250 mg CaCO3 L−1). Adult females, synchronized in their reproductive cycles, were placed at a density of 20 individuals per beaker. The medium was renewed daily, and the daphnids were fed Chlorella vulgaris (2.0 mg carbon L−1 day−1). Neonates from the third brood were collected for subsequent experiments.The chronic toxicity test (27 days) was conducted using D. magna (<24 h old) at 20 °C under a 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 h light:dark cycle. One D. magna was added to a 50 mL glass beaker containing 50 mL of M4 medium, with ten replicates for each concentration of PE NP fragments [0.1, 1, 10, and 100 µg mL−1, including the control (M4 medium)]. With renewal of M4 medium every two days, Chlorella vulgaris was fed daily at 0.2 mg carbon L−1 to minimize potential food-mediated interference with NP toxicity detection. Carbon content was calculated using a conversion factor for freshwater green algae18 as follows:
8 h light:dark cycle. One D. magna was added to a 50 mL glass beaker containing 50 mL of M4 medium, with ten replicates for each concentration of PE NP fragments [0.1, 1, 10, and 100 µg mL−1, including the control (M4 medium)]. With renewal of M4 medium every two days, Chlorella vulgaris was fed daily at 0.2 mg carbon L−1 to minimize potential food-mediated interference with NP toxicity detection. Carbon content was calculated using a conversion factor for freshwater green algae18 as follows:
| Carbon content = 0.1204 × V1.051 (mg carbon cell−1), |
where A, B, and C refer to the number of females, males, and stillbirths, respectively. The body lengths of the neonates were measured immediately after all offspring from each brood had been released, and adult body lengths were recorded at the end of the test. Images of the PE NP fragments in D. magna were analyzed using an inverted fluorescence microscope (EVOS, Thermo Fisher Scientific, Waltham, MA, USA). The body length of D. magna was measured from immediately above the eyespot to the base of the spine. Population growth rates were calculated using the Euler–Lotka equation:19
where lx represents the fraction of individual D. magna surviving until age x, mx represents the number of offspring per surviving female D. magna between the ages x and x + 1, and r represents the population growth rate. Finally, the effective concentrations causing 10 and 50% effects (EC10 and EC50, respectively) on sublethal endpoints were calculated using nonlinear regression analyses in CETIS (v.1.8.7; Tidepool Scientific Software).
2.3. Statistical analyses
All statistical analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA). Daily survival rates were compared using the log-rank test with the Kaplan–Meier procedure. Statistical differences among the concentrations of PE NP fragments were analyzed using one-way analysis of variance (ANOVA) followed by post-hoc Tukey's honestly significant difference test, and the Kruskal–Wallis test followed by the post-hoc Mann–Whitney test with Bonferroni correction for parametric and non-parametric data, respectively. Data were checked for normality of distribution using the Shapiro–Wilk test and homogeneity of variance using Levene's test. For all statistical tests, the significance level was set at α = 0.05. Statistical differences were determined when p < α. Data were described as the mean ± standard deviation (SD). Figures are presented as box-and-whisker plots, displaying the interquartile range (IQR, the box), mean (×symbol), median (horizontal bar within the box) and whiskers extending to 1.5 times the IQR. Outliers are shown as individual points.3. Results
3.1. Lethal toxicity of PE NP fragments
Survival of D. magna remained high across all PE NP fragment concentrations during the 27-day exposure, with only one mortality observed at 100 µg mL−1 on day 19 (Fig. 1A). No statistical differences (p > 0.05) in survival were observed between treatments. Despite the ingestion of PE NP fragments by D. magna (Fig. 1B), the particles did not induce lethal toxicity.3.2. Sublethal toxicity of PE NP fragments
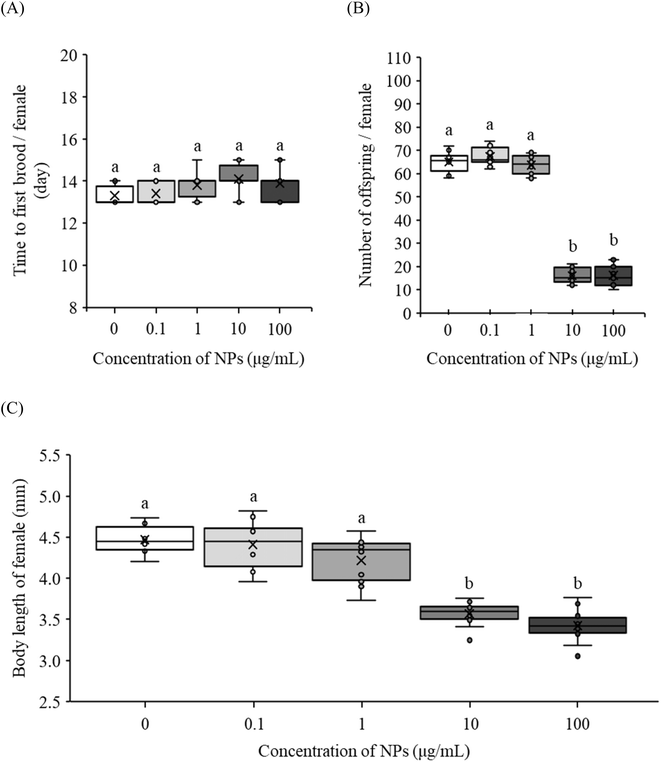
The PE NP fragments did not delay the time to first brood at any concentration (Fig. 2A) with EC10 and EC50 values exceeding 100 µg mL−1 (Table 1). However, the number of offspring was the most sensitive endpoint, with EC10 and EC50 values of 1.32 and 4.98 µg mL−1 (Table 1). However, the number of offspring and body length of adults statistically decreased (p < 0.05) at higher concentrations (10 and 100 µg mL−1) compared to the control (Fig. 2B and C).| Endpoints | EC10 (µg mL−1) with 95% CI | EC50 (µg mL−1) with 95% CI |
|---|---|---|
| Time to first brood | >100 | >100 |
| Number of offspring per female | 1.32 (1.10–1.51) | 4.98 (4.65–5.40) |
| Embryonic development | 3.66 (2.80–6.40) | >100 |
High concentrations (10 and 100 µg mL−1) of PE NP fragments statistically altered the sex ratio (Fig. 3A) and impaired embryonic development of offspring (Fig. 3B). For embryonic development, EC10 was 3.66 µg mL−1, while the EC50 exceeded 100 µg mL−1 (Table 1). These concentrations statistically increased (p < 0.05) male offspring production to 18.4 ± 13.8 and 14.8 ± 7.6%, respectively, compared to the control (0.3 ± 0.6%). Stillborn offspring production also statistically increased (p < 0.05) to 13.6 ± 10.1% and 18.7 ± 14.4% at 10 and 100 µg mL−1, respectively, compared to the control (0.3 ± 1.0%). Despite low embryonic development, well-developed offspring showed no statistical changes (p > 0.05) in body length with any concentration of PE NP fragments (Fig. 3C). The population growth rate, which integrates the effects on the survival and reproductive outputs of female D. magna, was altered at most concentrations of NP fragments (Fig. 4).
4. Discussion
4.1. Lethal toxicity of PE NP fragments
The low mortality observed in our study may be attributed to the behavior of PE NP fragments in the water column. Although HDPE had a density (0.99–1.01 g cm−3) close to that of water, visual observations revealed that agglomerated particles gathered at the water surface (Fig. S1). This surface accumulation potentially limited contact between particles and D. magna in the water column. Supporting our results, Coady et al.20 reported no statistically significant mortality in D. magna exposed to ethylene acrylic acid copolymer (0.92 g cm−3) NP beads (103 nm; 0.46 and 4.6 µg mL−1) compared to the control for 21 days. In contrast, several studies have reported statistical mortality in D. magna exposed to PS (1.04–1.07 g cm−3) NP beads.21–23 These divergent outcomes in mortality may be attributed to polymer density differences. PS is denser than water and settles more readily in the water column, potentially increasing ingestion rates and lethal toxicity compared to PE.244.2. Sublethal toxicity of PE NP fragments
Similar reproductive and growth impairments have been reported in daphnids exposed to PS NP beads for 21 days.16,25,26 For instance, Liu et al.26 observed that PS NP beads (75 nm) statistically decreased the number of offspring and body length of adult Daphnia pulex at low concentrations (0.1, 0.5, 1, and 2 µg mL−1). In comparison, our study with PE NP fragments showed adverse effects only at relatively higher concentrations (10 and 100 µg mL−1). This difference in effective concentrations may be attributed to multiple factors that distinguish these studies: (1) polymer density, with PS (1.05 g cm−3) exhibiting better suspension than HDPE (0.99–1.01 g cm−3);24 (2) particle size distribution, with monodisperse PS beads being more efficiently ingested than polydisperse PE fragments (25–350 nm);27 and (3) species sensitivity, as D. pulex may be more sensitive than D. magna.28 The mechanisms underlying these reproductive and growth impairments may involve oxidative stress responses.16,26 Sanpradit et al.16 demonstrated that reproductive capacity of D. magna was impaired by oxidative stress induced by PS NP bead exposure (50 nm).Zhang et al.29 demonstrated that NPs upregulated sex determining genes (DMRTA and DMRT1), statistically altering the sex ratio by increasing male offspring production in D. pulex exposed to PS NPs (75 nm; 1 µg mL−1) for 21 days. Furthermore, Cui et al.30 ascertained that Daphnia galeata exposed to PS NPs (52 ± 5 nm; 5 µg mL−1) for 5 days stored significantly fewer and smaller lipid droplets in their bodies than the control, resulting in extremely high stillbirth rates (83 ± 25%).
Two studies have reported reduced body size of offspring D. magna exposed to PS NP beads for 21 days, suggesting different reproductive strategies.16,25 Sanpradit et al.16 reported that NP exposure resulted in fewer and smaller offspring. In contrast, Besseling et al.25 reported a trade-off between clutch size and offspring size, with a greater number of smaller offspring being released. In this study, PE NP fragment exposure at 10 µg mL−1 resulted in a reduced number of offspring across all broods, indicating reproductive stress. Notably, the second brood exhibited fewer but longer offspring (Fig. S2), while other broods showed reduced numbers without changes in body length (data not shown). However, body length provides limited information on offspring quality, as it does not account for body width or mass. Therefore, body width or dry weight measurements would be needed to determine whether this second brood pattern represents a reproductive trade-off or stress-induced morphological alteration.
Except for 0.1 µg mL−1, all concentrations (1, 10, and 100 µg mL−1) of PE NP fragments statistically decreased (p < 0.05) the population growth rate of D. magna compared to the control. Previous studies have reported population growth rate reductions in daphnids exposed to PS NP beads for 21 days. Liu et al.14 found statistical effects in D. pulex at 2 µg mL−1 (71.18 nm) compared to the control. Lyu et al.22 observed a statistically reduced population growth rate in D. magna at 1 and 2 µg mL−1 (70 nm) at standard food levels (1.5 mg C L−1) compared to the control, with similar effects at 2 µg mL−1 under low food conditions (0.3 mg C L−1).
To evaluate the environmental risk of PE NP fragments, we applied the GHS (UN Globally Harmonized System) classification criteria for long-term aquatic hazards.31 Both EC10 values for the number of offspring and embryonic development exceeded the threshold for Category Chronic 2 (≤1 mg L−1), indicating that PE NP fragments do not meet the criteria for GHS chronic hazard classification (Category Chronic 1: ≤0.1 mg L; Category Chronic 2: ≤1 mg L−1). However, it is important to note several experimental considerations that may influence the interpretation of these results. In the present study, we did not follow OECD TG NO. 211 guidelines,32 as food levels (0.2 mg C L−1) were intentionally reduced to minimize potential interference with NP toxicity detection. However, this approach may have influenced the ability of D. magna to withstand potential stress from NP exposure. Lyu et al.22 demonstrated that at low food levels (0.3 mg C L−1), toxic effects of NPs on reproduction may be partially masked because organisms have already reached minimal reproductive output. Similarly, Sun et al.33 showed that below 0.3 mg C L−1 food levels, the negative effects of ZnO nanoparticles on survival time and reproduction of D. magna were masked by food limitation. Under standard food conditions, D. magna may exhibit greater sensitivity to NP exposure, potentially yielding lower EC10 values.
4.3. Implications and limitations
Numerous studies have assessed the physiological effects of NPs on D. magna (Table 2), focusing on PS beads. Few studies have investigated the chronic toxicity of PE NPs with fragmented shapes in daphnids. Ekvall et al.34 reported that HDPE (0.97 g cm−3) NP fragments (112 ± 4 nm) statistically decreased survival of D. magna for 134 days. This study used commercial packaging material mixed with tap water and fragmented with a hand-held food blender, which was filtered using an 800 nm syringe filter. The filtrate contained all molecules and materials that could pass through the filter. Thus, the concentrations and additives of NPs were unspecified, indicating that the properties of NPs are toxic to D. magna. Additionally, Monikh et al.35 investigated the reproductive toxicity of PE (50 nm; 0.00025 µg mL−1), PP (50 nm; 0.00022 µg mL−1), PS (200 and 600 nm; 0.13 and 3.5 µg mL−1, respectively), and PVC (200 nm; 0.17 µg mL−1) NPs in D. magna for 21 days. All NPs statistically altered the number of offspring in the fourth and fifth broods of D. magna, whereas earlier broods remained largely unaffected. However, the authors focused on characterizing the changes in NPs caused by co-occurring benzo(α)pyrene and on toxicity to Danio rerio. While the NP morphology was not explicitly stated in the paper, microscopic images revealed fragmented shapes. Toxicological studies (Table 2) have used different NP materials, sometimes without providing specific information on their characteristics, which makes comparing the toxic effects of NPs across studies challenging. Sørensen et al.36 suggested that test materials should incorporate environmental weathering processes and physicochemical characteristics to enhance environmental relevance.| NPs | Species | Exposure duration (day) | Effects | References | |||
|---|---|---|---|---|---|---|---|
| Shape | Polymer | Size (nm) | Conc. (µg mL−1) | ||||
| a HDPE: high density polyethylene; f: fluorescence; EAA: ethylene acrylic acid copolymer; PS: polystyrene; PE: polyethylene; PP: polypropylene; PVC: polyvinyl chloride. | |||||||
| Fragment | HDPE(f) | 85.14 ± 5.37 (25–350) | 0.1, 1, 10, 100 | D. magna | 27 | Number of offspring/female: ↓ (10, 100) | This study |
| Body length of adult: ↓ (10, 100) | |||||||
| Sex ratio: ↑ (10, 100) | |||||||
| Embryonic development: ↓ (10, 100) | |||||||
| Population growth rate: ↓ (1, 10, 100) | |||||||
| Survival, time to 1st brood, number of brood, body length of offspring: no significant changes | |||||||
| HDPE | 112 ± 4 (90–200) | 0.53 | D. magna | 134 | Survival: ↓ | 34 | |
| Number of offspring/female: no significant change | |||||||
| Bead | EAA | 103 | 0.46 and 4.6 | D. magna | 21 | Survival, number of offspring/female, body length of adult: no significant change | 20 |
| PS-COOH(f) | 20 | 0.1, 50 | D. magna | 21 | Time to 1st brood: ↑ (50) | 15 | |
| Body length of adult: ↓ | |||||||
| Number of offspring/female: no significant change | |||||||
| 200 | 50 | D. magna | 21 | Body length of adult: ↓ | |||
| Time to 1st brood: no significant change | |||||||
| PS(f) | 100 | 0.1, 0.5, 1 | D. magna | 21 | Number of offspring/female, time to 1st brood, number of brood, body length of adult: no significant changes | 42 | |
| 94.5 ± 3.9 | 1.7, 3.5, 7.1, 14.3, 28.6 | D. magna | 21 | Survival: ↓ (14.3, 28.6) | 23 | ||
| Time to 1st brood: ↑ (14.3) | |||||||
| Body length of adult: ↓ (14.3) | |||||||
| PS | 90.1 ± 4.9 | 1.6, 3.3, 6.6, 13.3, 26.6 | D. magna | 21 | Survival: ↓ (26.6) | ||
| Time to 1st brood: ↑ (13.3, 26.6) | |||||||
| Body length of adult: ↓ (26.6) | |||||||
| 70 | 1, 2 | D. magna | 27 | Survival: ↓ | 22 | ||
| Number of offspring/female: ↓ | |||||||
| 50 | 1, 2 | D. magna | 21 | Number of offspring/female: ↓ | 16 | ||
| - Body length of adult: ↓ (2) | |||||||
| Body length of offspring: ↓(1st brood) | |||||||
| Time to 1st brood: no significant change | |||||||
| 50 | 5 | D. magna | 21 | Survival: ↓ | 21 | ||
| Time to 1st brood: ↑ | |||||||
| Number of brood: ↓ | |||||||
| Number of offspring/female: no significant change | |||||||
| 72.84 ± 6.81 | 1.65, 3.31, 6.62, 13.24 | D. magna | 21 | Survival, time to 1st brood, body length of adult: no significant changes | 43 | ||
| 71.18 | 0.1, 0.5, 1, 2 | D. pulex | 21 | Number of offspring/20 females: ↓ (0.5, 2) | 14 | ||
| 71.18 | 0.001 | D. pulex | 21 | Number of offspring/female, number of brood, body length of adult: no significant changes | 37 | ||
| 75 | 0.1, 0.5, 1, 2 | D. pulex | 21 | Number of offspring/female: ↓ | 26 | ||
| Time to 1st brood: ↑ | |||||||
| Number of brood: ↓ | |||||||
| Body length of adult: ↓ | |||||||
| Unclear | PE | 50 ± 10 | 0.00025 | D. magna | 21 | Number of offspring/female: ↓ (5th brood) | 35 |
| Survival: no significant change | |||||||
| PP | 53 ± 5 | 0.00022 | D. magna | 21 | Number of offspring/female: ↑ (5th brood) | ||
| Survival: no significant change | |||||||
| PS | 207 ± 12 | 0.13 | D. magna | 21 | Number of offspring/female: ↑ (4th brood), ↓ (5th brood) | ||
| Survival: no significant change | |||||||
| 650 ± 35 | 3.5 | D. magna | 21 | Number of offspring/female: ↑ (5th brood) | |||
| Survival: no significant change | |||||||
| PVC | 241 ± 28 | 0.17 | D. magna | 21 | Number of offspring/female: ↑ (5th brood) | ||
| Survival: no significant change | |||||||
| PS | 75 | 1 | D. pulex | 21 | Sex ratio: ↑ | 29 | |
Cross-study comparisons are further complicated by variability in experimental protocols among published NP toxicity studies. Although most NP toxicity studies listed in Table 2 report adherence to OECD TG NO. 211 guidelines,32 several studies have modified the standard chronic test protocols by altering test vessel volume per organism,15,21,22 conducting group rather than individual exposures,14,22 or extending test duration.22,34 Additionally, some studies did not follow standardized chronic toxicity protocols,14,26,34,37 including the present study. These methodological differences should be considered when comparing toxicity results across studies.
The present study evaluated the chronic toxicity of PE NP fragments to the freshwater cladoceran D. magna, using materials representing one of the predominant polymer types and morphologies observed in natural aquatic environments.12 Although no significant mortality was observed, this study revealed sublethal toxicities, including reduced reproduction and altered offspring sex ratio at high concentrations (10 and 100 µg mL−1), indicating potential population-level impacts. The concentrations tested in this study require careful consideration regarding environmental relevance. Materić et al.10 reported NP concentrations ranging from 24 to 1588 µg L−1 in freshwater environments. The lower concentrations tested in this study (0.1 and 1 µg mL−1) fall within the range of heavily contaminated sites, while the higher concentrations (10 and 100 µg mL−1) substantially exceed currently reported environmental levels. Several factors suggest that actual environmental NP concentrations may be underestimated: (1) current detection methods have limited sensitivity for sub-100 nm particles,38 and (2) continued growth in plastic production and environmental accumulation may lead to elevated concentrations in the future.39 Conversely, environmental NP concentrations may be overestimated due to analytical limitations in distinguishing plastic from other nanoparticles40 and laboratory contamination during sample processing.41 Given these methodological uncertainties in environmental NP quantification, the lack of significant effects at lower concentrations in this study should be considered in the context of environmental risk assessment.
Although MP studies have demonstrated that the particle shape affects toxicity outcomes, direct evidence for shape-dependent toxicity at the nanoscale remains limited. The present study used fragmented NPs but did not include a direct comparison with spherical particles under identical experimental conditions. Therefore, it is not possible to determine whether the observed effects are attributable to particle shape or other factors such as polymer type and particle size. In addition, the toxicity mechanisms were not investigated in the present study. Future studies should address these limitations to better understand NP toxicity in aquatic organisms.
5. Conclusions
This study provides chronic toxicity data for PE NP fragments using well-characterized particles with food levels at 0.2 mg C L−1. In a 27-day exposure study with D. magna, we found that PE NP fragments caused no mortality but induced significant sublethal effects, including reduced offspring production (10 and 100 µg mL−1), decreased adult body length (10 and 100 µg mL−1), and diminished population growth rates (1, 10, and 100 µg mL−1). These findings demonstrate that PE NPs can impair life-history parameters and population growth rates of D. magna inhabiting environments with limited food availability, where concentrations of PENPs are higher than 1 µg mL−1. These concentrations substantially exceed currently reported environmental levels, and the sublethal effects should be interpreted within this context. Future investigations should examine cellular and molecular mechanisms and consider environmentally relevant concentration ranges.Author contributions
Jinyoung Song: investigation, data curation, validation, writing – original draft. In Young Kim: investigation, data curation, validation. Seonae Hwangbo: formal analysis, methodology. Tae Geol Lee: conceptualization, supervision, writing – review & editing. June-Woo Park: supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that the data supporting the findings of this study are available within the article.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5va00329f.
Acknowledgements
This study was supported by the Nano & Material Technology Development Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (RS-2024-00452934); a grant from the National Research Council of Science & Technology (NST) funded by the Korea government (MSIT) (No. CAP20025-000); a research fund of the Korea Research Institute of Standards and Science (KRISS-2025-GP2025-0007); and the Korea Institute of Toxicology, Republic of Korea (KK-2510).References
- S. Sharma, T. Barman, G. Mishra and A. Kumar, Micro and nanoplastics pollution: A review on global concern and its impacts on ecosystems, Land Degrad. Dev., 2024, 35(9), 2963–2981 CrossRef.
- R. Thakur, V. Joshi, G. C. Sahoo, N. Jindal, R. R. Tiwari and S. Rana, Review of mechanisms and impacts of nanoplastic toxicity in aquatic organisms and potential impacts on human health, Toxicol Rep, 2025, 102013 CrossRef PubMed.
- M. Hu and D. Palić, Micro-and nano-plastics activation of oxidative and inflammatory adverse outcome pathways, Redox Biol., 2020, 37, 101620 CrossRef CAS PubMed.
- D. An, J. Na, J. Song and J. Jung, Size-dependent chronic toxicity of fragmented polyethylene microplastics to Daphnia magna, Chemosphere, 2021, 271, 129591 CrossRef CAS PubMed.
- S. A. Lee, M. Esterhuizen, Y. Kim, M. Kim and Y. J. Kim, Assessing the acute differential toxicity of polystyrene microplastic particles and comparing the impacts of bead-shaped versus fragmented particles on Daphnia magna, Appl. Biol. Chem., 2025, 68(1), 34 CrossRef CAS.
- M. Ogonowski, C. Schür, Å. Jarsén and E. Gorokhova, The effects of natural and anthropogenic microparticles on individual fitness in Daphnia magna, PloS One, 2016, 11(5), e0155063 CrossRef PubMed.
- F. Jahedi and N. J. H. Fard, Micro-and nanoplastic toxicity in humans: Exposure pathways, cellular effects, and mitigation strategies, Toxicol Rep, 2025, 102043 Search PubMed.
- O. Pikuda, E. Roubeau Dumont, Q. Chen, J.-R. Macairan, S. A. Robinson, D. Berk and N. Tufenkji, Toxicity of microplastics and nanoplastics to Daphnia magna: Current status, knowledge gaps and future directions, Trac-Trends Anal. Chem., 2023, 167, 117208 CrossRef CAS.
- K. Pelegrini, T. C. B. Pereira, T. G. Maraschin, L. D. S. Teodoro, N. R. D. S. Basso, G. L. B. De Galland, R. A. Ligabue and M. R. Bogo, Micro-and nanoplastic toxicity: A review on size, type, source, and test-organism implications, Sci. Total Environ., 2023, 878, 162954 Search PubMed.
- D. Materić, M. Peacock, J. Dean, M. Futter, T. Maximov, F. Moldan, T. Röckmann and R. Holzinger, Presence of nanoplastics in rural and remote surface waters, Environ. Res. Lett., 2022, 17, 054036 Search PubMed.
- A. E. Schwarz, T. N. Ligthart, E. Boukris and T. Van Harmelen, Sources, transport, and accumulation of different types of plastic litter in aquatic environments: a review study, Mar. Pollut. Bull., 2019, 143, 92–100 Search PubMed.
- Y. Wu, J. Yang, Z. Li, H. He, Y. Wang, H. Wu, L. Xie, D. Chen and L. Wang, How does bivalve size influence microplastics accumulation?, Environ. Res., 2022, 214, 113847 Search PubMed.
- S. Moon, L. M. Martin, S. Kim, Q. Zhang, R. Zhang, W. Xu and T. Luo, Direct observation and identification of nanoplastics in ocean water, Sci. Adv., 2024, 10(4), eadh1675 Search PubMed.
- Z. Liu, Y. Li, M. S. Sepúlveda, Q. Jiang, Y. Jiao, Q. Chen, Y. Huang, J. Tian and Y. Zhao, Development of an adverse outcome pathway for nanoplastic toxicity in Daphnia pulex using proteomics, Sci. Total Environ., 2021, 766, 144249 Search PubMed.
- O. Pikuda, E. R. Dumont, S. Matthews, E. G. Xu, D. Berk and N. Tufenkji, Sub-lethal effects of nanoplastics upon chronic exposure to Daphnia magna, J. Hazard. Mater. Adv., 2022, 7, 100136 Search PubMed.
- P. Sanpradit, E. Byeon, J.-S. Lee, H. Jeong, H. S. Kim, S. Peerakietkhajorn and J.-S. Lee, Combined effects of nanoplastics and elevated temperature in the freshwater water flea Daphnia magna, J. Hazard. Mater., 2024, 465, 133325–133474 CrossRef CAS PubMed.
- S. Hwangbo, I. Y. Kim, K. Ko, K. Park, J. Hong, G. Kang, J.-S. Wi, J. Kim and T. G. Lee, Preparation of fragmented polyethylene nanoplastics using a focused ultrasonic system and assessment of their cytotoxic effects on human cells, Environ. Pollut., 2024, 362, 125009 CrossRef CAS PubMed.
- O. Rocha and A. Duncan, The relationship between cell carbon and cell volume in freshwater algal species used in zooplanktonic studies, J. Plankton Res., 1985, 7(2), 279–294 CrossRef.
- C. Hernandez-Suarez and J. Rabinovich, Exact confidence intervals for population growth rate, longevity and generation time, Theor. Popul. Biol., 2024, 155, 1–9 Search PubMed.
- K. K. Coady, L. Burgoon, C. Doskey and J. W. Davis, Assessment of transcriptomic and apical responses of Daphnia magna exposed to a polyethylene microplastic in a 21-d chronic study, Environ. Toxicol. Chem., 2020, 39, 1578–1589 CrossRef CAS PubMed.
- Y. Lee, D.-H. Kim, J.-S. Lee, H. S. Kim, P. Maszczyk, M. Wang, Z. Yang, D.-Z. Wang and J.-S. Lee, Combined exposure to hypoxia and nanoplastics leads to negative synergistic oxidative stress-mediated effects in the water flea Daphnia magna, Mar. Pollut. Bull., 2024, 202, 116306 CrossRef CAS PubMed.
- K. Lyu, B. Yu, D. Li, L. Gu and Z. Yang, Increased food availability reducing the harmful effects of microplastics strongly depends on the size of microplastics, J. Hazard. Mater., 2022, 437, 129375 CrossRef PubMed.
- D. S. Vicentini, D. J. Nogueira, S. P. Melegari, M. Arl, J. S. Köerich, L. Cruz, N. M. Justino, B. V. Oscar, R. C. Puerari, M. L. N. da Silva and C. Simioni, Toxicological evaluation and quantification of ingested metal-core nanoplastic by Daphnia magna through fluorescence and inductively coupled plasma-mass spectrometric methods, Environ. Toxicol. Chem., 2019, 38(10), 2101–2110 CrossRef CAS PubMed.
- A. Luangrath, J. Na, P. Kalimuthu, J. Song, C. Kim and J. Jung, Ecotoxicity of polylactic acid microplastic fragments to Daphnia magna and the effect of ultraviolet weathering, Ecotox. Environ. Safe., 2024, 271, 115974 CrossRef CAS PubMed.
- E. Besseling, B. Wang, M. Lürling and A. A. Koelmans, Nanoplastic Affects Growth of S. obliquus and Reproduction of D. magna, Environ. Sci. Technol., 2014, 48, 12336–12343 CrossRef CAS PubMed.
- Z. Liu, P. Yu, M. Cai, D. Wu, M. Zhang, Y. Huang and Y. Zhao, Polystyrene nanoplastic exposure induces immobilization, reproduction, and stress defense in the freshwater cladoceran Daphnia pulex, Chemosphere, 2019, 215, 74–81 CrossRef CAS PubMed.
- P. Rosenkranz, Q. Chaudhry, V. Stone and T. F. Fernandes, A comparison of nanoparticle and fine particle uptake by Daphnia magna, Environ. Toxicol. Chem., 2009, 28(10), 2142–2149 CrossRef CAS PubMed.
- G. Jaikumar, N. R. Brun, M. G. Vijver and T. Bosker, Reproductive toxicity of primary and secondary microplastics to three cladocerans during chronic exposure, Environ. pollut., 2019, 249, 638–646 Search PubMed.
- W. Zhang, Z. Liu, S. Tang, D. Li, Q. Jiang and T. Zhang, Transcriptional response provides insights into the effect of chronic polystyrene nanoplastic exposure on Daphnia pulex, Chemosphere, 2020, 238, 124563 Search PubMed.
- R. Cui, S. W. Kim and Y.-J. An, Polystyrene nanoplastics inhibit reproduction and induce abnormal embryonic development in the freshwater crustacean Daphnia galeata, Sci. Rep., 2017, 7(1), 12095 CrossRef PubMed.
- UN, Globally Harmonized System of Classification and Labelling of Chemicals (GHS), 2023 Search PubMed.
- OECD, Test Guideline No. 211: Daphnia Magna Reproduction Test, 2012 Search PubMed.
- Y. Sun, Q. Liu, J. Huang, D. Li, Y. Huang, K. Lyu and Z. Yang, Food abundance mediates the harmful effects of ZnO nanoparticles on development and early reproductive performance of Daphnia magna, Ecotox. Environ. Safe., 2022, 236, 113475 CrossRef CAS PubMed.
- M. T. Ekvall, I. Gimskog, J. Hua, E. Kelpsiene, M. Lundqvist and T. Cedervall, Size fractionation of high-density polyethylene breakdown nanoplastics reveals different toxic response in Daphnia magna, Sci. Rep., 2022, 12(1), 3109 Search PubMed.
- F. A. Monikh, M. Durão, P. V. Kipriianov, H. Huuskonen, J. Kekäläinen, S. Uusi-Heikkilä, E. Uurasjärvi, J. Akkanen and R. Kortet, Chemical composition and particle size influence the toxicity of nanoscale plastic debris and their co-occurring benzo(α)pyrene in the model aquatic organisms Daphnia magna and Danio rerio, NanoImpact, 2022, 25, 100382 CrossRef CAS PubMed.
- L. Sørensen, M. H. Gerace and A. M. Booth, Small micro-and nanoplastic test and reference materials for research: Current status and future needs, Camb. prism., Plast., 2024, 2, e13 CrossRef.
- Z. Liu, M. Cai, D. Wu, P. Yu, Y. Jiao, Q. Jiang and Y. Zhao, Effects of nanoplastics at predicted environmental concentration on Daphnia pulex after exposure through multiple generations, Environ. Pollut., 2020, 256, 113506 CrossRef CAS PubMed.
- M. Kaur, C. T. Gibson, S. Miller, S. C. Leterme and M. Macgregor, A physical chemistry lens on environmental nanoplastics analysis challenges. Part II: Detection techniques-principles, limitations and future directions, Environ. Sci.-Nano, 2026 10.1039/D5EN00630A.
- L. Lebreton and A. Andrady, Future scenarios of global plastic waste generation and disposal, Palgr. Commun., 2019, 5(1), 1–11 Search PubMed.
- R. J. Peters, E. Relou, E. L. Sijtsma and A. K. Undas, Evaluation of the performance of Nanoparticle Tracking Analysis (NTA) for the measurement of nanoplastics, Food Safety and Risk, 2025, 12(1), 2 CrossRef.
- N. R. Jones, A. M. de Jersey, J. L. Lavers, T. Rodemann and J. Rivers-Auty, Identifying laboratory sources of microplastic and nanoplastic contamination from the air, water, and consumables, J. Hazard. Mater., 2024, 465, 133276 Search PubMed.
- S. Rist, A. Baun and N. B. Hartmann, Ingestion of micro-and nanoplastics in Daphnia magna–Quantification of body burdens and assessment of feeding rates and reproduction, Environ. Pollut., 2017, 228, 398–407 Search PubMed.
- D. J. Nogueira, A. C. D. O. da Silva, M. L. N. da Silva, D. S. Vicentini and W. G. Matias, Individual and combined multigenerational effects induced by polystyrene nanoplastic and glyphosate in Daphnia magna (Strauss, 1820), Sci. Total Environ., 2022, 811, 151360 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |